Revolutionizing Bendamustine Hydrochloride Production: A Stable, High-Yield Purification Strategy for Global Pharma
Introduction to Advanced Purification Technologies in Oncology
The development of robust purification processes for potent oncology agents is critical for ensuring patient safety and therapeutic efficacy. Patent CN101948437B introduces a groundbreaking refining method for bendamustine hydrochloride, a dual-functional alkylating agent widely used in the treatment of chronic lymphocytic leukemia and non-Hodgkin lymphoma. This technology addresses the inherent instability of the bendamustine molecule during traditional recrystallization processes, which often suffer from significant degradation and impurity formation. By utilizing a specific concentration range of hydrochloric acid solution as the refining solvent, the process effectively suppresses hydrolytic degradation pathways that plague conventional water or organic solvent-based methods. This innovation not only enhances the chemical stability of the active pharmaceutical ingredient (API) during processing but also ensures that the final product meets the rigorous purity specifications required for injectable formulations, thereby setting a new standard for reliability in the supply of high-quality anticancer drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for bendamustine hydrochloride have historically relied on solvents such as purified water, ethanol-water mixtures, or tetrahydrofuran (THF) systems, each presenting distinct chemical challenges that compromise product quality. When purified water is used, the bis(2-chloroethyl)amine moieties within the bendamustine structure are highly susceptible to hydrolysis, leading to the formation of hydroxyethyl impurities that are difficult to remove and reduce the overall potency of the drug. Similarly, the use of ethanol-water mixtures introduces the risk of esterification reactions at elevated temperatures, where the carboxylic acid group reacts with the alcohol to form ester byproducts, further complicating the impurity profile. Furthermore, processes utilizing THF involve significant operational hazards due to the solvent's low boiling point and tendency to cause bumping during water addition, alongside concerns regarding residual solvent toxicity and environmental impact. These conventional approaches often result in low refining yields, typically necessitating multiple recrystallization cycles that erode production efficiency and increase manufacturing costs.
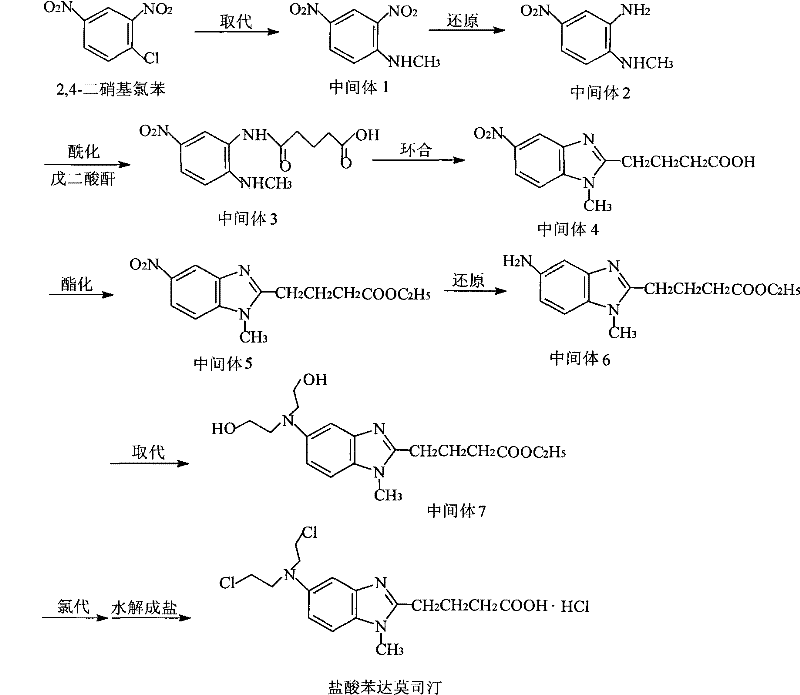
The synthetic complexity of bendamustine, as illustrated in the reaction pathway, underscores the necessity for a gentle yet effective purification step to preserve the integrity of the final molecule after multiple transformation stages. The presence of reactive functional groups makes the crude product particularly vulnerable to the harsh conditions often encountered in standard solvent systems. For instance, the hydrolysis mechanism depicted in the degradation pathway highlights how water molecules attack the chloroethyl groups, cleaving the carbon-chlorine bonds and rendering the alkylating capability of the drug ineffective. This chemical vulnerability means that any purification method exposing the crude material to neutral or basic aqueous environments will inevitably lead to a decline in assay values and an increase in related substances, failing to meet the strict regulatory thresholds for single impurities which are often required to be below 0.1% for parenteral products.
