Scalable Manufacturing of High-Purity Dexlansoprazole Intermediates via Novel Asymmetric Oxidation
Scalable Manufacturing of High-Purity Dexlansoprazole Intermediates via Novel Asymmetric Oxidation
The pharmaceutical industry continuously seeks robust synthetic routes for proton pump inhibitors, particularly for the enantiomerically pure forms that offer superior therapeutic profiles. A pivotal development in this domain is detailed in patent CN102731478A, which discloses an advanced preparation method for (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole. This compound serves as the critical chiral intermediate for Dexlansoprazole, a potent anti-ulcer medication. The disclosed technology addresses long-standing challenges in stereoselective synthesis by employing a modified Sharpless asymmetric oxidation protocol. By utilizing a specific chiral titanium catalyst system alongside cumene hydroperoxide, the method achieves exceptional optical purity and chemical yield simultaneously. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-value gastrointestinal therapeutics. The ability to produce this key building block with minimal impurities and high efficiency directly translates to reduced manufacturing costs and enhanced product reliability for downstream API production.
![Chemical structure of the key intermediate (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole](/insights/img/dexlansoprazole-intermediate-asymmetric-oxidation-pharma-supplier-20260309090111-02.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active benzimidazole sulfoxides has relied heavily on resolution techniques, which are inherently inefficient for large-scale manufacturing. Conventional chemical resolution involves derivatizing the racemate to form diastereomers, followed by separation and hydrolysis, a process that theoretically caps the yield at 50% and often results in actual yields as low as 40-50%. Furthermore, enzymatic resolution, while selective, introduces significant cost burdens due to the expense of biocatalysts and the stringent reaction conditions required to maintain enzyme activity. Another prevalent method involves the asymmetric oxidation of precursors lacking the 4-nitro group, which frequently leads to over-oxidation issues where the desired sulfoxide is further oxidized to the sulfone. Separating these sulfone byproducts is notoriously difficult due to their similar physical and chemical properties, often necessitating expensive and time-consuming column chromatography that is impractical for ton-scale production.
The Novel Approach
The innovative strategy presented in the patent circumvents these bottlenecks by performing the asymmetric oxidation directly on the 4-nitro substituted thioether precursor. This specific substrate configuration, when subjected to the optimized chiral titanium catalyst system, demonstrates remarkable resistance to over-oxidation while maintaining high stereoselectivity. The reaction proceeds smoothly under mild conditions, typically between 10°C and 50°C, avoiding the extreme temperatures that degrade sensitive functional groups. By carefully controlling the stoichiometry of the oxidant, specifically keeping the molar ratio of substrate to oxidant between 1:1 and 1:2.0, the process effectively halts at the sulfoxide stage. This precision eliminates the need for complex purification steps like chromatography, allowing the product to be isolated simply by crystallization or filtration. The result is a streamlined workflow that drastically reduces solvent consumption and processing time compared to legacy resolution methods.
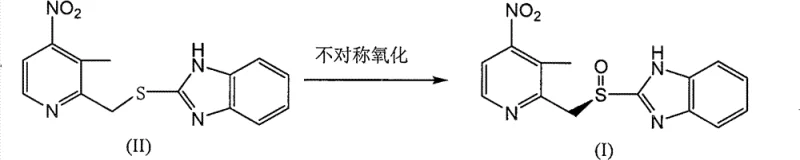
Mechanistic Insights into Titanium-Catalyzed Asymmetric Oxidation
The core of this technological breakthrough lies in the formation of a highly organized chiral titanium-tartrate complex that dictates the stereochemical outcome of the oxygen transfer. The catalyst is generated in situ by reacting titanium isopropoxide with a chiral diol, specifically L-(+)-diethyl tartrate, in the presence of a controlled amount of water. This ternary complex creates a rigid chiral environment around the titanium center, which coordinates with the sulfur atom of the thioether substrate. When the oxidant, cumene hydroperoxide, approaches this complex, the chiral ligands sterically hinder one face of the sulfur atom, forcing the oxygen transfer to occur exclusively from the less hindered face. This mechanism ensures the preferential formation of the (R)-enantiomer with extremely high enantiomeric excess, often exceeding 97% ee as demonstrated in the patent examples. The presence of the electron-withdrawing nitro group on the pyridine ring further modulates the electronic density of the sulfur, making it less susceptible to non-selective background oxidation and enhancing the catalyst's control over the reaction pathway.
Impurity control is another critical aspect managed by this mechanistic design. In traditional Sharpless oxidations, the primary impurity concern is the over-oxidized sulfone derivative, which is chemically similar to the target sulfoxide. However, the specific reaction conditions outlined in this patent, including the use of diisopropylethylamine as a base and the precise temperature control during oxidant addition, kinetically favor the formation of the sulfoxide. The base likely serves to neutralize acidic byproducts that could otherwise catalyze non-selective oxidation or decompose the chiral catalyst. Furthermore, the choice of solvent, such as toluene or ethyl acetate, provides an optimal medium for the solubility of the reactants while facilitating the precipitation of the product upon completion. This phase behavior aids in driving the reaction to completion and simplifies the isolation of the high-purity solid, ensuring that the final impurity profile meets the stringent requirements for pharmaceutical intermediates without requiring additional recrystallization cycles.
