Scalable Resolution of Optically Active Diazaspiro[4.5]decane Derivatives for Commercial API Production
Scalable Resolution of Optically Active Diazaspiro[4.5]decane Derivatives for Commercial API Production
The pharmaceutical industry continuously seeks robust methodologies for producing chiral intermediates with high optical purity, particularly for targets involving serotonin regulation. Patent CN110177790B discloses a groundbreaking process for the resolution of optically active diazaspiro[4.5]decane derivatives, which serve as critical intermediates in the synthesis of Tryptophan Hydroxylase 1 (TPH1) inhibitors. These inhibitors are pivotal for treating diseases associated with peripheral serotonin, including gastrointestinal disorders and metabolic conditions. The disclosed technology addresses the significant challenge of separating enantiomers in spirocyclic systems, offering a pathway that transitions effectively from laboratory discovery to industrial application. By leveraging diastereomeric salt formation with specific chiral acids, the process achieves exceptional enantiomeric enrichment without relying on costly preparative chiral chromatography. This innovation represents a substantial leap forward for manufacturers aiming to secure a reliable supply chain for high-value neurological and metabolic therapeutics.
![General structure of optically active diazaspiro[4.5]decane isomers I-(S) and I-(R)](/insights/img/diazaspiro-decane-resolution-pharma-intermediate-supplier-20260308200344-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the separation of enantiomers for complex spirocyclic scaffolds like diazaspiro[4.5]decane has relied heavily on chiral high-performance liquid chromatography (HPLC) or enzymatic kinetic resolution. While effective on a small scale, chiral HPLC is notoriously difficult to scale due to the high cost of chiral stationary phases and the limited loading capacity of columns, leading to prohibitive operational expenses in commercial manufacturing. Furthermore, enzymatic methods often suffer from narrow substrate specificity, requiring extensive screening to find a biocatalyst that accepts the bulky spirocyclic structure, which can delay project timelines significantly. Another common issue with conventional resolution techniques is the inability to recycle the unwanted isomer efficiently, resulting in a maximum theoretical yield of only 50% unless dynamic kinetic resolution is employed, which adds further complexity and cost to the process. These limitations create bottlenecks in the supply chain, making it difficult for procurement teams to secure large quantities of intermediates at a viable cost point for widespread therapeutic use.
The Novel Approach
The novel approach detailed in the patent utilizes a classical yet highly optimized diastereomeric salt resolution strategy using 2,3:5,6-di-O-isopropylidene-2-keto-L-gulonic acid (gulonic acid) as the resolving agent. This method capitalizes on the distinct solubility differences between the salt of the desired (S)-isomer and the undesired (R)-isomer in specific solvent systems like 2-methyltetrahydrofuran. A key innovation is the use of catalytic amounts of an aldehyde, such as benzaldehyde, which appears to facilitate the equilibration or crystallization kinetics, thereby enhancing the efficiency of the resolution. This process allows for the enrichment of the (S)-isomer to levels exceeding 90% ee in the initial salt formation, which can be further purified to >99% ee through recrystallization. By shifting the separation mechanism from adsorption-based chromatography to precipitation-based crystallization, the process drastically reduces solvent consumption and equipment footprint, offering a more sustainable and economically attractive route for large-scale production.
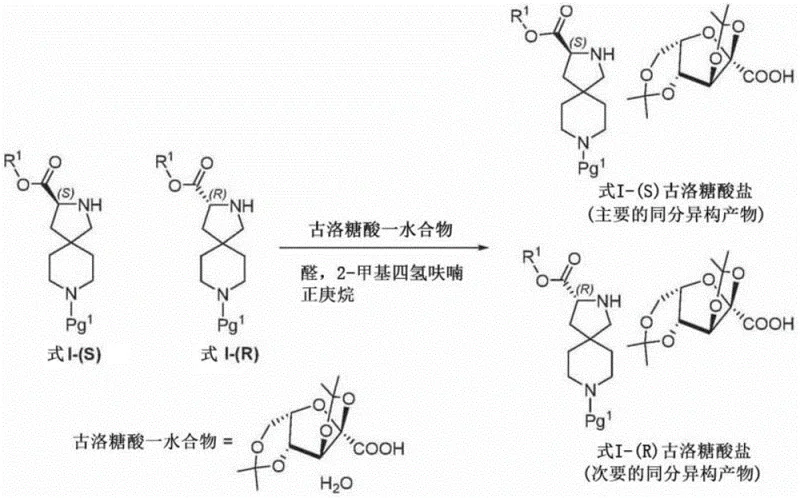
Mechanistic Insights into Diastereomeric Salt Resolution
The core mechanism driving this resolution lies in the stereoselective interaction between the basic nitrogen of the diazaspiro[4.5]decane scaffold and the chiral carboxylic acid moiety of the gulonic acid derivative. When the racemic mixture reacts with the chiral acid, two diastereomeric salts are formed: one comprising the (S)-amine and the L-gulonic acid, and the other comprising the (R)-amine and the L-gulonic acid. Due to the rigid spirocyclic geometry, these two salts pack differently in the crystal lattice, leading to divergent thermodynamic stability and solubility profiles in organic solvents. The presence of the acetonide protecting groups on the gulonic acid enhances its lipophilicity, ensuring that the resulting salts are soluble enough in ethers like 2-methyltetrahydrofuran to allow for dynamic equilibration during the heating phase, yet insoluble enough to precipitate upon cooling. The addition of benzaldehyde likely acts as a transient catalyst or crystal habit modifier, potentially interacting with the amine or the solvent matrix to lower the activation energy for the nucleation of the less soluble (S)-salt, thereby kinetically favoring its precipitation over the (R)-salt.