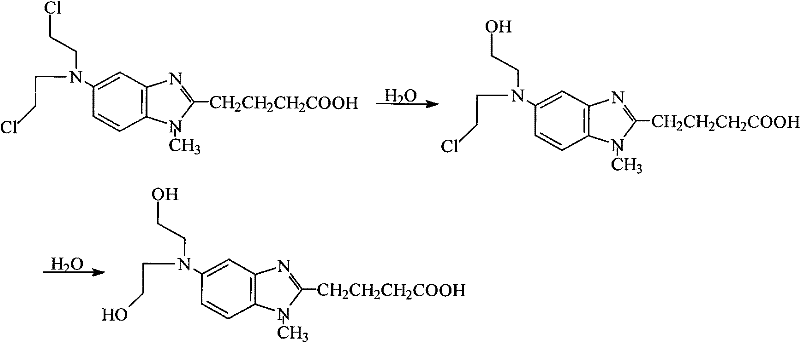
The Novel Approach
The novel approach detailed in the patent data circumvents these stability issues by employing a dilute hydrochloric acid solution with a concentration ranging from 0.01 mol/L to 2 mol/L as the exclusive refining medium. This acidic environment plays a crucial role in protonating the nitrogen atoms within the benzimidazole ring and the amine side chains, thereby electronically deactivating the molecule towards nucleophilic attack by water molecules. By maintaining the pH in an acidic range, the rate of hydrolysis of the mustard groups is drastically reduced, allowing the crude material to be dissolved at temperatures between 30°C and 100°C without significant decomposition. This method enables a single-step recrystallization process that effectively separates the target API from both organic byproducts and inorganic salts, achieving a purity level exceeding 99.5% with a refining yield that consistently remains above 90%. The simplicity of using a common, non-toxic reagent like hydrochloric acid also eliminates the need for complex solvent recovery systems associated with THF or ethanol, streamlining the manufacturing workflow and enhancing the overall economic viability of the production process.
Mechanistic Insights into Acid-Stabilized Recrystallization
The core mechanistic advantage of this purification technology lies in the stabilization of the bendamustine hydrochloride structure through acid-base chemistry interactions. As shown in the molecular structure, the compound features a carboxylic acid tail and a nitrogen mustard head, both of which are sensitive to pH changes. In a neutral aqueous environment, the lone pair electrons on the nitrogen atoms can facilitate the intramolecular cyclization or intermolecular hydrolysis of the chloroethyl groups. However, in the presence of excess protons provided by the hydrochloric acid solvent, these nitrogen centers become protonated, forming stable ammonium salts that resist nucleophilic substitution reactions. This protonation effect effectively 'locks' the reactive mustard groups, preventing the formation of hydroxyethyl degradation products that are commonly observed in water-based refinements. Additionally, the high solubility of the hydrochloride salt in the acidic medium at elevated temperatures ensures complete dissolution of the crude material, while the sharp decrease in solubility upon cooling promotes the formation of large, well-defined crystals that exclude impurities from the lattice structure.
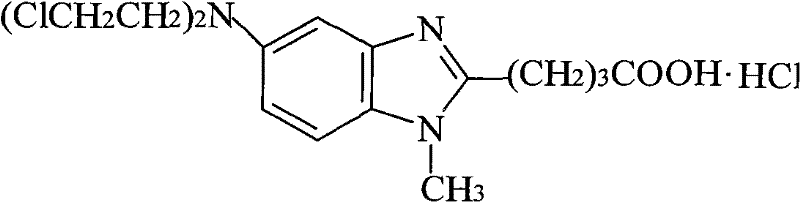
Furthermore, the control of impurity profiles is significantly enhanced by the selective solubility characteristics of the acidic system. Impurities generated during the upstream synthesis, such as unreacted intermediates or over-alkylated byproducts, often possess different pKa values or solubility profiles compared to the target bendamustine hydrochloride. The acidic recrystallization medium exploits these differences, keeping unwanted impurities in the mother liquor while allowing the pure API to precipitate out as a white crystalline solid. The process also allows for the optional addition of activated carbon during the hot dissolution phase, which provides an additional layer of purification by adsorbing colored impurities and high-molecular-weight organic contaminants. This combination of chemical stabilization via pH control and physical purification via crystallization and adsorption results in a product with exceptional optical clarity and chemical homogeneity, satisfying the stringent visual and chromatographic requirements of modern pharmacopoeias for injectable oncology therapies.
