Advanced Levetiracetam Manufacturing Process Enhancing Purity and Commercial Scalability for Global Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiepileptic medications, and patent CN110698379A presents a significant advancement in the preparation of levetiracetam. This specific intellectual property outlines a refined synthetic route that addresses longstanding challenges regarding product purity, specifically the control of the R-isomer and levetiracetam acid impurities, which are critical quality attributes for regulatory compliance. The disclosed method leverages a multi-step sequence starting from (S)-2-aminobutyric acid, utilizing specific additives like oleic acid and chlorobutanol to optimize reaction kinetics and thermodynamic outcomes. By integrating these novel process parameters, manufacturers can achieve a more consistent impurity profile compared to conventional methods described in prior art such as EP0162036B. The structural integrity of the final active pharmaceutical ingredient is paramount, as shown in the molecular configuration below, which dictates its binding affinity to synaptic vesicle protein 2A. 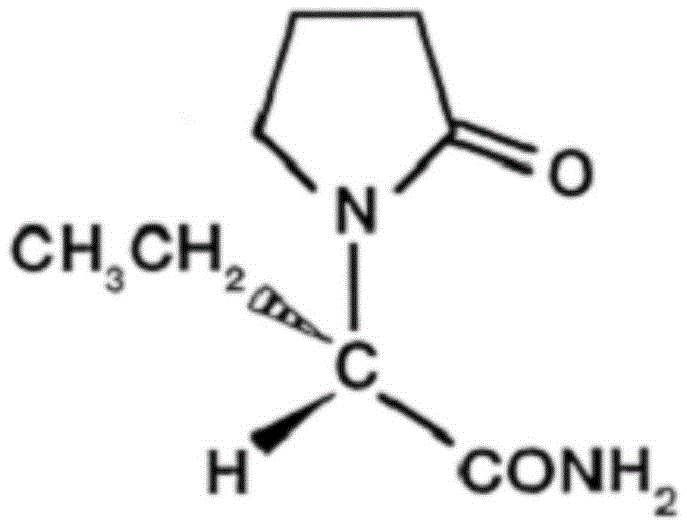
Furthermore, the economic implications of adopting this patented technology extend beyond mere yield improvements, touching upon the broader supply chain reliability for a reliable levetiracetam supplier. The process eliminates the need for complex chiral resolution steps often required in older methodologies, thereby streamlining the production timeline and reducing the consumption of expensive resolving agents. This simplification directly contributes to cost reduction in API manufacturing by minimizing unit operations and solvent usage during purification stages. For procurement managers, understanding the technical nuances of this synthesis is essential for evaluating potential partners who can demonstrate mastery over these specific reaction conditions. The ability to consistently produce high-purity levetiracetam with minimal enantiomeric excess deviations is a key differentiator in the competitive generic pharmaceutical market. Consequently, this report delves deep into the mechanistic and operational details to provide a comprehensive view for technical decision-makers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of levetiracetam has been plagued by issues related to low overall yields and difficult purification processes, particularly when attempting to control stereochemistry at the alpha-carbon position. Traditional routes often rely on harsh reaction conditions or expensive chiral catalysts that do not guarantee the suppression of the unwanted R-enantiomer, leading to costly recycling loops or material loss. Many prior art methods, such as those involving direct alkylation without specific additives, struggle to maintain impurity levels below the stringent thresholds required by pharmacopeias like the USP or EP. The use of thionyl chloride in earlier protocols often resulted in significant side reactions if temperature controls were not meticulously maintained, generating chlorinated byproducts that are difficult to remove via standard crystallization. Additionally, the lack of specific impurity suppressors in conventional cyclization steps frequently leads to elevated levels of levetiracetam acid, necessitating multiple recrystallization steps that erode overall process efficiency. These inefficiencies translate into higher production costs and longer lead times, creating bottlenecks for supply chain heads managing inventory for high-demand neurological medications.
The Novel Approach
In contrast, the methodology disclosed in CN110698379A introduces a strategic intervention through the use of oleic acid and chlorobutanol, which fundamentally alters the reaction landscape to favor the desired S-enantiomer. This novel approach mitigates the formation of key impurities at the source rather than relying solely on downstream purification, representing a paradigm shift in process chemistry design. By carefully modulating the reaction environment with these specific additives, the process achieves a remarkable improvement in crude product quality, reducing the burden on final purification steps. The integration of a phase transfer catalyst in the cyclization stage further enhances the reaction rate under milder conditions, preserving the integrity of the chiral center. This results in a more robust process window where variations in raw material quality have less impact on the final outcome, ensuring greater batch-to-batch consistency. For commercial scale-up of complex pharmaceutical intermediates, this level of control is indispensable for maintaining regulatory approval and market supply continuity without unexpected quality deviations.
