Scalable Synthesis of High-Purity 1-Bromocarbazole for Advanced Electronic Materials
The pharmaceutical and organic electronics industries are constantly seeking more efficient pathways to synthesize critical heterocyclic intermediates, and the recent disclosure in patent CN113582910B offers a transformative approach to producing 1-bromocarbazole. This specific compound serves as a pivotal building block for organic light-emitting diodes (OLEDs), hole transport materials, and various active pharmaceutical ingredients due to its unique electron-transfer characteristics. Historically, the manufacturing landscape for 1-bromocarbazole has been plagued by inefficient multi-step sequences involving hazardous reagents and complex purification protocols that drive up costs and environmental liabilities. The new methodology detailed in this patent circumvents these legacy issues by utilizing a direct lithiation strategy on a protected carbazole scaffold, achieving yields exceeding 80% and purity levels greater than 99.5%. This technical advancement represents a significant leap forward for reliable agrochemical intermediate supplier networks and fine chemical manufacturers aiming to optimize their supply chains for high-value aromatic nitrogen heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing 1-bromocarbazole have long been hindered by severe economic and environmental bottlenecks that make large-scale production challenging. One prevalent historical method involves the Suzuki coupling of 2,6-dibromonitrobenzene with phenylboronic acid followed by a Cadogan cyclization, a process heavily reliant on stoichiometric amounts of phosphorus-containing reagents such as triphenylphosphine or triethyl phosphite. As illustrated in the reaction scheme below, this pathway not only introduces toxic phosphorus waste streams that require costly disposal but also suffers from difficult byproduct control and low overall yields due to the complexity of the cyclization step.
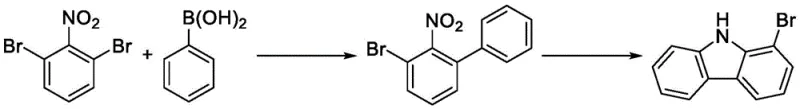
Furthermore, alternative approaches utilizing o-bromoaniline or o-bromohydrazinobenzene often necessitate expensive oxidants like DDQ for oxidative dehydrogenation, which drastically inflates the raw material costs and complicates the purification process. Other methods relying on palladium-catalyzed C-H activation or Ullmann couplings introduce heavy metal contamination risks, requiring rigorous and expensive metal scavenging steps to meet pharmaceutical grade specifications. These conventional strategies collectively result in high production costs, complex synthesis process flows, and substantial three-waste generation, creating a pressing need for a cleaner, more direct manufacturing technology that can support the growing demand for high-purity OLED material precursors.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel synthesis method disclosed in CN113582910B streamlines production into a concise three-step sequence that begins with readily available carbazole. The core innovation lies in the strategic introduction of an N-acyl protecting group, which fundamentally alters the electronic landscape of the carbazole ring to enable highly regioselective functionalization. By converting carbazole into an N-substituted intermediate, the subsequent lithiation reaction can be precisely directed to the C1 position, avoiding the thermodynamically favored but undesired C3 and C6 positions. This approach eliminates the need for transition metal catalysts and toxic phosphorus reagents entirely, replacing them with standard organolithium chemistry that is well-understood and scalable in industrial settings.
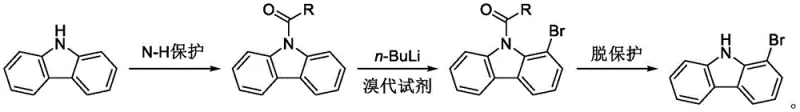
The operational simplicity of this new route is a major advantage for cost reduction in electronic chemical manufacturing, as the post-treatment involves straightforward hydrolysis and crystallization rather than complex chromatographic separations. The process demonstrates exceptional robustness, consistently delivering product purity above 99.5% with yields surpassing 80%, thereby significantly reducing the material loss associated with purification. By adopting this streamlined pathway, manufacturers can achieve substantial cost savings through reduced reagent consumption, simplified waste treatment, and higher throughput, positioning this method as the preferred choice for the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Mechanistic Insights into N-Acyl Directed Ortho Lithiation
The success of this synthesis hinges on the precise mechanistic control exerted by the N-acyl protecting group during the lithiation phase, which serves as a powerful directing group for the incoming nucleophile. In the absence of protection, direct bromination of carbazole typically occurs at the C3 and C6 positions due to their higher electron density, making selective C1 functionalization nearly impossible through electrophilic aromatic substitution. However, by first converting the N-H bond into an N-acyl bond using reagents like acetic anhydride, the nitrogen lone pair is delocalized into the carbonyl group, reducing the electron density of the ring and altering the coordination environment for the lithium reagent. This modification allows the bulky butyllithium species to coordinate with the carbonyl oxygen, effectively guiding the lithiation to the adjacent C1 position through a cyclic transition state.
