Advanced Aryltriazolinone Manufacturing: Scalable Routes for Global Herbicide Production
Advanced Aryltriazolinone Manufacturing: Scalable Routes for Global Herbicide Production
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical heterocyclic intermediates. Patent CN1927827A introduces a transformative methodology for preparing 1-aryltriazolinones of formula (I), which serve as indispensable precursors in the production of commercial herbicides. This intellectual property outlines a dual-pathway strategy involving either the carbonylation of an amidrazone or the condensation of a hydrazonoyl derivative, both offering superior yield and purity profiles compared to legacy techniques. By leveraging these novel chemical transformations, manufacturers can achieve significant process intensification, reducing waste and enhancing the economic viability of large-scale agrochemical production. The structural versatility of the intermediates allows for the incorporation of various substituents, including halogens and nitro groups, ensuring broad applicability across different herbicide families.
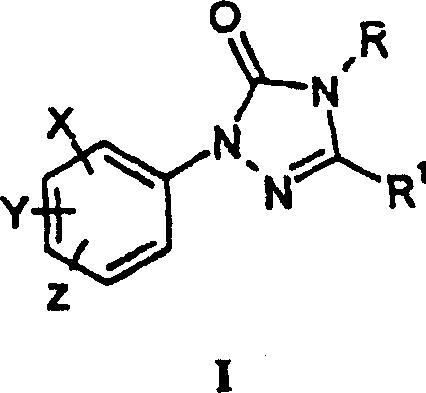
For procurement specialists and supply chain managers, understanding the underlying chemistry of these intermediates is crucial for securing a reliable agrochemical intermediate supplier. The patent details specific embodiments where substituents such as chlorine, fluorine, and methylsulfonylamino groups are strategically positioned to optimize biological activity. This level of molecular precision requires a manufacturing partner capable of handling complex halogenated substrates with rigorous quality control. The ability to produce these compounds with unexpectedly high yield and purity directly impacts the downstream formulation stability and efficacy of the final herbicide product, making the choice of synthesis route a critical strategic decision for any agrochemical enterprise seeking cost reduction in herbicide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 1-aryltriazolinones has been plagued by inefficient multi-step sequences that detrimentally impact overall throughput and cost structures. Traditional methods often necessitate the initial formation of a 1-aryl triazole alkane ketone ring, which must subsequently undergo further chemical modification to yield the desired triazolinone core. This indirect approach introduces unnecessary unit operations, each carrying its own risks of yield loss, impurity generation, and solvent consumption. Furthermore, the formation of by-products during these extended sequences complicates purification protocols, often requiring energy-intensive recrystallization or chromatography steps to meet the stringent purity specifications demanded by the pharmaceutical and agrochemical industries. The cumulative effect of these inefficiencies results in higher production costs and longer lead times, creating bottlenecks in the supply chain for high-purity agrochemical intermediates.
The Novel Approach
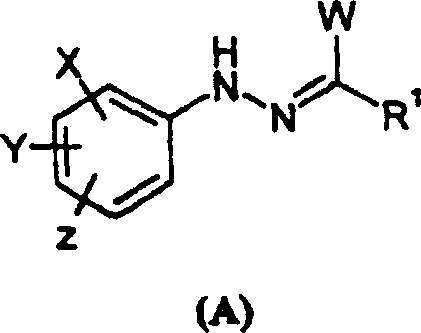
In stark contrast, the methodology disclosed in CN1927827A streamlines the synthesis by enabling the direct construction of the triazolinone ring from readily accessible precursors. The process utilizes either a carbonylation reaction of an amidrazone of formula (A) or a condensation reaction of a hydrazonoyl derivative with a ring-forming agent. This direct cyclization strategy effectively bypasses the need for intermediate ring conversion steps, thereby collapsing the synthetic timeline and reducing the overall material footprint. The use of robust reagents such as diphenyl carbonate or potassium cyanate ensures that the reaction proceeds with high atom economy. Additionally, the patent highlights the novelty of certain compounds of formula (A), which act as superior substrates for these transformations, further enhancing the selectivity of the reaction and minimizing the formation of difficult-to-remove impurities.
Mechanistic Insights into Carbonylation and Cyclization Chemistry
The mechanistic elegance of this process lies in the activation of the amidrazone or hydrazonoyl functionality towards nucleophilic attack or electrophilic substitution, depending on the specific pathway chosen. In the carbonylation route, the amidrazone nitrogen attacks the carbonyl carbon of the activating agent, such as diphenyl carbonate, facilitated by the presence of a catalyst. The patent specifies that acid catalysts like boron trifluoride or base catalysts like potassium carbonate can be employed to accelerate this cyclization. The choice of catalyst is pivotal; for instance, the use of Lewis acids can activate the carbonyl group, while basic conditions may deprotonate the amidrazone to increase its nucleophilicity. This flexibility allows chemists to tune the reaction conditions to suit the electronic nature of the substituents on the aromatic ring, ensuring consistent performance across a diverse range of substrates.
Impurity control is inherently built into the design of this synthetic route through the selection of high-purity starting materials and optimized reaction parameters. The patent emphasizes that the formation of the triazolinone ring occurs with high regioselectivity, preventing the generation of isomeric by-products that often plague heterocyclic synthesis. For example, in the condensation of hydrazonoyl chlorides with potassium cyanate, the reaction proceeds cleanly to form the desired ring system without significant side reactions. The use of specific solvents like DMAC or glyme not only solubilizes the reagents but also stabilizes the transition states, further suppressing unwanted degradation pathways. This inherent cleanliness of the reaction profile translates to simpler work-up procedures and higher isolated yields, which is a critical factor for maintaining cost competitiveness in the commercial scale-up of complex agrochemical intermediates.
