Advanced Asymmetric Oxidation for High-Purity Dexlansoprazole Intermediates and Commercial Scale-Up
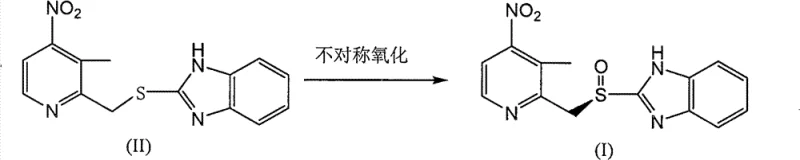
The pharmaceutical industry's relentless pursuit of more efficient synthetic routes for proton pump inhibitors has found a significant breakthrough in patent CN102731478A. This document discloses a robust preparation method for (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole, a critical chiral intermediate in the synthesis of Dexlansoprazole. Unlike traditional approaches that rely on cumbersome resolution techniques or harsh oxidation conditions, this invention leverages a modified Sharpless asymmetric oxidation system to deliver exceptional stereocontrol. The technical significance lies in its ability to suppress over-oxidation to the corresponding sulfone while maintaining high reaction rates under mild thermal conditions. For R&D directors and process chemists, this represents a pivotal shift towards more atom-economical and environmentally benign manufacturing protocols. By utilizing a specific titanium-tartrate catalyst complex alongside cumene hydroperoxide, the process achieves optical purities that meet the stringent regulatory requirements for modern anti-ulcer medications without the need for extensive downstream purification.
![Chemical structure of the key chiral intermediate (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole](/insights/img/dexlansoprazole-intermediate-asymmetric-oxidation-supplier-20260309085903-02.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active benzimidazole sulfoxides has been plagued by inherent inefficiencies associated with racemic resolution strategies. As detailed in the background art, chemical resolution methods described in documents like DE 403545 require multi-step derivatization and hydrolysis sequences, resulting in theoretical yield losses where the maximum obtainable yield is capped at 50 percent, with practical industrial yields often hovering between 40 to 50 percent. Furthermore, enzymatic resolution, while selective, introduces prohibitive costs due to the expense of biocatalysts and the necessity for strict reaction condition controls that complicate scale-up. Inclusion resolution methods using expensive binaphthol hosts add another layer of complexity, requiring chromatographic separation of solid complexes which is economically unviable for bulk API intermediate production. Additionally, earlier asymmetric oxidation attempts, such as those cited in WO9602535, struggled with selectivity issues where the desired sulfoxide was easily over-oxidized to the sulfone byproduct, necessitating difficult purification steps due to the similar physicochemical properties of the oxidation states.
The Novel Approach
The methodology presented in CN102731478A fundamentally addresses these bottlenecks by optimizing the stoichiometry and catalytic environment of the Sharpless oxidation. By employing a molar ratio of the sulfide substrate to the oxidant ranging from 1:1 to 1:2.0, specifically preferring a tighter range of 1:1 to 1:1.5, the process effectively minimizes the formation of the undesired sulfone impurity. The innovation allows the reaction to proceed at温和 temperatures between 10 to 50 degrees Celsius, with a particular preference for room temperature operations, thereby eliminating the energy-intensive cryogenic conditions (-10 to 10 degrees Celsius) required by previous iterations like those in WO200911489. This novel approach not only simplifies the thermal management infrastructure required for the reactor but also significantly enhances the safety profile of the operation by avoiding extreme cold chains. The result is a streamlined process that delivers the target chiral sulfoxide with yields exceeding 94 percent and enantiomeric excess values approaching 99 percent, effectively rendering the costly and wasteful resolution steps obsolete.
Mechanistic Insights into Sharpless Asymmetric Oxidation
The core of this technological advancement lies in the precise formation and activity of the chiral titanium-tartrate complex. The mechanism involves the in situ generation of a catalytically active species formed by the coordination of titanium isopropoxide with a chiral diol, specifically L-(+)-diethyl tartrate, in the presence of a controlled amount of water. This ternary complex creates a chiral pocket that directs the approach of the oxidant, cumene hydroperoxide, to the sulfur atom of the thioether substrate with high facial selectivity. The presence of the nitro group at the 4-position of the pyridine ring plays a crucial electronic role, potentially influencing the nucleophilicity of the sulfur center and stabilizing the transition state through electronic effects that differ from unsubstituted analogs. The water content, maintained at a molar ratio relative to titanium of approximately 0.1 to 2, is critical for activating the catalyst without causing hydrolysis that would degrade the chiral ligand integrity. This delicate balance ensures that the oxygen transfer occurs exclusively to generate the (R)-enantiomer, suppressing the formation of the (S)-enantiomer and preventing the non-selective background oxidation that typically leads to racemic mixtures or over-oxidation products.
