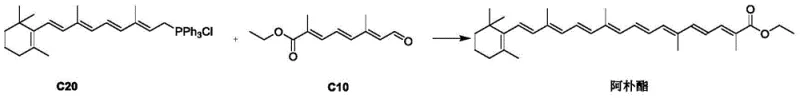
Revolutionizing Apoester Production: A Scalable Route for High-Purity C10 Aldehyde Ester
The global demand for high-performance carotenoids in the food and feed industries continues to drive innovation in intermediate synthesis, particularly for Apoester, a vital C30 conjugated unsaturated carbon chain compound. Patent CN114805066B, published in March 2023, introduces a groundbreaking preparation method for the key intermediate, C10 aldehyde ester, which serves as the critical building block in the C20+C10 synthesis strategy for Apoester. This technology addresses long-standing bottlenecks in carotenoid manufacturing by replacing complex, low-yield pathways with a streamlined, three-step protocol that leverages inexpensive starting materials such as acetoacetaldehyde and glyoxal. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize the supply chain for fat-soluble yellow pigments used in broiler chicken coloring and human nutritional supplements. The following analysis details how this novel route enhances process efficiency while maintaining the stringent purity specifications required for food-grade applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
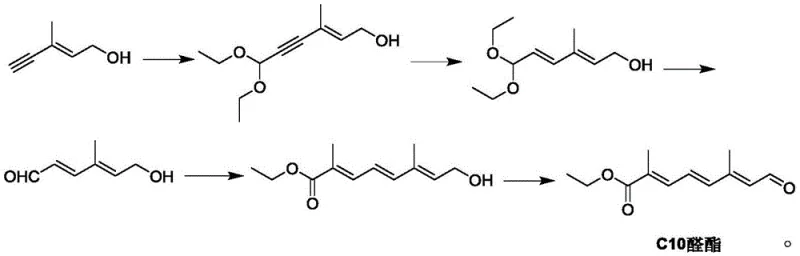
Historically, the industrial synthesis of C10 aldehyde ester has been plagued by intricate reaction sequences that hinder cost-effective mass production. As illustrated in the prior art, traditional routes often rely on acetylene chemistry involving multiple protection and deprotection steps, which drastically increases the number of unit operations and solvent consumption. These legacy methods typically suffer from low overall yields due to the accumulation of losses at each stage, alongside the generation of substantial amounts of hazardous waste, often referred to as "three wastes" in chemical manufacturing. The reliance on specialized alkyne precursors not only elevates raw material costs but also introduces significant safety risks associated with handling unstable intermediates. Furthermore, the complexity of these routes makes process control difficult, leading to variability in impurity profiles that can compromise the quality of the final Apoester product. Such inefficiencies create a fragile supply chain where lead times are extended, and production costs remain prohibitively high for widespread application in the competitive feed additive market.
The Novel Approach
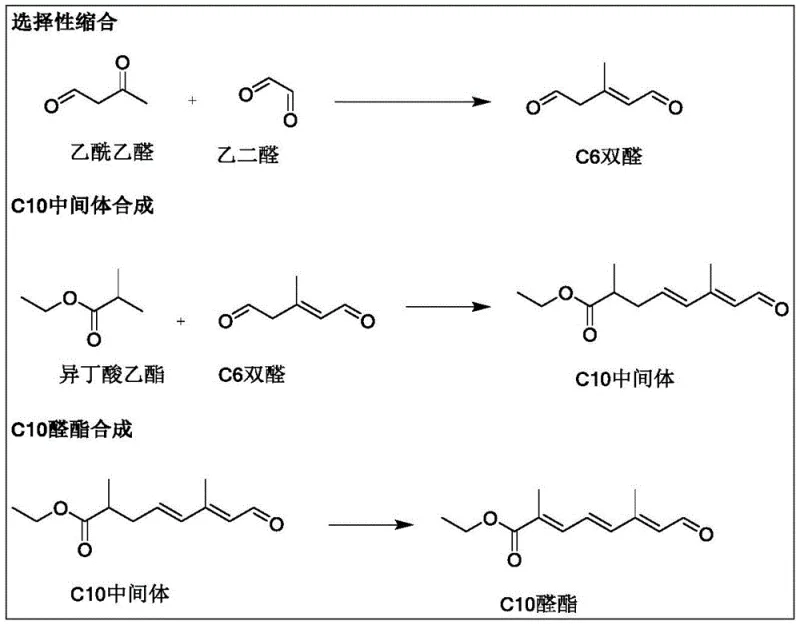
In stark contrast to the cumbersome traditional pathways, the methodology disclosed in CN114805066B offers a radically simplified approach that fundamentally restructures the synthesis logic. The new route initiates with a highly selective condensation reaction between acetoacetaldehyde and glyoxal under alkali catalysis to directly construct the C6 dialdehyde skeleton, bypassing the need for complex alkyne manipulations. This C6 intermediate is then coupled with ethyl isobutyrate via a Grignard-type reaction to extend the carbon chain to the C10 framework, followed by a final reduction-elimination step to establish the requisite conjugated aldehyde ester functionality. By reducing the entire synthesis to just three distinct chemical transformations, the process minimizes the potential for side reactions and simplifies downstream purification. The use of commodity chemicals as starting materials ensures a stable and cost-efficient supply base, while the shortened reaction sequence significantly reduces the operational footprint. This strategic shift not only lowers the barrier to entry for manufacturers but also aligns with modern green chemistry principles by reducing solvent usage and waste discharge, making it an ideal candidate for sustainable industrial scale-up.
