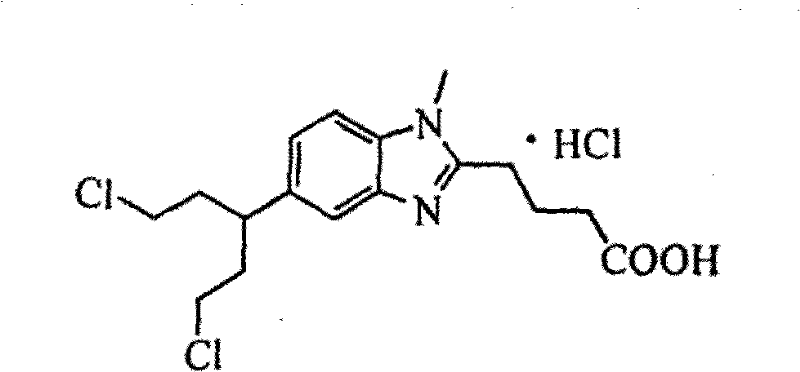
Advanced Synthesis of Bendamustine Hydrochloride for Commercial API Production
Advanced Synthesis of Bendamustine Hydrochloride for Commercial API Production
The global demand for oncology therapeutics continues to surge, placing immense pressure on supply chains to deliver high-quality active pharmaceutical ingredients (APIs) with consistent purity profiles. A pivotal advancement in this domain is detailed in Chinese Patent CN101691359A, which discloses a robust method for synthesizing highly pure bendamustine hydrochloride, the active ingredient in the blockbuster drug Treanda. This patent addresses critical bottlenecks in traditional manufacturing by introducing a streamlined four-step process that replaces hazardous and impurity-prone reagents with more manageable alternatives. By utilizing [1-methyl-2(4′-butanoic acid ethyl)-5-amino]-1H-benzimidazole as a starting material and employing phosphorus oxychloride for the key chlorination step, the inventors have achieved a purity profile exceeding 99.5% with single impurities controlled below 0.1%. This technical breakthrough not only meets stringent regulatory standards for raw materials but also offers a scalable pathway for reliable API intermediate suppliers aiming to optimize their production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of bendamustine hydrochloride faced significant hurdles regarding impurity control and operational complexity. As highlighted in the background technology of the patent, earlier methods, such as those disclosed in the 'Chinese Journal of New Drugs' (2007), relied heavily on thionyl chloride (SOCl2) for the chlorination of the hydroxyethyl side chains. While chemically feasible, this approach proved problematic on an industrial scale. The reaction with SOCl2 was observed to generate a substantial amount of by-products, leading to reaction mixtures that were difficult to purify. Furthermore, subsequent hydrolysis steps often utilized concentrated sulfuric acid under harsh conditions, resulting in dark, viscous residues that complicated filtration and isolation. Consequently, the final product from these conventional routes typically exhibited purity levels around 96.8%, failing to meet the rigorous single impurity threshold of less than 0.1% required for modern pharmaceutical applications.
The Novel Approach
The methodology presented in CN101691359A represents a paradigm shift in process chemistry for this alkylating agent. Instead of relying on thionyl chloride, the novel approach employs phosphorus oxychloride (POCl3) as the chlorinating agent. This substitution is not merely a change of reagent but a fundamental optimization of the reaction thermodynamics and kinetics. The new process eliminates the need for strict low-temperature control during the reagent addition phase, which was a prerequisite in older methods to mitigate exothermic risks and side reactions. By operating under reflux conditions in solvents like toluene or xylene, the reaction proceeds more smoothly, minimizing the formation of polymeric or degradation impurities. Additionally, the hydrolysis step is optimized using concentrated hydrochloric acid rather than sulfuric acid, which simplifies the workup procedure and prevents the charring associated with strong dehydrating agents. This holistic redesign of the synthetic route ensures that the final bendamustine hydrochloride is obtained as a clean, white solid suitable for direct formulation.
