Scalable Metal-Catalyzed Synthesis of 1-Bromocarbazole Derivatives for High-Performance OLEDs
The chemical industry is witnessing a significant paradigm shift in the synthesis of heterocyclic intermediates, driven by the urgent demand for high-purity materials in the organic light-emitting diode (OLED) and pharmaceutical sectors. Patent CN113372261A introduces a groundbreaking methodology for the preparation of 1-bromo/chlorocarbazole and its derivatives, addressing long-standing challenges in regioselective halogenation. This innovation utilizes a transition metal salt catalyst to facilitate the direct reaction between carbazole substrates and N-halosuccinimides (NBS/NCS) under relatively mild thermal conditions. Unlike traditional multi-step sequences that rely on cumbersome protection strategies, this approach leverages the intrinsic electronic properties of the carbazole nucleus, enhanced by metal coordination, to achieve selective functionalization at the 1-position. The implications for manufacturing high-performance hole transport materials are profound, offering a pathway to reduce process complexity while enhancing the overall economic viability of producing these critical electronic chemical intermediates.
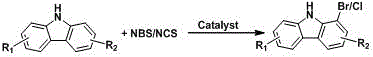
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-substituted carbazoles has been plagued by inefficiencies and operational hazards inherent to indirect synthetic routes. Prior art, such as the methods described in WO2011105161 and CN106397304A, often necessitates the use of o-bromophenylhydrazine hydrochloride or involves the installation and subsequent removal of bulky tert-butyl protecting groups. These conventional pathways are not only atom-uneconomical but also introduce significant safety risks due to the handling of hydrazine derivatives and the use of corrosive Lewis acids like aluminum trichloride for deprotection. Furthermore, the requirement for multiple isolation and purification steps between protection, halogenation, and deprotection drastically increases the cumulative yield loss and extends the production lead time. The generation of substantial amounts of acidic waste and the need for specialized corrosion-resistant equipment further elevate the capital and operational expenditures, making these traditional methods less attractive for modern, sustainable large-scale manufacturing environments.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent data streamlines the synthesis into a direct, catalytic C-H functionalization process that bypasses the need for protecting groups entirely. By employing inexpensive and readily available metal salts as catalysts, the reaction proceeds efficiently in common organic solvents such as toluene, xylene, or mesitylene at temperatures ranging from 60 to 200°C. This direct halogenation strategy capitalizes on the weak coordination between the metal center and the nitrogen atom of the carbazole ring, effectively directing the electrophilic halogen source to the desired ortho-position. The operational simplicity is a key differentiator; the reaction can often be conducted under an air atmosphere, eliminating the need for rigorous inert gas handling systems. This reduction in unit operations translates directly into a more robust and scalable process, capable of delivering moderate to good yields of the target 1-halo carbazoles with significantly reduced environmental footprint and processing time.
Mechanistic Insights into Metal-Salt Catalyzed Regioselective Halogenation
The core of this technological advancement lies in the sophisticated interplay between the metal catalyst and the carbazole substrate, which overrides the natural electronic bias of the molecule. While the nitrogen atom in carbazole typically directs electrophilic substitution to the 3 and 6 positions due to resonance effects, the presence of a transition metal salt alters this landscape through coordination chemistry. The metal cation, acting as a Lewis acid, coordinates with the lone pair of electrons on the carbazole nitrogen, thereby modulating the electron density distribution across the aromatic rings. This coordination effect, combined with hydrogen bonding interactions, activates the 1-position towards electrophilic attack by the N-halosuccinimide species. The result is a highly regioselective transformation that favors the formation of the 1-bromo or 1-chloro isomer, a feat that is difficult to achieve with high selectivity using standard halogenation reagents alone without the catalytic influence.

Furthermore, the mechanistic robustness of this system is evidenced by its remarkable tolerance to diverse functional groups, ensuring high purity profiles essential for electronic applications. The catalytic cycle operates effectively even in the presence of electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as esters and halogens, without inducing side reactions like over-halogenation or ring degradation. For instance, substrates like 3,6-dichlorocarbazole can be selectively converted to 1-bromo-3,6-dichlorocarbazole, demonstrating the catalyst's ability to distinguish between different reactive sites on the polycyclic framework. This selectivity is crucial for minimizing the formation of difficult-to-remove impurities, thereby simplifying the downstream purification processes such as column chromatography or recrystallization. The ability to maintain structural integrity while introducing the halogen handle makes this method superior for synthesizing complex precursors for polymerization or cross-coupling reactions.
How to Synthesize 1-Bromocarbazole Efficiently
The practical implementation of this synthesis route offers a straightforward protocol that balances reaction efficiency with operational safety, making it ideal for both laboratory optimization and industrial scale-up. The process begins with the precise mixing of the carbazole starting material and the halogenating agent, typically N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), in a molar ratio optimized to minimize excess reagent waste while driving the reaction to completion. A catalytic amount of a metal salt, such as Iron(III) sulfate or Palladium nitrate, is introduced to initiate the transformation, with the choice of solvent playing a critical role in solubilizing the reactants and stabilizing the transition states. The detailed standardized synthesis steps, including specific temperature ramps, stirring rates, and workup procedures required to achieve the reported yields of up to 83%, are outlined in the technical guide below.