Mechanistic Insights into Selective Condensation and Grignard Coupling
The core innovation of this patent lies in the precise control of the initial condensation reaction, where acetoacetaldehyde reacts with glyoxal in the presence of a strong base catalyst such as bis(trimethylsilyl)potassium amide (KHMDS). The reaction is conducted at low temperatures, specifically between -10°C and 10°C, to ensure high regioselectivity and prevent the polymerization of the reactive aldehyde groups. This careful thermal management allows for the formation of the C6 dialdehyde intermediate with exceptional conversion rates exceeding 99%, as evidenced by the experimental data. Following the reaction, continuous vacuum distillation is employed to isolate the product, achieving purity levels between 90% and 98%, which is critical for preventing impurity carryover into subsequent steps. The second phase involves the generation of a Grignard reagent from ethyl isobutyrate using pyridinium tribromide and magnesium powder in tetrahydrofuran. This organometallic species then attacks the C6 dialdehyde, extending the carbon chain. The subsequent treatment with phosphoric acid at elevated temperatures (85-120°C) facilitates the dehydration and rearrangement necessary to form the C10 intermediate. Finally, the introduction of a bulky base like potassium tert-butoxide in chloroform triggers an elimination reaction that restores the conjugated double bond system, yielding the target C10 aldehyde ester with high stereochemical integrity.
Impurity control is meticulously managed throughout this cascade, particularly during the quenching and elimination stages. The protocol specifies the use of acetic acid to quench excess magnesium and organomagnesium compounds before the acid-catalyzed rearrangement, preventing the formation of unwanted by-products that could arise from uncontrolled Grignard reactions. Furthermore, the stoichiometric excess of the base reagent in the final step serves a dual purpose: it neutralizes residual phosphoric acid from the previous stage and drives the elimination equilibrium towards the desired conjugated product. This integrated approach to reaction design ensures that the final C10 aldehyde ester possesses a clean impurity profile, which is paramount for its downstream application in the synthesis of Apoester. The robustness of these mechanistic steps provides a reliable foundation for consistent batch-to-batch reproducibility, a key requirement for regulatory compliance in the food and feed additive sectors.
How to Synthesize C10 Aldehyde Ester Efficiently
The synthesis of C10 aldehyde ester via this patented route requires strict adherence to reaction parameters to maximize yield and purity. The process begins with the preparation of the C6 dialdehyde intermediate, followed by its conversion to the C10 skeleton through organometallic coupling, and concludes with a base-mediated elimination. Each step demands precise control over temperature, reagent addition rates, and stoichiometry to avoid side reactions. For detailed operational protocols, including specific solvent ratios and distillation parameters, refer to the standardized guide below which outlines the critical process parameters derived from the patent examples.
- Perform selective condensation of acetoacetaldehyde and glyoxal using KHMDS catalyst in n-butanol at low temperature (-5 to 5°C) to obtain C6 dialdehyde intermediate.
- React ethyl isobutyrate with pyridinium tribromide and magnesium to form a Grignard reagent, then couple with the C6 dialdehyde intermediate followed by acid treatment.