Impurity control in this system is managed through both kinetic and thermodynamic factors inherent to the catalyst design. The use of a slight excess of oxidant, carefully calibrated to not exceed a 1:2.0 molar ratio, ensures complete conversion of the starting sulfide while minimizing the residence time of the product sulfoxide in the presence of the oxidizing agent, thus curbing further oxidation to the sulfone. The addition of an organic base, such as diisopropylethylamine, serves to neutralize any acidic byproducts generated during the decomposition of the hydroperoxide, which could otherwise catalyze racemization of the sensitive sulfoxide bond. Furthermore, the choice of solvent, with toluene being particularly effective, provides an optimal medium for the solubility of the organometallic catalyst while facilitating the precipitation of the final product upon cooling. This crystallization-driven isolation acts as a secondary purification step, further upgrading the optical purity of the crude material to meet the rigorous specifications required for subsequent coupling reactions in the synthesis of Dexlansoprazole.
How to Synthesize (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-efficiency results in a pilot or production setting. The procedure emphasizes the importance of pre-forming the catalyst complex before introducing the oxidant to ensure consistent stereoinduction throughout the batch. Operators must strictly adhere to the temperature profiles during the catalyst aging phase to guarantee the formation of the active dimeric or oligomeric titanium species. Following the oxidation step, the workup involves simple filtration or crystallization, bypassing the need for column chromatography which is a major cost driver in fine chemical manufacturing. For a comprehensive understanding of the specific operational parameters, including addition rates and quenching procedures, please refer to the standardized synthesis guide below.
- Prepare the chiral catalyst by mixing titanium isopropoxide, L-(+)-diethyl tartrate, and water in a solvent like toluene at 50-55°C.
- Add the sulfide substrate 2-[[[3-methyl-4-nitro-2-pyridyl]methyl]thio]benzimidazole and a base such as diisopropylethylamine to the mixture.
- Slowly add cumene hydroperoxide as the oxidant at controlled temperatures (20-40°C) to achieve high enantiomeric excess and yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this asymmetric oxidation technology translates directly into substantial cost reduction in pharmaceutical intermediate manufacturing. The primary economic driver is the dramatic improvement in material throughput; shifting from a resolution-based process with a maximum theoretical yield of 50 percent to a direct asymmetric synthesis with yields consistently above 90 percent effectively doubles the output from the same quantity of raw materials. This efficiency gain reduces the burden on upstream supply chains for starting materials like the sulfide precursor and significantly lowers the cost of goods sold (COGS). Moreover, the elimination of expensive chiral resolving agents, such as binaphthol derivatives or specialized enzymes, removes a volatile cost component from the bill of materials, stabilizing pricing structures for long-term contracts. The simplified downstream processing, which avoids complex chromatographic separations, also reduces the consumption of solvents and silica gel, contributing to both direct cost savings and a reduced environmental footprint.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chiral resolving agents and reduces solvent consumption by avoiding chromatographic purification steps. By achieving yields nearly double that of resolution methods, the effective cost per kilogram of the active intermediate is drastically lowered, allowing for more competitive pricing in the global API market without sacrificing margin.
- Enhanced Supply Chain Reliability: Operating at mild temperatures between 10 to 50 degrees Celsius reduces the dependency on specialized cryogenic equipment and energy-intensive cooling systems. This operational flexibility ensures that production can be maintained consistently across different geographical locations and seasons, mitigating the risk of batch failures due to thermal control issues and ensuring a steady supply of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The use of common industrial solvents like toluene and the avoidance of heavy metal catalysts or biological waste streams simplifies waste treatment protocols. The high selectivity of the reaction minimizes the generation of hazardous sulfone byproducts, reducing the load on effluent treatment plants and facilitating easier regulatory approval for commercial scale-up of complex pharmaceutical intermediates in environmentally regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric oxidation technology. These answers are derived directly from the experimental data and claims within patent CN102731478A, providing a factual basis for evaluating the process viability. Understanding these nuances is essential for technical teams assessing the fit of this route within their existing manufacturing infrastructure.
Q: What optical purity can be achieved with this asymmetric oxidation method?
A: According to patent CN102731478A, the method achieves extremely high optical purity, with enantiomeric excess (ee) values reaching up to 99.2% in optimized examples, significantly surpassing traditional resolution methods.
Q: How does this process compare to chemical resolution in terms of yield?
A: Traditional chemical resolution methods often suffer from theoretical maximum yields of 50% and practical yields around 40-50%. In contrast, this asymmetric oxidation route demonstrates yields exceeding 94%, effectively doubling the material efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (10-50°C) and avoids complex chromatographic separations required by inclusion resolution methods, making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the successful development of next-generation proton pump inhibitors. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and compliant. We are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm enantiomeric excess values matching the high standards set by the patent literature. Our commitment to quality assurance means that every batch of (R)-2-[[[3-methyl-4-nitro-2-pyridyl]methyl]sulfinyl]benzimidazole is thoroughly characterized to support your regulatory filings and clinical trial needs.
We invite you to engage with our technical procurement team to discuss how this advanced oxidation route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this high-yield methodology for your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a reliable partnership for your long-term API sourcing strategies.