Temperature control and solvent selection play equally critical roles in maintaining this delicate regioselectivity and preventing side reactions such as multiple lithiations or decomposition of the aryl lithium intermediate. The patent specifies conducting the lithiation at cryogenic temperatures ranging from -80°C to -30°C, which kinetically traps the mono-lithiated species and suppresses thermal degradation. Additionally, the inclusion of ligands like Tetramethylethylenediamine (TMEDA) in non-polar solvents like n-hexane enhances the stability and nucleophilicity of the butyllithium reagent, ensuring complete conversion before the addition of the brominating agent. This meticulous control over reaction conditions ensures that the subsequent quenching with bromine sources like 1,2-dibromoethane or NBS occurs exclusively at the C1 site, yielding the desired N-substituted 1-bromocarbazole intermediate with minimal isomeric impurities.
How to Synthesize 1-Bromocarbazole Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and strict adherence to temperature profiles to maximize yield and purity. The process begins with the acylation of carbazole, followed by the critical low-temperature lithiation-bromination sequence, and concludes with an acidic hydrolysis to reveal the final free amine product. Detailed standard operating procedures for each stage, including specific molar ratios and solvent volumes, are essential for reproducing the high efficiency reported in the patent data. For a comprehensive breakdown of the exact experimental parameters and safety considerations required for execution, please refer to the standardized guide below.
- Perform N-H acylation of carbazole using acetic anhydride and an acid catalyst to form N-acyl carbazole intermediate.
- Conduct low-temperature lithiation (-50 to -80°C) with n-BuLi followed by bromination to selectively substitute the C1 position.
- Execute hydrolytic deprotection using dilute hydrochloric acid under heating to remove the acyl group and isolate 1-bromocarbazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this lithiation-based synthesis offers compelling economic benefits that extend far beyond simple reagent costs. By shifting away from palladium-catalyzed or phosphorus-dependent chemistries, the process eliminates the need for expensive precious metal recovery systems and hazardous waste disposal protocols associated with phosphine oxides. This transition results in a drastically simplified downstream processing workflow, where the final product can often be isolated through simple crystallization and washing, thereby reducing energy consumption and solvent usage significantly. The reliance on carbazole, a commodity chemical with a stable and abundant global supply, further insulates the production process from the volatility often seen with specialized coupling partners or exotic catalysts.
- Cost Reduction in Manufacturing: The elimination of high-cost catalysts such as palladium complexes and stoichiometric phosphorus reagents directly lowers the bill of materials for every batch produced. Furthermore, the high atom economy of the lithiation route minimizes raw material waste, meaning less money is spent on purchasing excess starting materials to compensate for poor yields. The simplified workup procedure reduces labor hours and utility costs associated with distillation and chromatography, leading to substantial cost savings that improve the overall margin profile of the final API intermediate.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of carbazole and common acylating agents like acetic anhydride, which are available from multiple global suppliers with consistent quality. Unlike methods dependent on single-source specialty reagents or sensitive organometallic catalysts that may face logistics disruptions, this robust chemistry ensures continuous production capability. The reduced sensitivity to trace impurities compared to palladium-catalyzed systems also means that standard industrial grade solvents can often be utilized, further securing the supply chain against specification bottlenecks.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as low-temperature dosing and crystallization that are standard in modern multipurpose chemical plants. From an environmental perspective, the absence of heavy metals and persistent phosphorus waste simplifies regulatory compliance and reduces the burden on wastewater treatment facilities. This green chemistry profile aligns perfectly with corporate sustainability goals, facilitating easier permitting for expanding production capacity to meet rising market demand for high-purity OLED material precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the comparative data and experimental examples provided in the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply route. The answers reflect the specific advantages in purity, yield, and operational safety that distinguish this method from prior art.
Q: Why is the lithiation method superior to traditional Cadogan cyclization for 1-bromocarbazole?
A: Traditional Cadogan cyclization requires toxic phosphorus reagents like triphenylphosphine and generates significant hazardous waste. The lithiation method described in CN113582910B avoids phosphorus entirely, utilizes inexpensive carbazole as a starting material, and achieves higher purity (>99.5%) with simpler post-treatment.
Q: How does the new synthesis route ensure regioselectivity at the C1 position?
A: The process employs an N-acyl protecting group which directs the lithiation reagent specifically to the C1 position due to steric and electronic effects. Combined with strict low-temperature control (-50 to -80°C) and the use of stabilizers like TMEDA, the reaction suppresses substitution at the more reactive C3 and C6 positions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It uses commercially available raw materials, avoids expensive transition metal catalysts like palladium, and features straightforward workup procedures such as crystallization and washing, making it economically viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Bromocarbazole Supplier
As the demand for high-performance organic electronic materials and complex pharmaceutical intermediates continues to surge, having a manufacturing partner with deep expertise in advanced heterocyclic synthesis is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the cryogenic conditions and sensitive organolithium chemistry required by this patent, while our stringent purity specifications and rigorous QC labs guarantee that every batch of 1-bromocarbazole meets the exacting standards required for OLED and pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your total cost of ownership for this critical intermediate. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this lithiation-based route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that secure your production continuity and enhance your product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
