Advanced Industrial Synthesis of Deoxycholic Acid: A Cost-Effective Route for Pharmaceutical Manufacturing
Introduction to Novel Deoxycholic Acid Manufacturing
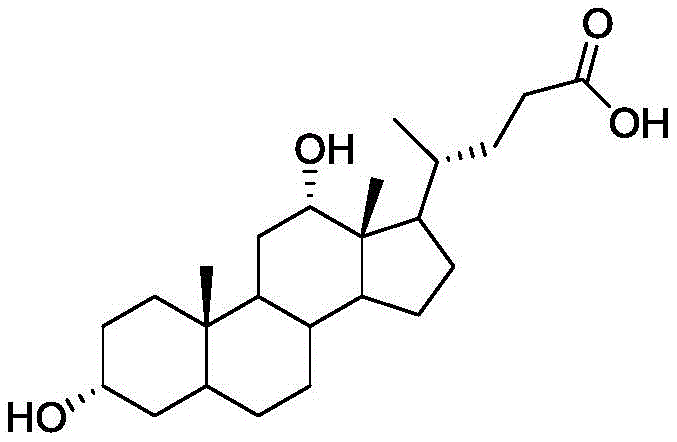
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of high-value active pharmaceutical ingredients (APIs) and their intermediates. Patent CN113024624A, published in June 2021, introduces a transformative synthetic method for deoxycholic acid, a critical compound widely recognized as the first FDA-approved injectable agent for local fat dissolution. This innovation addresses the longstanding challenges associated with steroid modification, specifically the removal of the 7-hydroxyl group from the cholic acid backbone. By leveraging a strategic sequence of esterification, selective oxidation, and elimination reactions, this patent outlines a process that is not only chemically elegant but also industrially pragmatic. The methodology shifts away from complex, low-yielding routes towards a streamlined approach that prioritizes operational simplicity and high total yield, making it an attractive candidate for commercial adoption by forward-thinking chemical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deoxycholic acid has been plagued by inefficiencies that hinder large-scale commercialization. As highlighted in the background of the patent, previous methodologies, such as the one disclosed in WO 2008157635, relied on excessively long reaction sequences comprising up to 11 distinct steps. A major bottleneck in these traditional routes was the dependence on precious metal catalysts, specifically platinum oxide (PtO), which drastically inflates raw material costs and introduces complexities in catalyst recovery and metal residue control. Furthermore, multi-step syntheses inherently accumulate yield losses at each stage, leading to suboptimal overall productivity and generating significant volumes of chemical waste. The purification of intermediates in these older processes often required rigorous chromatographic techniques, which are difficult to translate from laboratory benchtops to multi-ton industrial reactors, thereby creating a barrier to entry for mass production.
The Novel Approach
In stark contrast, the synthetic route detailed in CN113024624A offers a concise and efficient alternative that fundamentally restructures the manufacturing logic. The new process condenses the synthesis into just eight manageable steps, initiating from the abundant and renewable feedstock, cholic acid. By replacing expensive noble metal catalysts with common, cost-effective reagents such as N-bromosuccinimide (NBS), zinc powder, and palladium on carbon (Pd/C), the method achieves a dramatic reduction in direct material costs. The reaction conditions are notably mild, typically operating between 20°C and 70°C, which minimizes energy consumption and enhances operational safety. This streamlined approach not only accelerates the production cycle but also simplifies downstream processing, allowing for easier isolation of high-purity intermediates through standard crystallization techniques rather than complex separations.
