Advanced Synthesis of C10 Aldehyde Esters for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries constantly seek robust pathways for constructing complex carbon skeletons, particularly for terpenoid derivatives. Patent CN102391114A introduces a highly efficient method for synthesizing C10 aldehyde esters, which serve as critical building blocks for high-value active pharmaceutical ingredients. This technology addresses long-standing challenges in the production of intermediates used for compounds like beta-Apo-8'-carotenal derivatives. By leveraging a combination of Wittig-Horner olefination and modern catalytic oxidation, the process offers a streamlined alternative to legacy methods. For a reliable pharmaceutical intermediate supplier, adopting such a route ensures consistent quality and supply continuity. The following analysis details how this innovation transforms the manufacturing landscape for these essential chemical entities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C10 aldehyde esters has been plagued by significant economic and technical hurdles. Early methodologies, such as those reported by Schwieter et al., relied on expensive six-carbon alcohol starting materials that drove up the overall cost of goods. Furthermore, these routes often necessitated the use of shortening agents in secondary steps due to the inherent instability and high reactivity of the intermediates involved. A major bottleneck in these traditional processes was the final oxidation step, which frequently utilized stoichiometric amounts of manganese dioxide. This not only resulted in low yields of the target aldehyde but also generated substantial quantities of hazardous titanium dioxide and manganese waste residues, creating severe environmental disposal issues. Additionally, alternative routes proposed by entities like BASF AG depended on specialized raw materials such as 4-chloro ethyl tiglate, which were not commercially available domestically and posed significant supply chain risks for manufacturers seeking localization.
The Novel Approach
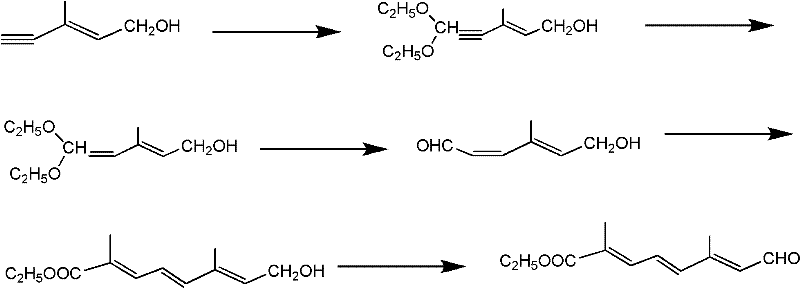
The methodology disclosed in patent CN102391114A represents a paradigm shift towards sustainable and economically viable manufacturing. This novel approach utilizes readily available starting materials, specifically E-4-acetoxy-2-methyl-2-butenal, which can be sourced locally, thereby drastically reducing raw material costs and lead times. The synthetic strategy employs a clever sequence of acetalization and vinyl ether addition to construct the carbon chain with high precision. Crucially, the final oxidation step replaces the wasteful manganese dioxide protocol with a catalytic system using TEMPO and copper chloride under an oxygen atmosphere. This modification not only improves the atom economy but also simplifies the downstream processing by eliminating the filtration of heavy metal sludge. For procurement managers, this translates to cost reduction in fine chemical manufacturing through both material savings and reduced waste treatment expenses.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Wittig-Horner Coupling
The core of this synthetic breakthrough lies in the precise orchestration of carbon-carbon bond formation and functional group interconversion. The process initiates with the protection of the aldehyde functionality via acetalization using trimethyl or triethyl orthoformate under acidic conditions, ensuring stability during subsequent transformations. Following hydrolysis, the intermediate undergoes a Lewis acid-catalyzed reaction with vinyl ethers, extending the carbon chain while introducing necessary ether linkages that are later hydrolyzed to reveal the aldehyde group. The pivotal step involves the preparation of a phosphonate ester via the Arbuzov reaction between triethyl phosphite and ethyl 2-chloropropionate. This phosphonate is then coupled with the aldehyde intermediate through a Wittig-Horner reaction in the presence of sodium ethoxide. This reaction is highly stereoselective, favoring the formation of the desired trans-conjugated system essential for the biological activity of the final terpenoid products.