Impurity control in this system is managed through the rigorous optimization of stoichiometry and temperature profiles. The patent specifies using approximately 1 molar equivalent of the resolving acid relative to the total amine mixture, which ensures that the equilibrium is driven towards salt formation without excessive excess reagent that could co-crystallize. Following the initial salt formation, a recrystallization step is employed where the crude salt mixture is dissolved and slowly cooled, allowing the purer (S)-salt to crystallize while the (R)-salt remains in the mother liquor. This fractional crystallization is highly effective at rejecting impurities, including the unwanted enantiomer and any residual starting materials or byproducts from the upstream hydrogenation step. The final liberation of the free base using a mild base like sodium carbonate ensures that the sensitive ester functionalities on the spirocycle remain intact, preserving the integrity of the molecule for downstream coupling reactions required to build the final TPH1 inhibitor.
How to Synthesize Diazaspiro[4.5]decane Intermediates Efficiently
The synthesis of these high-purity intermediates begins with the preparation of the racemic amine mixture, typically achieved via hydrogenation of an oxadiazaspiro precursor using Raney Nickel, followed by the critical resolution step described above. The process is designed to be telescoped where possible, minimizing isolation steps to improve overall yield and reduce waste generation. Operators must carefully control the temperature during the salt formation, maintaining ranges between 30°C and 40°C to ensure complete dissolution before initiating the crystallization phase. Detailed standardized synthetic steps for the preparation of the racemic precursor, the resolution conditions, and the subsequent deprotection strategies are outlined in the comprehensive guide below, providing a clear roadmap for technical teams to replicate this high-efficiency protocol in their own facilities.
- Prepare a starting mixture containing isomeric compounds of formula I-(S) and I-(R) via hydrogenation of the corresponding oxadiazaspiro precursor.
- React the starting mixture with 2,3: 5,6-di-O-isopropylidene-2-keto-L-gulonic acid monohydrate in the presence of a catalytic amount of benzaldehyde to form a diastereomeric salt mixture.
- Purify the salt mixture through recrystallization in solvents like 2-methyltetrahydrofuran to enrich the I-(S) gulonate salt, followed by basification to recover the free base with >90% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology offers transformative benefits regarding cost structure and supply security. By replacing chromatographic purification with crystallization, the process eliminates the need for expensive chiral columns and the associated high-volume solvent disposal costs, leading to a significantly reduced cost of goods sold (COGS). The reliance on commercially available resolving agents like gulonic acid derivatives ensures that raw material sourcing is stable and not subject to the volatility often seen with specialized chiral catalysts. Furthermore, the robustness of the crystallization process means that batch-to-batch variability is minimized, reducing the risk of production delays caused by failed quality control tests. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the demanding delivery timelines of global pharmaceutical partners.
- Cost Reduction in Manufacturing: The shift from chromatography to crystallization fundamentally alters the economic model of producing chiral spiro-intermediates. Crystallization equipment is standard in almost every chemical plant, whereas chiral prep-HPLC requires specialized, high-capital investment units that are often bottlenecks in production. By utilizing standard reactors and filters, manufacturers can leverage existing infrastructure to produce tons of material rather than kilograms, achieving economies of scale that drive down the unit price. Additionally, the ability to recycle the mother liquor containing the unwanted (R)-isomer, potentially racemizing it for re-entry into the process, further enhances the atom economy and reduces raw material waste, contributing to substantial long-term cost savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the chemical steps involved. The reagents used, such as benzaldehyde and sodium carbonate, are commodity chemicals with multiple global suppliers, mitigating the risk of single-source dependency. The process operates under mild conditions (30°C to 40°C) and ambient pressure, reducing the safety risks associated with high-pressure hydrogenation or cryogenic reactions, which in turn lowers insurance and compliance overheads. This operational safety profile ensures that production facilities can run continuously without unplanned shutdowns, guaranteeing a steady flow of intermediates to downstream API manufacturers and preventing stockouts that could disrupt clinical trials or commercial launches.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns well with green chemistry principles by reducing solvent intensity. Crystallization typically requires less solvent volume per kilogram of product compared to column chromatography, resulting in a smaller environmental footprint and lower waste treatment costs. The use of 2-methyltetrahydrofuran, a solvent derived from renewable resources, further enhances the sustainability profile of the manufacturing route. As regulatory bodies increasingly scrutinize the environmental impact of pharmaceutical manufacturing, adopting such eco-friendly processes provides a competitive advantage, facilitating smoother regulatory approvals and enhancing the corporate social responsibility standing of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What is the primary advantage of using 2,3:5,6-di-O-isopropylidene-2-keto-L-gulonic acid for resolution?
A: This resolving agent forms diastereomeric salts with significantly different solubility profiles, allowing for effective enrichment of the desired (S)-isomer through simple crystallization rather than expensive chromatography.
Q: Can this resolution process be scaled for industrial manufacturing?
A: Yes, the process utilizes standard unit operations such as crystallization and filtration, avoiding complex chiral columns, which makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What level of enantiomeric excess can be achieved with this method?
A: The patent demonstrates that after purification steps, the enantiomeric excess of the I-(S) isomer can reach greater than 99%, meeting stringent purity specifications for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazaspiro[4.5]decane Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing chiral resolution processes like the one described in CN110177790B, ensuring that we meet stringent purity specifications with rigorous QC labs dedicated to chiral analysis. We understand the critical nature of TPH1 inhibitor intermediates and are committed to delivering materials that support your drug development timelines with unmatched consistency and quality assurance.
We invite you to engage with our technical procurement team to discuss how we can tailor this resolution process to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing protocols can reduce your overall project costs. Contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex pharmaceutical intermediate manufacturing.