Mechanistic Insights into Phosphorus Oxychloride-Mediated Chlorination
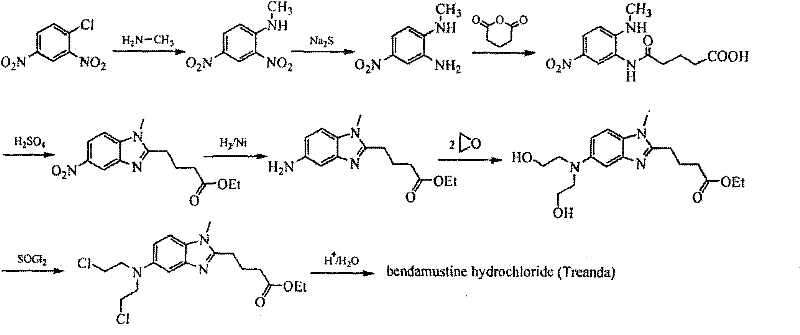
The core of this synthetic advancement lies in the efficient conversion of the dihydroxy intermediate into the bis-chloroethyl derivative. Mechanistically, the reaction begins with the nucleophilic attack of the amino group on ethylene oxide to form the N,N-bis(2-hydroxyethyl) species. In the subsequent chlorination step, phosphorus oxychloride acts as a potent electrophile, activating the hydroxyl groups for nucleophilic substitution by chloride ions. The patent specifies a molar ratio of the benzimidazole intermediate to phosphorus oxychloride ranging from 1:10 to 1:30, with a preferred range of 1:12 to 1:16. This excess of POCl3 serves a dual purpose: it drives the equilibrium towards complete conversion of the diol and acts as a solvent medium that stabilizes the transition state. Unlike SOCl2, which can generate gaseous by-products like SO2 and HCl that require scrubbing and can lead to pressure build-up, the POCl3 system remains largely contained within the liquid phase until the quenching stage. This containment reduces the risk of cross-contamination and allows for a more controlled reaction environment, which is critical for maintaining the integrity of the sensitive benzimidazole ring system.
Furthermore, the purification mechanism relies heavily on the differential solubility of the target compound versus its impurities in specific solvent systems. The patent details a recrystallization protocol using a mixture of tetrahydrofuran (THF) and water. The optimization of the volume ratio between these two solvents is crucial; a ratio of 8:1 to 20:1 (optimally 12:1 to 16:1) creates a solvent environment where the bendamustine hydrochloride has limited solubility upon cooling, while polar impurities and residual organic by-products remain dissolved in the mother liquor. This selective crystallization is the final barrier ensuring that the single impurity content remains below the critical 0.1% limit. The use of activated carbon during the hydrolysis workup further aids in removing colored impurities and trace organics, contributing to the high visual and chemical quality of the final API. This multi-stage purification strategy demonstrates a deep understanding of physical organic chemistry applied to process development.
How to Synthesize Bendamustine Hydrochloride Efficiently
Implementing this synthesis requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process is divided into four distinct stages: the initial alkylation with ethylene oxide, the chlorination with phosphorus oxychloride, the acid hydrolysis to form the salt, and the final recrystallization. Each step builds upon the previous one to incrementally increase purity while maintaining yield. Operators must pay close attention to temperature controls, particularly during the ethylene oxide addition which is conducted at -5 to 0°C, and the reflux times for the chlorination step which can range from 2 to 10 hours depending on the solvent choice. The following guide summarizes the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams looking to adopt this superior manufacturing route.
- React [1-methyl-2(4'-butanoic acid ethyl)-5-amino]-1H-benzimidazole with ethylene oxide in water/acetic acid at -5 to 0°C to form the dihydroxy intermediate.
- Perform chlorination using phosphorus oxychloride (POCl3) in toluene or xylene under reflux conditions, maintaining a molar ratio of 1: 10 to 1:30.
- Hydrolyze the chlorinated residue with concentrated hydrochloric acid under reflux, followed by decolorization with activated carbon.
- Purify the crude product via recrystallization using a tetrahydrofuran and water mixture (volume ratio 8-20: 1) to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The shift away from thionyl chloride and sulfuric acid mitigates several logistical and safety risks inherent in the legacy process. By simplifying the reaction conditions and eliminating the need for extreme cryogenic control during the chlorination phase, the process becomes significantly more robust and easier to scale. This robustness translates directly into reduced operational downtime and lower energy consumption, as the facility does not need to maintain specialized low-temperature infrastructure for extended periods. Moreover, the higher purity of the crude product prior to recrystallization means that fewer resources are expended on downstream purification, effectively lowering the cost of goods sold (COGS) through improved process efficiency.
