Advanced Three-Step Synthesis of Boseine: Technical Breakthroughs for Commercial Scale-Up
Advanced Three-Step Synthesis of Boseine: Technical Breakthroughs for Commercial Scale-Up
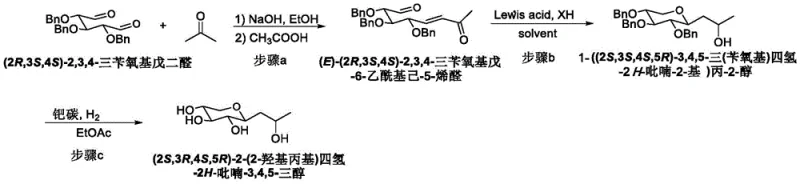
The global demand for high-performance anti-aging ingredients has placed Boseine, chemically known as (2S,3R,4S,5R)-2-(2-hydroxypropyl)tetrahydro-2H-pyran-3,4,5-triol, at the forefront of cosmetic chemistry innovation. Patent CN112812087A introduces a robust and highly efficient synthetic methodology that addresses critical bottlenecks in the current manufacturing landscape. This proprietary route leverages a chiral pool strategy starting from (2R,3S,4S)-2,3,4-tribenzyloxyglutaraldehyde, proceeding through a novel Lewis acid-catalyzed tandem reaction to construct the core tetrahydropyran scaffold. The process is distinguished by its exceptional total yield of approximately 78 percent across three distinct chemical transformations, offering a compelling alternative to legacy production methods. By integrating mild reaction conditions with precise stereochemical control, this technology enables the production of Boseine with superior purity profiles essential for premium skincare formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Boseine has been plagued by significant downstream processing challenges that compromise both economic efficiency and product quality. Traditional routes, such as those disclosed in earlier literature, often rely on the direct condensation of xylose with acetylacetone in alkaline aqueous solutions. While conceptually simple, these methods invariably produce Boseine as a dilute aqueous solution, making the isolation of the pure active ingredient extremely difficult and energy-intensive due to the high boiling point of water and the thermal sensitivity of the sugar derivative. Furthermore, alternative approaches utilizing homogeneous rare earth metal catalysts, while improving yields, introduce the risk of toxic metal residues that require costly and complex purification steps to meet stringent cosmetic safety regulations. Enzymatic methods, though green, frequently suffer from low substrate tolerance and difficult product recovery, limiting their viability for large-scale commercial supply chains.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112812087A employs a sophisticated organic synthesis strategy that prioritizes isolability and purity at every stage. The process initiates with a highly selective aldol condensation between a protected glutaraldehyde derivative and acetone, generating a key enone intermediate with excellent geometric control. The cornerstone of this innovation is the subsequent Lewis acid-catalyzed cascade reaction, which simultaneously effects carbon-oxygen bond formation and stereocenter establishment in a single operational step. This tandem transformation not only streamlines the synthetic sequence but also ensures that the intermediate remains in an organic phase, facilitating straightforward purification via crystallization or chromatography. The final deprotection step utilizes standard catalytic hydrogenation to cleanly remove benzyl groups, yielding the target molecule as a concentrated syrup rather than a dilute solution, thereby drastically reducing solvent removal costs and enhancing overall process throughput.
Mechanistic Insights into Lewis Acid-Catalyzed Tandem Cyclization
The heart of this synthetic breakthrough lies in the intricate mechanism of Step B, where a Lewis acid, such as boron trifluoride diethyl etherate, activates the electrophilic centers of the enone intermediate. Upon coordination of the Lewis acid to the carbonyl oxygen, the electron density of the conjugated system is significantly altered, rendering the beta-carbon highly susceptible to nucleophilic attack by the silane reagent. This initial hydride delivery triggers a cascade of electronic rearrangements that facilitate an intramolecular 1,4-Michael addition, effectively closing the six-membered tetrahydropyran ring with high diastereoselectivity. The chirality of the starting material, derived from the specific configuration of the tribenzyloxyglutaraldehyde, acts as a powerful directing group, ensuring that the newly formed stereocenters align perfectly with the natural configuration required for biological activity. This mechanistic precision minimizes the formation of diastereomeric impurities, which are notoriously difficult to separate in carbohydrate chemistry, thus securing a high-purity profile for the protected intermediate before the final deprotection even begins.
From an impurity control perspective, the choice of reagents and conditions in this tandem reaction is critical for maintaining a clean reaction profile. The use of triethylsilane or trimethylsilane as the hydride source provides a controlled reduction potential that avoids over-reduction of other sensitive functional groups within the molecule. Furthermore, the reaction is conducted at low temperatures initially, typically around minus 78 degrees Celsius, to manage the exothermicity and prevent side reactions such as polymerization or non-selective reduction. As the reaction warms to room temperature, the kinetic barrier for the cyclization is overcome, driving the equilibrium towards the thermodynamically stable pyran product. The subsequent quenching with saturated sodium bicarbonate effectively neutralizes the Lewis acid and any silyl byproducts, allowing for a clean phase separation that removes inorganic salts and catalyst residues, thereby simplifying the workup procedure and reducing the burden on downstream purification units.