Mechanistic Insights into Oleic Acid and Chlorobutanol Assisted Cyclization
The first critical stage involves the esterification of (S)-2-aminobutyric acid with thionyl chloride in a lower alcohol solvent, typically methanol, to form the ester intermediate. As illustrated in the reaction scheme, this step requires precise temperature management, initially maintaining 20-30°C during addition and then heating to 40-50°C to drive the reaction to completion while removing generated gases. 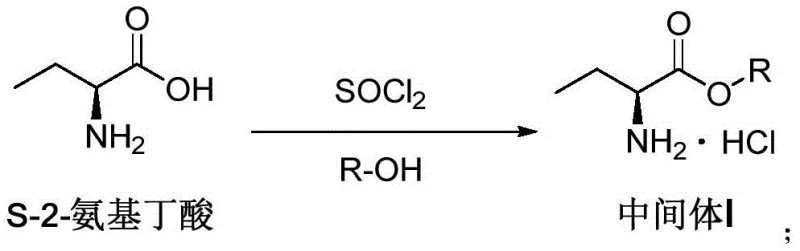
Following esterification, the subsequent ammonolysis reaction converts the ester into the corresponding amide, a transformation that is significantly enhanced by the presence of oleic acid. The mechanism by which oleic acid improves yield is believed to involve the stabilization of intermediate species or the modification of the solvent interface, although the exact physicochemical interactions remain a subject of specialized study. This step proceeds with aqueous ammonia at low temperatures, followed by salification with hydrochloric acid to isolate the intermediate as a stable salt, ensuring high recovery rates before proceeding to the final cyclization. The second reaction scheme details this conversion, highlighting the retention of stereochemistry which is crucial for the biological activity of the final drug substance. 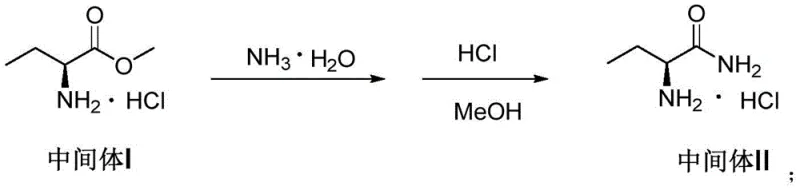
The final and most critical step is the intramolecular cyclization where the amino group attacks the terminal chloride of the butyryl chain to form the pyrrolidone ring. This reaction is conducted in dichloromethane with potassium hydroxide as the base and a quaternary ammonium salt as the phase transfer catalyst to facilitate ion transport between phases. Crucially, the addition of chlorobutanol during the addition of 4-chlorobutyryl chloride acts as an impurity suppressor, likely by competing for side reaction pathways or stabilizing the transition state to prevent racemization. The reaction is kept at sub-zero temperatures, specifically between -15°C and 0°C, to minimize thermal degradation and ensure high enantiomeric purity. The final reaction pathway demonstrates the convergence of these intermediates into the target molecule, showcasing the elegance of this optimized synthetic route. 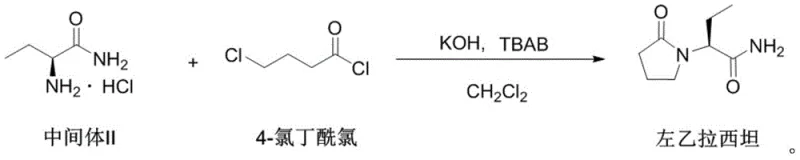
How to Synthesize Levetiracetam Efficiently
To implement this synthesis effectively, operators must adhere to strict stoichiometric ratios and temperature profiles as defined in the patent examples to replicate the high yields and purity levels reported. The process begins with the suspension of the starting amino acid in methanol, followed by the controlled dropwise addition of thionyl chloride to manage exothermic heat release effectively. Subsequent steps involve precise pH adjustments and solvent swaps, requiring experienced technical oversight to ensure that each intermediate meets the necessary quality specifications before moving to the next stage. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in validating this route within their own facilities.