- Execute a reduction-elimination reaction on the resulting C10 intermediate using potassium tert-butoxide in chloroform to finalize the C10 aldehyde ester structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative benefits that extend beyond simple technical feasibility. The shift from complex alkyne-based chemistry to a condensed three-step process using commodity feedstocks fundamentally alters the cost structure of C10 aldehyde ester manufacturing. By eliminating the need for expensive and hazardous specialty reagents, the process significantly reduces the direct material costs associated with production. Moreover, the reduction in the number of synthetic steps translates to a drastic simplification of the manufacturing workflow, which inherently lowers labor costs, energy consumption, and equipment occupancy time. This efficiency gain allows for higher throughput within existing facilities, effectively increasing capacity without the need for capital-intensive expansion. The simplified waste profile also reduces the burden on environmental treatment systems, leading to substantial operational savings in waste disposal and compliance management.
- Cost Reduction in Manufacturing: The utilization of low-cost raw materials such as acetoacetaldehyde, glyoxal, and ethyl isobutyrate replaces the expensive precursors required in traditional routes, driving down the overall bill of materials. The high yields reported in the patent examples, exceeding 85% for the initial condensation and over 90% for the final coupling, minimize raw material waste and maximize output per batch. Additionally, the avoidance of complex protection-deprotection sequences eliminates the costs associated with additional reagents and the extra purification steps they necessitate. This lean manufacturing approach ensures that the final cost of goods sold is optimized, providing a competitive edge in the pricing of downstream carotenoid products.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key starting materials are widely available commodity chemicals with established global supply chains, reducing the risk of shortages associated with niche intermediates. The robustness of the reaction conditions, which tolerate standard industrial solvents like n-butanol and tetrahydrofuran, ensures that production is not vulnerable to the supply constraints of exotic reagents. Furthermore, the shortened synthesis timeline accelerates the production cycle, allowing for faster response to market demand fluctuations and reduced inventory holding times. This agility strengthens the overall resilience of the supply chain, ensuring consistent availability of high-purity C10 aldehyde ester for downstream Apoester synthesis.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as vacuum distillation and conventional stirred-tank reactors that are common in fine chemical plants. The significant reduction in the number of steps and the use of less hazardous reagents simplify the safety management protocols, making it easier to obtain regulatory approvals for large-scale production. The minimized generation of "three wastes" aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with effluent treatment. This environmentally friendly profile not only facilitates smoother permitting processes but also enhances the sustainability credentials of the final product, appealing to eco-conscious customers in the food and feed industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in patent CN114805066B, ensuring accuracy and relevance for decision-makers evaluating this process for adoption.
Q: What are the primary advantages of the new C10 aldehyde ester synthesis route compared to traditional methods?
A: The new route described in patent CN114805066B utilizes readily available commodity chemicals like acetoacetaldehyde and glyoxal, significantly reducing raw material costs. It shortens the synthetic sequence to just three main steps, minimizing unit operations and waste generation compared to the complex multi-step alkyne-based routes previously reported.
Q: How does the process ensure high purity of the C6 dialdehyde intermediate?
A: High purity is achieved through precise temperature control during the condensation reaction (-10 to 10°C) and the use of specific strong base catalysts like bis(trimethylsilyl)potassium amide. Subsequent continuous vacuum distillation allows for the isolation of the C6 dialdehyde with purity levels ranging from 90% to 98%.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids hazardous reagents and complex protection-deprotection sequences, relying instead on robust reactions like Grignard formation and base-mediated elimination. The use of common solvents like tetrahydrofuran and chloroform further facilitates easy scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C10 Aldehyde Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient production of value-added carotenoids like Apoester. Our team of expert chemists has thoroughly analyzed the pathway described in CN114805066B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative route to life. We are committed to delivering high-purity C10 aldehyde ester that meets rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle the specific solvent systems and temperature controls necessary for this synthesis, ensuring that every batch delivered reflects the superior yield and purity profile demonstrated in the patent literature.
We invite global partners to collaborate with us to leverage this cost-effective technology for their carotenoid supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your production efficiency and reduce your overall operational costs. Let us be your trusted partner in securing a stable and economical supply of this essential fine chemical intermediate.