- Cost Reduction in Manufacturing: The replacement of thionyl chloride with phosphorus oxychloride eliminates the need for complex gas scrubbing systems required to handle sulfur dioxide emissions, thereby reducing capital expenditure on environmental control equipment. Additionally, the simplified workup procedure, which avoids the formation of sticky, black residues, minimizes product loss during filtration and transfer operations. This improvement in material throughput ensures that a higher percentage of raw materials are converted into saleable API, driving down the effective cost per kilogram. The ability to use common industrial solvents like toluene and xylene, which are readily available and cost-effective, further enhances the economic viability of the process compared to routes requiring specialized or exotic reagents.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including ethylene oxide, phosphorus oxychloride, and tetrahydrofuran, are commodity chemicals with stable global supply chains. This reduces the risk of production stoppages due to raw material shortages, a common issue with specialized reagents. The shortened synthesis cycle time, as noted in the patent, allows for faster batch turnover, enabling manufacturers to respond more agilely to market demand fluctuations. By establishing a production line based on this reliable method, companies can secure a consistent supply of high-purity bendamustine hydrochloride, ensuring continuity for downstream drug product manufacturing and reducing the risk of stockouts for critical cancer therapies.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, with examples demonstrating successful execution in multi-liter reactors. The absence of harsh sulfuric acid hydrolysis reduces the generation of acidic waste streams that require neutralization and disposal, aligning the process with increasingly stringent environmental regulations. The cleaner reaction profile means less solvent is needed for washing and purification, reducing the overall solvent footprint of the manufacturing campaign. This environmental efficiency not only lowers waste disposal costs but also positions the manufacturer as a sustainable partner, a key consideration for multinational pharmaceutical companies evaluating their supplier base for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of bendamustine hydrochloride using the patented POCl3 method. These answers are derived directly from the experimental data and comparative analysis provided in CN101691359A, offering clarity on why this route is superior to legacy methods. Understanding these nuances is essential for technical teams evaluating process transfers and for procurement professionals assessing supplier capabilities.
Q: Why is phosphorus oxychloride preferred over thionyl chloride for bendamustine synthesis?
A: The use of thionyl chloride (SOCl2) in conventional methods often leads to excessive impurity formation, resulting in black, sticky residues that are difficult to purify. In contrast, phosphorus oxychloride (POCl3) allows for milder reaction conditions without strict low-temperature control during feeding, significantly reducing side reactions and enabling the production of API-grade material with single impurities below 0.1%.
Q: What is the critical parameter for achieving high purity in the final recrystallization step?
A: The solvent system and its ratio are paramount. The patent specifies using a mixed solvent of tetrahydrofuran (THF) and water. Optimization experiments determined that a volume ratio between 8:1 and 20:1 (preferably 12:1 to 16:1) is essential. This specific polarity balance ensures that impurities remain in the mother liquor while the target bendamustine hydrochloride crystallizes out as a high-purity white solid.
Q: How does this novel route impact the overall yield compared to prior art?
A: Conventional routes using SOCl2 and sulfuric acid hydrolysis reported total yields around 59.0% with purity levels only reaching 96.8%. The optimized process described in CN101691359A utilizes POCl3 chlorination and optimized hydrolysis conditions to achieve total yields exceeding 60%, with specific examples demonstrating yields up to 68.7% and purity greater than 99.5%, representing a significant improvement in both efficiency and quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bendamustine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex oncology APIs like bendamustine hydrochloride demands more than just chemical knowledge; it requires a commitment to excellence in every batch. Our technical team has extensively analyzed the pathways described in CN101691359A and possesses the expertise to implement this high-purity route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical single impurity limits of less than 0.1%, guaranteeing that every shipment meets the highest global pharmacopeial standards.
We invite potential partners to engage with us to explore how our advanced manufacturing capabilities can support your supply chain needs. By leveraging our optimized processes, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how our efficient route translates into value for your organization. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on transparent, high-quality data.