How to Synthesize (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole Efficiently
Implementing this synthesis requires careful attention to the preparation of the catalytic species and the addition rate of the oxidant to maintain thermal stability. The process begins with the activation of the titanium catalyst in an anhydrous environment under nitrogen protection to prevent premature hydrolysis. Once the active chiral complex is formed, the thioether substrate is introduced, followed by the slow, dropwise addition of the oxidant to manage the exotherm. Maintaining the reaction temperature within the specified range of 20°C to 40°C is vital for maximizing both yield and optical purity. Detailed standardized operating procedures regarding reagent grades, mixing speeds, and quenching protocols are essential for reproducibility. For a comprehensive breakdown of the specific experimental steps and safety considerations, please refer to the technical guide below.
- Prepare the chiral catalyst by mixing titanium isopropoxide, L-(+)-diethyl tartrate, and water in a solvent like toluene at 50-55°C.
- Add the substrate 2-[[[3-methyl-4-nitro-2-pyridyl]methyl]thio]benzimidazole and a base such as diisopropylethylamine to the mixture.
- Slowly add cumene hydroperoxide as the oxidant while maintaining the temperature between 20-40°C to ensure high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The shift from resolution-based methods to direct asymmetric synthesis fundamentally alters the cost structure of the intermediate by eliminating the 50% yield loss inherent in resolving racemates. This efficiency gain means that less raw material is required to produce the same amount of active intermediate, directly lowering the cost of goods sold. Additionally, the removal of chromatographic purification steps significantly reduces solvent usage and waste disposal costs, aligning with modern green chemistry initiatives and environmental compliance standards. The robustness of the reaction conditions also implies a more reliable supply chain, as the process is less sensitive to minor fluctuations in temperature or reagent quality compared to enzymatic methods.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and the reduction in solvent volume for purification lead to significant operational savings. By achieving high yields in a single step, manufacturers can reduce the number of reactor batches required to meet demand, thereby optimizing facility utilization rates. The avoidance of column chromatography not only saves on silica gel and solvents but also reduces the labor hours associated with running and monitoring these separations. Consequently, the overall production cost per kilogram of the intermediate is drastically lowered, providing a competitive pricing advantage in the global market for proton pump inhibitor ingredients.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as cumene hydroperoxide and titanium isopropoxide ensures a consistent supply of inputs without reliance on specialized biological materials that may have shelf-life constraints. The mild reaction temperatures reduce the energy load on manufacturing facilities and minimize the risk of thermal runaways, leading to safer and more predictable production schedules. This stability allows for better forecasting and inventory management, ensuring that downstream API manufacturers receive their materials on time without unexpected delays caused by batch failures or complex purification bottlenecks.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing common organic solvents that are easily recovered and recycled. The high selectivity of the reaction minimizes the generation of hazardous waste streams associated with sulfone byproducts, simplifying wastewater treatment processes. This environmental friendliness facilitates regulatory approval in jurisdictions with strict emission standards. Furthermore, the simplicity of the workup procedure, often involving simple filtration or crystallization, allows for seamless transition from pilot plant to multi-ton commercial production, ensuring that supply can be rapidly ramped up to meet market surges without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric oxidation technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The focus remains on practical application, yield optimization, and quality assurance metrics that matter most to production stakeholders.
Q: How does this new method improve upon traditional resolution techniques for Dexlansoprazole intermediates?
A: Traditional resolution methods often suffer from theoretical maximum yields of 50% and require complex separation steps. This asymmetric oxidation method achieves yields exceeding 90% with optical purity greater than 97% ee in a single synthetic step, significantly reducing material waste and processing time.
Q: What are the critical parameters for controlling over-oxidation to the sulfone derivative?
A: Controlling the molar ratio of the oxidant to the substrate is crucial. The patent specifies a ratio between 1:1 and 1:2.0, preferably 1:1 to 1:1.5. Additionally, maintaining moderate reaction temperatures (20-40°C) prevents the thermal runaway that typically drives the formation of unwanted sulfone byproducts.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (room temperature to 50°C) and common solvents like toluene or ethyl acetate. The elimination of column chromatography for purification and the use of stable reagents make it highly amenable to commercial scale-up in standard reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole meets the highest international standards for optical purity and chemical integrity. Our commitment to excellence extends beyond mere compliance; we actively collaborate with clients to optimize processes for cost and efficiency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production goals, ensuring a seamless and profitable partnership for your pharmaceutical supply chain.