Mechanistic Insights into Selective Deoxygenation and Steroid Modification
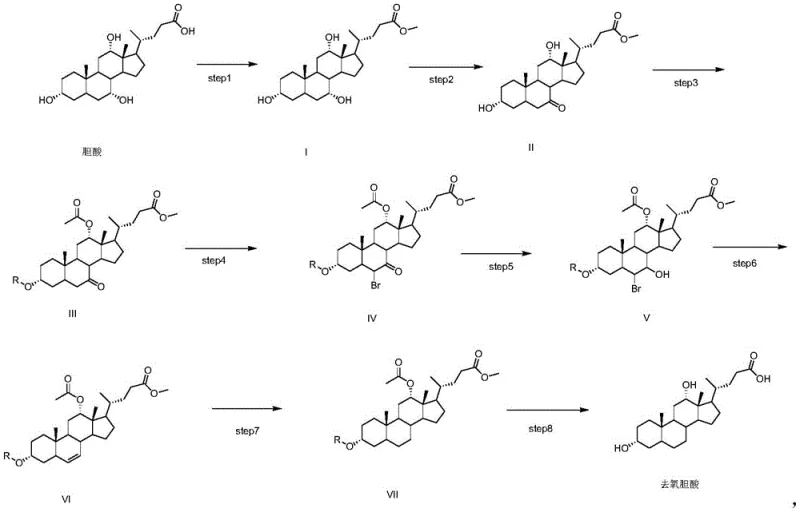
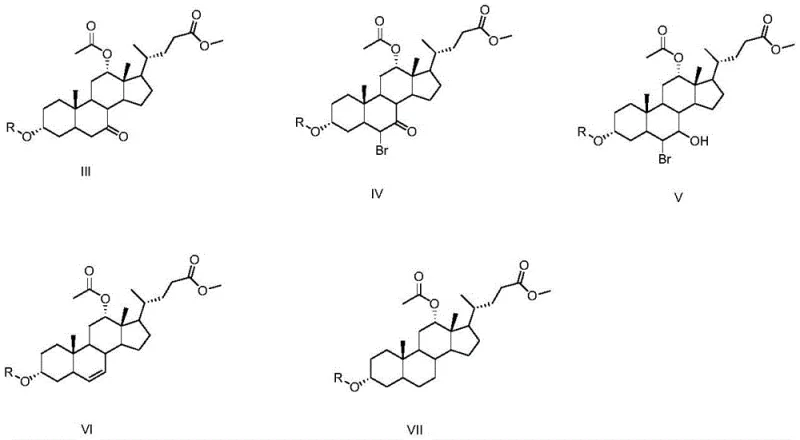
The core chemical innovation of this patent lies in its sophisticated handling of the steroid nucleus, particularly the selective manipulation of the hydroxyl groups at positions 3, 7, and 12. The process begins with the esterification of cholic acid to protect the carboxylic acid tail, followed by a crucial oxidation step using NBS in acetone. This step selectively targets the 7-hydroxyl group, converting it into a ketone functionality while leaving the 3 and 12-hydroxyl groups intact or subsequently protected. This selectivity is paramount, as it sets the stage for the subsequent deoxygenation. Following protection of the remaining hydroxyls with benzoyl or tosyl groups, the 7-keto intermediate undergoes alpha-bromination. This introduction of a bromine atom adjacent to the carbonyl group activates the molecule for a subsequent elimination reaction.
The removal of the oxygen functionality is achieved through a clever reduction-elimination sequence. Treatment with zinc powder in acetic acid facilitates the reductive elimination of the bromohydrin or bromoketone species, generating a double bond within the steroid ring system (specifically at the 5-6 position). This alkene intermediate is then subjected to catalytic hydrogenation using Pd/C, which saturates the double bond and effectively removes the oxygen atom originally present at the C-7 position, restoring the saturated steroid skeleton characteristic of deoxycholic acid. Finally, basic hydrolysis cleaves the ester and protecting groups to reveal the free hydroxyls and carboxylic acid. This mechanistic pathway ensures high stereochemical fidelity, minimizing the formation of epimers or regioisomers that could complicate purification and compromise the biological efficacy of the final API.
How to Synthesize Deoxycholic Acid Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity at every stage. The protocol initiates with the acid-catalyzed esterification of cholic acid in methanol, followed by the critical NBS oxidation which must be monitored closely to prevent over-oxidation. Subsequent protection steps utilize standard acylation chemistry, while the bromination and elimination steps require careful temperature management to ensure the correct regioselectivity of the double bond formation. The final hydrogenation and hydrolysis steps are robust but demand high-quality catalysts and controlled pH environments to prevent degradation of the sensitive steroid core. For process chemists looking to implement this route, the detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and compliance with Good Manufacturing Practices (GMP).