How to Synthesize Boseine Efficiently
The synthesis of Boseine via this patented route represents a paradigm shift in how high-value cosmetic actives are manufactured, balancing chemical elegance with industrial practicality. The protocol is designed to be scalable, utilizing readily available solvents like ethanol and dichloromethane, and common reagents such as acetone and sodium hydroxide, which ensures a stable and cost-effective supply chain. The detailed standardized synthetic steps provided below outline the precise molar ratios, temperature controls, and workup procedures necessary to replicate the high yields reported in the patent data. Adhering to these parameters is essential for maximizing the efficiency of the tandem cyclization and ensuring the final product meets the rigorous quality standards expected by global cosmetic brands.
- Perform aldol condensation between (2R,3S,4S)-2,3,4-tribenzyloxyglutaraldehyde and acetone using alkali in ethanol.
- Execute Lewis acid-catalyzed tandem nucleophilic addition and 1,4-Michael addition with silane to form the protected pyran ring.
- Conduct catalytic hydrogenation using Pd/C or Pd(OH)2/C to remove benzyl protecting groups and yield pure Boseine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits that extend far beyond simple yield improvements. The elimination of aqueous reaction media in the critical bond-forming steps significantly reduces the energy consumption associated with solvent recovery and product drying, leading to a lower carbon footprint and reduced utility costs per kilogram of output. Moreover, the avoidance of rare earth metals and complex enzymatic systems simplifies the raw material sourcing strategy, mitigating the risks associated with geopolitical supply constraints or price volatility of specialized catalysts. The ability to isolate intermediates as stable solids or oils rather than unstable solutions enhances inventory management capabilities, allowing for flexible production scheduling and the potential for campaign manufacturing to meet fluctuating market demands without compromising product integrity.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and the use of commodity chemicals as starting materials. By consolidating multiple bond-forming events into a single tandem reaction step, the process drastically reduces the number of unit operations required, which translates directly into lower labor costs and reduced equipment occupancy time. The high yield of the final hydrogenation step ensures that the expensive chiral starting material is converted into the final product with minimal loss, maximizing the return on investment for every batch produced. Additionally, the simplified purification requirements mean less consumption of chromatography media and filtration aids, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of the reaction conditions, which tolerate minor variations in temperature and reagent quality without catastrophic failure. The reliance on standard hydrogenation technology and common Lewis acids means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations without the need for specialized proprietary equipment. This flexibility ensures continuity of supply even in the face of regional disruptions, as the technical know-how is based on fundamental organic synthesis principles widely understood in the industry. Furthermore, the stability of the benzyl-protected intermediate allows for the decoupling of the synthesis stages, enabling the stockpiling of key precursors to buffer against raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is facilitated by the absence of hazardous reagents and the mild operating pressures used in the final step. The hydrogenation is performed at low pressure, typically around 12 psi, which reduces the safety engineering requirements and insurance costs associated with high-pressure reactors. From an environmental standpoint, the process generates less aqueous waste compared to traditional methods, as the majority of the byproducts are organic and can be incinerated for energy recovery or recycled. The use of palladium catalysts, which can be recovered and reused, aligns with green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations regarding heavy metal discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Boseine synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN112812087A, providing a factual basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating process transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: How does this synthesis method improve product purity compared to traditional xylose routes?
A: Unlike traditional methods that often result in aqueous solutions difficult to purify, this patent utilizes a benzyl-protected intermediate strategy. The final hydrogenation step yields a solid syrup of high-purity Boseine, eliminating the complex separation issues associated with water-soluble byproducts found in direct xylose condensation.
Q: What are the specific advantages of the Lewis acid tandem reaction in Step B?
A: The use of Lewis acids like boron trifluoride etherate facilitates a highly efficient tandem reaction combining nucleophilic addition and 1,4-Michael addition. This one-pot transformation constructs the tetrahydropyran core with excellent stereocontrol and high yield (up to 87%), significantly reducing processing time compared to multi-step protection-deprotection sequences.
Q: Is the hydrogenation step safe for large-scale manufacturing?
A: Yes, the process is designed for safety and scalability. The hydrogenation operates at mild pressures (as low as 12 psi) and room temperature using standard Pd/C or Pd(OH)2/C catalysts. These conditions minimize safety risks associated with high-pressure hydrogenation while ensuring complete removal of benzyl groups without over-reduction of the sugar backbone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boseine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and reliable supply in the fast-paced cosmetic ingredients market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in this patent can be seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch of Boseine we produce. Our commitment to excellence means that we do not just supply a chemical; we deliver a validated solution that meets the exacting standards of global personal care brands.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability rather than marketing promises.