- Prepare Intermediate I by reacting (S)-2-aminobutyric acid with thionyl chloride in lower alcohol at controlled temperatures between 20-50°C.
- Convert Intermediate I to salified Intermediate II using ammonia water and hydrochloric acid, incorporating oleic acid to significantly improve reaction yield.
- Perform cyclization with 4-chlorobutyryl chloride in dichloromethane using KOH and phase transfer catalysts, adding chlorobutanol to reduce key impurities.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this optimized synthesis route offers substantial benefits regarding supply chain resilience and cost structure optimization for buyers of antiepileptic intermediates. The reliance on commodity chemicals such as methanol, dichloromethane, and potassium hydroxide ensures that raw material availability remains high, reducing the risk of supply disruptions caused by niche reagent shortages. Furthermore, the elimination of complex chiral resolution steps significantly reduces the consumption of expensive resolving agents and solvents, leading to a leaner manufacturing process with a smaller environmental footprint. This efficiency translates into a more competitive pricing structure without compromising on the stringent quality standards required for pharmaceutical grade materials. For supply chain heads, the predictability of this process allows for more accurate demand forecasting and inventory planning, essential for maintaining continuous production of life-saving medications.
- Cost Reduction in Manufacturing: The streamlined process design eliminates several unit operations associated with traditional resolution methods, directly lowering labor and utility costs per kilogram of produced API. By improving the crude product quality through the use of chlorobutanol, the need for extensive recrystallization is reduced, saving both time and solvent resources during the finishing stages. The higher overall yield means that less starting material is required to produce the same amount of final product, optimizing the cost of goods sold significantly. These cumulative efficiencies allow suppliers to offer more attractive commercial terms while maintaining healthy margins for reinvestment in quality assurance and capacity expansion.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials like (S)-2-aminobutyric acid ensures that production is not bottlenecked by the supply of exotic or proprietary reagents that may have long lead times. The robustness of the reaction conditions, with wide operating windows for temperature and stoichiometry, reduces the likelihood of batch failures due to minor process deviations. This reliability is critical for reducing lead time for high-purity antiepileptic drugs, ensuring that downstream formulation manufacturers receive their materials on schedule. A stable supply of high-quality intermediates supports the overall security of the global pharmaceutical supply chain against unforeseen disruptions.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, avoiding the use of highly toxic benzene or other hazardous solvents that pose significant waste disposal challenges. The simplified workup procedures generate less chemical waste, aligning with modern green chemistry principles and reducing the cost of environmental compliance and waste treatment. Scaling this process from pilot to commercial tonnage is straightforward due to the use of standard reactor types and common separation techniques like filtration and distillation. This ease of scale-up facilitates rapid capacity expansion to meet growing global demand for levetiracetam without requiring massive capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific levetiracetam synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical evaluators. Understanding these details helps stakeholders assess the feasibility of integrating this supply source into their existing vendor qualification programs. Comprehensive responses below cover aspects of impurity control, yield optimization, and regulatory compliance.
Q: How does the addition of oleic acid impact the levetiracetam synthesis yield?
A: According to patent CN110698379A, adding 0.2 to 0.3 equivalents of oleic acid during the ammonolysis step surprisingly improves the total yield of the first two steps from approximately 68% to over 92%, ensuring better material efficiency.
Q: What specific impurity control measures are implemented in this novel process?
A: The process incorporates 0.1 to 0.2 equivalents of chlorobutanol during the acylation and cyclization stages, which significantly reduces the content of the R-isomer impurity to below 0.1% and minimizes levetiracetam acid levels.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method utilizes readily available starting materials like (S)-2-aminobutyric acid and common solvents such as dichloromethane and methanol, avoiding highly toxic reagents and complex resolution steps, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levetiracetam Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in actual manufacturing output. Our rigorous QC labs and stringent purity specifications guarantee that every batch of levetiracetam meets or exceeds international pharmacopeial standards for enantiomeric excess and related substances. We understand the critical nature of antiepileptic medications and are committed to providing a stable, high-quality supply that supports your patient populations globally. Our technical team is well-versed in the nuances of this specific synthesis route, allowing us to troubleshoot and optimize production continuously for maximum efficiency.
We invite you to engage with our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. By initiating a dialogue now, you can secure a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this optimized supply source. Let us demonstrate how our manufacturing capabilities can enhance your supply chain resilience and product quality simultaneously. Contact us today to discuss your upcoming project needs and establish a long-term partnership built on technical excellence and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