- Esterify cholic acid with methanol and sulfuric acid to form the methyl ester intermediate (Compound I).
- Perform selective oxidation using N-bromosuccinimide (NBS) in acetone to generate the 7-keto derivative (Compound II).
- Protect hydroxyl groups using benzoyl chloride and acetic anhydride, followed by bromination, reduction, elimination to alkene, catalytic hydrogenation, and final hydrolysis to yield pure deoxycholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this patented methodology offers compelling advantages that directly impact the bottom line and operational resilience. By transitioning from a precious metal-dependent process to one utilizing base metals and organic oxidants, manufacturers can insulate themselves from the volatility of platinum group metal prices. The reduction in step count not only lowers labor and utility costs but also shortens the overall manufacturing lead time, allowing for faster response to market demand fluctuations. Furthermore, the use of cholic acid as a starting material leverages a supply chain rooted in natural extraction, which is generally more stable and scalable than fully synthetic petrochemical routes. The simplified purification protocols reduce the reliance on expensive chromatography resins and solvents, contributing to a greener and more cost-efficient production profile.
- Cost Reduction in Manufacturing: The elimination of platinum oxide (PtO) from the catalyst list represents a significant financial benefit, as noble metals constitute a major portion of variable costs in fine chemical synthesis. Additionally, the use of inexpensive reagents like zinc powder and NBS, combined with the high yields reported in the patent examples, drives down the cost of goods sold (COGS). The ability to isolate intermediates via simple crystallization rather than column chromatography further reduces solvent consumption and waste disposal costs, creating a leaner manufacturing operation that is highly competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Utilizing cholic acid as the primary feedstock ensures a robust supply chain, as it is a naturally abundant bile acid with established global production capacity. Unlike specialized synthetic precursors that may have single-source suppliers, cholic acid is commoditized, reducing the risk of supply disruptions. The mild reaction conditions (mostly below 70°C) mean that the process can be run in standard glass-lined or stainless steel reactors without requiring exotic high-pressure or cryogenic equipment, thereby increasing the number of qualified contract manufacturing organizations (CMOs) capable of producing this intermediate.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, avoiding hazardous reagents or extreme conditions that pose risks during scale-up. The reduction in reaction steps inherently lowers the E-factor (mass of waste per mass of product), aligning with modern green chemistry principles and regulatory expectations for environmental sustainability. The straightforward workup procedures, involving aqueous washes and crystallizations, facilitate easier wastewater treatment and solvent recovery, ensuring that the manufacturing process remains compliant with increasingly stringent environmental regulations across different jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN113024624A, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new method for their own production lines or for procurement teams assessing the quality and consistency of the supply.
Q: What are the primary advantages of this new synthesis route over prior art?
A: The new method described in CN113024624A significantly reduces the number of reaction steps from 11 to 8 and eliminates the need for expensive platinum oxide (PtO) catalysts, resulting in lower production costs and easier purification.
Q: Is the starting material cholic acid readily available for large-scale production?
A: Yes, cholic acid is a naturally occurring bile acid that is abundantly available and cost-effective, ensuring a stable and reliable supply chain for the continuous manufacturing of deoxycholic acid.
Q: How does this process ensure high stereochemical purity?
A: The route utilizes specific protection and elimination strategies, including zinc-mediated elimination and palladium-catalyzed hydrogenation, which effectively control the stereochemistry at the C-7 position to match the natural configuration of deoxycholic acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our team of expert process chemists has thoroughly analyzed the technology described in CN113024624A and is well-positioned to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch of deoxycholic acid meets stringent purity specifications, adhering to the highest international standards for pharmaceutical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain efficiency and reduce overall project costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality chemical solutions that drive your business forward.