How to Synthesize Bendamustine Hydrochloride Efficiently
The implementation of this purification protocol requires precise control over solvent concentration and temperature parameters to maximize yield and purity. The process begins with the preparation of the hydrochloric acid solution within the specified molarity range, followed by the dissolution of the crude bendamustine hydrochloride under heated conditions to ensure a homogeneous mixture. This streamlined approach minimizes unit operations and reduces the potential for product loss during transfer and filtration steps, making it highly suitable for large-scale industrial application where consistency and throughput are paramount.
- Dissolve 1 part by weight of crude bendamustine hydrochloride in 3-10 parts of 0.01-2 mol/L hydrochloric acid solution at 30-100°C.
- Perform hot filtration to remove insoluble impurities, optionally adding activated carbon for decolorization if necessary.
- Cool the filtrate to induce crystallization, filter the resulting white crystals, and dry to obtain high-purity bendamustine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acid-based purification method offers substantial strategic benefits regarding cost efficiency and supply reliability. By eliminating the need for expensive and hazardous organic solvents like THF, the process significantly reduces raw material costs and simplifies waste management protocols, leading to a lower overall cost of goods sold (COGS). The high refining yield of over 90% means that less starting crude material is required to produce a given amount of finished API, optimizing the utilization of valuable precursors and reducing the burden on upstream synthesis capacity. Moreover, the robustness of the process against degradation ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by out-of-specification results and reprocessing requirements. This reliability is crucial for maintaining continuous supply lines to downstream formulation partners, especially in the competitive oncology market where drug shortages can have severe clinical consequences.
- Cost Reduction in Manufacturing: The substitution of complex solvent systems with dilute hydrochloric acid removes the necessity for specialized solvent recovery infrastructure and reduces the energy consumption associated with distilling high-boiling or azeotropic mixtures. This simplification of the downstream processing train translates directly into lower utility costs and reduced capital expenditure for plant equipment. Additionally, the high purity achieved in a single crystallization step negates the need for repetitive refining cycles, further saving on labor and processing time while maximizing the output from each production batch.
- Enhanced Supply Chain Reliability: The use of commodity chemicals like hydrochloric acid ensures that the supply of refining reagents is secure and unaffected by the volatility often seen in the specialty solvent market. The process's tolerance to a wide range of operating temperatures and acid concentrations provides operational flexibility, allowing manufacturers to adapt to varying production scales without compromising product quality. This resilience contributes to a more stable supply chain, reducing the likelihood of disruptions and ensuring that pharmaceutical partners can rely on timely deliveries of high-quality bendamustine hydrochloride for their drug product manufacturing schedules.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener alternative to THF-based processes by avoiding the use of Class 2 solvents with strict residual limits. The aqueous acidic waste streams generated are easier to treat and neutralize compared to organic solvent wastes, facilitating compliance with increasingly stringent environmental regulations. The scalability of the crystallization process is excellent, as the thermodynamics of salt precipitation in acidic media are well-understood and easily transferred from pilot scale to multi-ton commercial production, supporting the growing global demand for bendamustine-based therapies without requiring extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability and quality parameters of bendamustine hydrochloride produced via this innovative method. Understanding these details is essential for quality assurance teams evaluating the suitability of this material for clinical and commercial use. These insights demonstrate the thorough validation of the process against international quality standards.
Q: Why is hydrochloric acid preferred over water or ethanol for refining bendamustine?
A: Bendamustine contains reactive mustard groups that undergo hydrolysis in neutral water and esterification in ethanol at elevated temperatures. Dilute hydrochloric acid stabilizes the molecule, preventing these degradation pathways and ensuring higher purity.
Q: What purity levels can be achieved with this acidic recrystallization method?
A: This method consistently achieves a product purity of greater than 99.5%, with single impurity levels controlled below 0.1%, meeting stringent ICH guidelines for injectable raw materials.
Q: Does this process improve the overall yield compared to traditional solvent systems?
A: Yes, by minimizing degradation during the dissolution and crystallization phases, the refining yield is maintained at not lower than 90%, significantly reducing material loss compared to aqueous or THF-based methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bendamustine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced purification technologies like the acid-mediated recrystallization process to deliver bendamustine hydrochloride of unparalleled quality and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch exceeds the 99.5% purity threshold with single impurities controlled below 0.1%, guaranteeing safety and efficacy for the end patient. Our commitment to technical excellence allows us to navigate the complexities of oncology API manufacturing with precision and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our superior process capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our refined bendamustine hydrochloride can enhance your drug development pipeline and commercial production efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