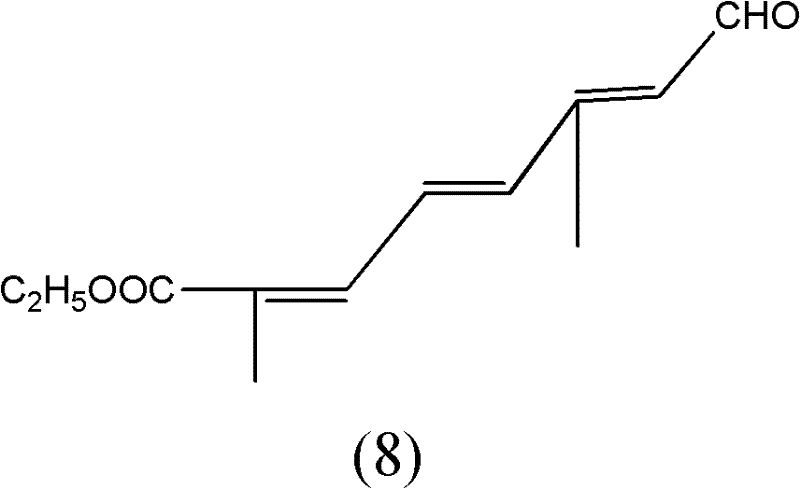
The terminal transformation involves the oxidation of the primary alcohol to the corresponding aldehyde using a TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) catalytic cycle. In this mechanism, TEMPO acts as a stable nitroxyl radical mediator, shuttling electrons between the substrate and the terminal oxidant, molecular oxygen. The presence of cuprous chloride facilitates the regeneration of the active oxoammonium species of TEMPO. This catalytic cycle operates efficiently at room temperature, minimizing thermal degradation of the sensitive conjugated polyene system. By avoiding harsh stoichiometric oxidants, the process maintains high purity specifications and minimizes the formation of over-oxidized byproducts such as carboxylic acids. This level of control is vital for producing high-purity C10 aldehyde ester suitable for downstream pharmaceutical applications.
How to Synthesize C10 Aldehyde Ester Efficiently
The synthesis of this valuable intermediate is achieved through a sequential six-step process that balances reactivity with operational simplicity. The route begins with the protection of the starting aldehyde, followed by chain extension and deprotection to yield a key aldehyde precursor. Simultaneously, a phosphonate coupling partner is synthesized via thermal Arbuzov rearrangement. These two fragments are united through a base-mediated olefination to establish the conjugated backbone. Finally, the primary alcohol moiety is selectively oxidized to the aldehyde using the green TEMPO system. Detailed standardized synthetic steps see the guide below.
- Perform acetalization of E-4-acetoxy-2-methyl-2-butenal with orthoformate, followed by hydrolysis to obtain the protected intermediate.
- React the intermediate with vinyl ether under Lewis acid catalysis, then hydrolyze to form the aldehyde precursor.
- Conduct Arbuzov reaction to synthesize the phosphonate, couple via Wittig-Horner, and oxidize the resulting alcohol using TEMPO/CuCl/O2.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this patented methodology offers compelling strategic benefits beyond mere technical feasibility. The shift from imported, specialty chemicals to commoditized, locally sourced reagents fundamentally alters the cost structure of the production line. By eliminating dependencies on single-source suppliers for exotic starting materials, manufacturers can mitigate the risk of supply disruptions and price volatility. Furthermore, the simplification of the workup procedures, particularly the removal of heavy metal filtration steps, reduces the operational burden on production facilities. This efficiency gain allows for faster batch turnover and improved asset utilization, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The replacement of expensive six-carbon alcohols and stoichiometric manganese dioxide with cheap orthoformates and catalytic TEMPO significantly lowers the variable cost per kilogram. The elimination of hazardous waste disposal fees associated with manganese sludge further enhances the economic profile. Additionally, the high conversion rates observed in the Wittig-Horner step minimize the loss of valuable intermediates, ensuring that raw material input is maximized into saleable output. These factors combine to deliver substantial cost savings without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: Utilizing raw materials that are produced domestically ensures a stable and continuous supply flow, insulating the manufacturing process from international logistics bottlenecks. The robustness of the reaction conditions, which tolerate ambient temperatures and standard pressures, means that production can be maintained across multiple facilities without requiring specialized high-pressure equipment. This flexibility allows for diversified production strategies, reducing the risk of single-point failures in the supply network and ensuring consistent delivery to downstream customers.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, avoiding exothermic runaways and unstable intermediates that complicate large-scale operations. The use of molecular oxygen as the oxidant aligns with green chemistry principles, reducing the environmental footprint of the facility. This compliance with stringent environmental regulations facilitates smoother permitting processes and long-term operational sustainability. The ability to scale from pilot batches to multi-ton production runs is seamless, supporting the growing demand for complex terpenoid intermediates in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer.
Q: What are the advantages of the TEMPO oxidation method over traditional manganese dioxide oxidation?
A: The TEMPO catalytic oxidation system utilizes molecular oxygen as the terminal oxidant, significantly reducing hazardous waste generation compared to stoichiometric manganese dioxide methods, which produce large amounts of solid residue.
Q: Are the raw materials for this C10 aldehyde ester synthesis readily available?
A: Yes, the process utilizes localized and commercially available raw materials such as E-4-acetoxy-2-methyl-2-butenal, orthoformates, and vinyl ethers, avoiding the supply chain risks associated with specialized imported reagents.
Q: How does this method improve scalability for industrial production?
A: The route eliminates complex purification steps and avoids unstable intermediates found in older methods, allowing for simpler operational control and easier commercial scale-up of complex terpenoid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C10 Aldehyde Ester Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the TEMPO-mediated oxidation pathway to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of C10 aldehyde ester meets the exacting standards required for pharmaceutical grade intermediates. Our commitment to quality and consistency makes us the preferred partner for global enterprises seeking dependable supply chains.
We invite you to collaborate with us to optimize your production costs and secure your raw material supply. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your operational efficiency. Let us help you navigate the complexities of fine chemical manufacturing with confidence and precision.