How to Synthesize 1-Aryltriazolinones Efficiently
The practical execution of this synthesis involves a sequence of well-defined steps that begin with the preparation of the key hydrazine precursor. Typically, a substituted phenylhydrazine is reacted with an imidate ester or acylating agent to generate the amidrazone or hydrazonoyl intermediate. This intermediate is then subjected to the cyclization conditions described in the patent, utilizing solvents such as toluene or DMAC at temperatures ranging from ambient to 160°C. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry and processing conditions required to replicate the high yields reported in the patent examples. Adhering to these parameters is essential for achieving the reproducibility necessary for GMP-compliant manufacturing environments.
- Prepare the key amidrazone intermediate (Formula A) by reacting a substituted phenylhydrazine with an imidate ester or acylating agent in a suitable solvent like dichloromethane.
- Select an appropriate carbonylating agent such as diphenyl carbonate or a ring-forming agent like potassium cyanate depending on the specific substitution pattern of the starting material.
- Conduct the cyclization reaction in a polar aprotic solvent like DMAC or toluene at elevated temperatures (100-135°C) with optional acid or base catalysis to achieve high purity yields.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers substantial strategic benefits for organizations focused on cost reduction in herbicide manufacturing. By eliminating the need for transition metal catalysts in certain embodiments and utilizing commodity chemicals like potassium cyanate and diphenyl carbonate, the process significantly lowers the raw material cost basis. The avoidance of expensive noble metals also simplifies the purification process, as there is no need for costly heavy metal scavenging steps, which are often a regulatory and financial burden in fine chemical production. This streamlined approach allows for a more predictable cost structure, shielding the supply chain from the volatility associated with precious metal markets and complex catalyst recovery systems.
- Cost Reduction in Manufacturing: The direct cyclization methodology reduces the total number of processing steps, which directly correlates to lower labor, energy, and equipment utilization costs. By consolidating the synthesis into fewer unit operations, manufacturers can achieve higher throughput rates within existing infrastructure, effectively increasing capacity without capital expenditure. The high yields reported, such as the 96.8% yield from the hydrazonoyl chloride intermediate in Example 1, demonstrate the material efficiency of the process, ensuring that minimal raw material is wasted as by-product. This efficiency is compounded by the ability to recover and recycle solvents like toluene and ethyl acetate, further driving down the variable cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The reliance on widely available reagents such as phenylhydrazines, acyl chlorides, and inorganic salts ensures a robust and resilient supply chain. Unlike processes dependent on bespoke or scarce reagents, this methodology leverages a chemical ecosystem with multiple global suppliers, mitigating the risk of single-source bottlenecks. The stability of the intermediates, particularly the novel amidrazones described, allows for potential batch storage or transport if decentralized manufacturing strategies are employed. This flexibility empowers supply chain heads to optimize inventory levels and respond rapidly to fluctuations in market demand for the final herbicide products.
- Scalability and Environmental Compliance: The process conditions are amenable to large-scale batch or continuous processing, utilizing standard stainless steel reactors common in the fine chemical industry. The use of solvents with manageable boiling points facilitates efficient distillation and recovery, aligning with modern green chemistry principles and environmental regulations. Furthermore, the high purity of the crude product reduces the load on wastewater treatment facilities by minimizing the volume of mother liquors and wash streams containing organic impurities. This environmental compatibility is increasingly critical for maintaining operating licenses and meeting the sustainability goals of major agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this aryltriazolinone synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is vital for R&D teams planning pilot plant trials and for procurement officers negotiating supply agreements for these critical intermediates.
Q: What are the primary advantages of the novel amidrazone route over conventional triazole formation?
A: The novel route described in CN1927827A eliminates the need for forming a 1-aryl triazole alkane ketone ring as a separate preliminary step, thereby reducing the total number of synthetic operations. This direct carbonylation or condensation approach significantly minimizes by-product formation and improves overall process efficiency compared to traditional multi-step methodologies.
Q: Which solvents are most effective for the carbonylation of amidrazones in this process?
A: The patent identifies toluene, DMAC (N,N-dimethylacetamide), and glymes as particularly effective solvents. Toluene is highlighted for its ability to balance cost and solubility, while polar aprotic solvents like DMAC facilitate the dissolution of reagents and promote reaction kinetics, especially when used with catalysts like potassium fluoride.
Q: Can this synthesis method be scaled for commercial herbicide production?
A: Yes, the process is explicitly designed for commercial utility, utilizing robust reagents like diphenyl carbonate and potassium cyanate which are readily available on an industrial scale. The reaction conditions, ranging from ambient temperature to moderate heating (up to 160°C), are compatible with standard stainless steel reactor infrastructure used in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryltriazolinones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of global agrochemical portfolios. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering aryltriazolinones with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay. Our capability to handle halogenated and nitro-substituted aromatics safely and efficiently makes us an ideal partner for executing the complex chemistry described in CN1927827A.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this novel manufacturing process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain resilience and competitive positioning in the herbicide market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