- Mix carbazole raw material with N-bromosuccinimide (NBS) and a metal salt catalyst (e.g., Fe2(SO4)3) in a suitable organic solvent like toluene or xylene.
- Heat the reaction mixture to 90-110°C under air atmosphere and stir for 1 to 6 hours to facilitate the halogenation reaction.
- Cool the system, extract with dichloromethane, dry over anhydrous sodium sulfate, and purify via column chromatography to isolate the target 1-bromocarbazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility in the fine chemical sector. The elimination of multi-step protection and deprotection sequences fundamentally alters the cost structure of manufacturing 1-halo carbazoles by removing entire categories of raw materials and processing time. By avoiding the use of expensive and hazardous reagents like aluminum trichloride and tert-butyl sources, manufacturers can significantly reduce the costs associated with raw material acquisition, hazardous waste disposal, and equipment maintenance. The simplified workflow also implies a reduction in labor hours and energy consumption per kilogram of product, contributing to a leaner and more competitive manufacturing model that can better absorb fluctuations in market pricing for upstream commodities.
- Cost Reduction in Manufacturing: The transition to a direct catalytic halogenation process removes the necessity for costly protecting group chemistry, which traditionally accounts for a substantial portion of the total production cost in carbazole derivative synthesis. By utilizing abundant and inexpensive metal salts like iron sulfate instead of precious metal catalysts or stoichiometric Lewis acids, the direct material costs are drastically lowered. Additionally, the higher atom economy of the direct route means less waste is generated per unit of product, leading to substantial savings in waste treatment and environmental compliance fees. This efficiency gain allows for a more aggressive pricing strategy while maintaining healthy margins, providing a distinct competitive edge in the global market for electronic chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as NBS, NCS, and common solvents like toluene and xylene ensures a resilient supply chain that is less susceptible to the bottlenecks often associated with specialized reagents. Since the reaction can proceed under air atmosphere and does not require stringent anhydrous or oxygen-free conditions, the logistical burden of transporting and storing sensitive materials is minimized. This operational flexibility allows for faster turnaround times and more reliable delivery schedules, as production is not held up by the availability of niche catalysts or the need for complex reactor setups. Consequently, procurement managers can secure a more stable supply of high-purity intermediates, mitigating the risk of production delays for downstream OLED or pharmaceutical clients.
- Scalability and Environmental Compliance: The robustness of this catalytic system underpins its exceptional scalability, enabling seamless transition from gram-scale laboratory experiments to multi-ton commercial production without significant process re-engineering. The use of less corrosive catalysts and the avoidance of harsh acidic deprotection steps significantly reduce the wear and tear on reactor vessels, extending equipment lifespan and lowering capital replacement costs. Moreover, the reduced generation of hazardous byproducts aligns with increasingly stringent global environmental regulations, facilitating easier permitting and smoother operations in regions with strict emission standards. This sustainability profile not only future-proofs the manufacturing asset but also enhances the brand value for clients seeking green chemistry solutions for their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-catalyzed halogenation technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this synthesis route for their specific applications. Understanding these nuances is critical for R&D teams planning process validation and for supply chain leaders assessing the feasibility of integrating this new intermediate into their existing procurement portfolios.
Q: What are the advantages of this metal-catalyzed method over traditional protection-deprotection routes?
A: This method eliminates the need for bulky protecting groups like tert-butyl and harsh deprotection reagents such as aluminum trichloride, significantly simplifying the workflow and reducing hazardous waste generation while maintaining moderate to good yields.
Q: Is this process compatible with sensitive functional groups on the carbazole ring?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating substrates with ether, ester, methoxy, halogen, methyl, and aryl substituents without compromising the integrity of these groups during halogenation.
Q: What types of catalysts are effective for this transformation?
A: A wide range of metal salts including Iron (Fe), Palladium (Pd), Copper (Cu), and Nickel (Ni) salts are effective, with Iron(III) sulfate being a particularly cost-effective and efficient option for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Bromocarbazole Supplier
As the demand for high-performance organic electronic materials continues to surge, the ability to supply ultra-pure 1-bromocarbazole derivatives becomes a critical differentiator for CDMO partners. NINGBO INNO PHARMCHEM leverages this advanced metal-catalyzed technology to deliver products with stringent purity specifications, ensuring optimal performance in OLED devices and pharmaceutical syntheses. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, supported by rigorous QC labs that validate every batch against the highest industry standards. We understand that consistency is key in the electronics sector, and our optimized processes guarantee the low impurity profiles necessary for next-generation hole transport materials.
We invite forward-thinking organizations to collaborate with us to unlock the full potential of this efficient synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our innovative manufacturing capabilities can drive value and reliability in your supply chain.
