Scalable Water-Based Synthesis of High-Purity Allyl Glycosides for Commercial Production
Introduction to Advanced Allyl Glycoside Manufacturing
The landscape of carbohydrate chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes that align with modern industrial sustainability goals. Patent CN115043890A introduces a groundbreaking methodology for the synthesis of allyl glycosides, specifically targeting the production of high-purity allyl pyranosides with exceptional beta-isomer selectivity. This innovation addresses long-standing challenges in the field by utilizing water as the sole reaction solvent, thereby eliminating the reliance on hazardous organic solvents and complex protection-deprotection sequences that have traditionally plagued this sector. For R&D directors and process chemists, this represents a pivotal shift towards atom-economic chemistry that simplifies workflow while maintaining rigorous quality standards. The technology enables the direct alkylation of unprotected pyranoses, such as glucose, galactose, and mannose, under mild conditions ranging from 15°C to 35°C, ensuring thermal stability and minimizing degradation pathways. By integrating this novel approach, manufacturers can achieve a reliable pharmaceutical intermediates supplier status, offering clients a product line that is both environmentally compliant and economically superior.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allyl glycosides has been constrained by methodologies that are either chemically inefficient or environmentally burdensome, creating significant bottlenecks for large-scale production. Traditional routes such as the Fischer glycosylation often suffer from poor stereoselectivity, predominantly yielding alpha-anomers due to the anomeric effect, which necessitates difficult and costly separation processes to isolate the desired beta-isomers. Alternatively, the Koenigs-Knorr reaction, while capable of producing trans-glycosides, relies heavily on stoichiometric amounts of toxic heavy metal salts like silver carbonate or mercury oxide, posing severe safety risks and generating hazardous waste streams that complicate regulatory compliance. Furthermore, the Helferich glycosylation requires fully acetylated sugar precursors and strong Lewis acids, introducing additional synthetic steps for acetylation and subsequent deacetylation that drastically reduce overall yield and increase material costs. These conventional methods also frequently employ high-boiling polar organic solvents to solubilize unprotected sugars, which demand energy-intensive distillation processes for removal, thereby inflating the carbon footprint and operational expenditure of the manufacturing facility.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN115043890A leverages a unique aqueous-phase alkylation strategy that bypasses the need for protecting groups and toxic catalysts entirely. This innovative protocol utilizes water as a green solvent, capitalizing on the differential solubility of reactants and products to drive the reaction forward without the aid of phase transfer catalysts, which were previously thought to be essential for such biphasic systems. By meticulously optimizing the molar ratios of pyranose to allyl bromide (ranging from 1:1.2 to 1:9) and the alkaline reagent (1:1.05 to 1:2), the process achieves a kinetic enhancement that overcomes the inherent solubility limitations of allyl bromide in water. This precise control allows for the direct conversion of various pyranose substrates into their corresponding allyl glycosides with remarkable beta-selectivity, consistently exceeding a beta-to-alpha ratio of 85:15.  The versatility of this approach is demonstrated by its applicability to a wide range of monosaccharides and disaccharides, including D-glucose, D-mannose, D-galactose, cellobiose, lactose, and maltobiose, making it a robust platform technology for cost reduction in pharmaceutical intermediates manufacturing.
The versatility of this approach is demonstrated by its applicability to a wide range of monosaccharides and disaccharides, including D-glucose, D-mannose, D-galactose, cellobiose, lactose, and maltobiose, making it a robust platform technology for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Aqueous Nucleophilic Substitution
The core of this technological breakthrough lies in the nuanced understanding of reaction kinetics within an aqueous medium, where the interplay between reagent concentration and solubility dictates the stereochemical outcome. Unlike traditional organic phase reactions where solvation effects stabilize specific transition states, this aqueous system relies on the localized concentration of hydroxide ions to deprotonate the anomeric hydroxyl group of the pyranose, generating a highly reactive alkoxide species in situ. The absence of a phase transfer catalyst is counterintuitive yet effective; the limited solubility of allyl bromide in water creates a micro-environment where the reaction occurs at the interface, preventing excessive side reactions such as poly-allylation of other hydroxyl groups on the sugar ring. This kinetic control is further refined by maintaining the reaction temperature between 15°C and 35°C, a range that is sufficiently warm to promote nucleophilic attack but cool enough to suppress hydrolysis of the allyl bromide or degradation of the sensitive glycosidic bond. The result is a clean transformation that favors the formation of the thermodynamically stable beta-anomer, likely due to the specific orientation of the intermediate and the steric constraints imposed by the aqueous solvation shell surrounding the sugar molecule.
Furthermore, the mechanism ensures exceptional impurity control by minimizing the formation of regioisomers and poly-substituted by-products that typically complicate downstream purification. The selective activation of the anomeric position is achieved through the careful balancing of the alkaline reagent, ensuring that only the most acidic proton at the C1 position is abstracted preferentially under the controlled conditions. This specificity eliminates the need for chromatographic separation of complex mixtures, allowing for simpler workup procedures such as extraction and recrystallization to yield products of high purity. 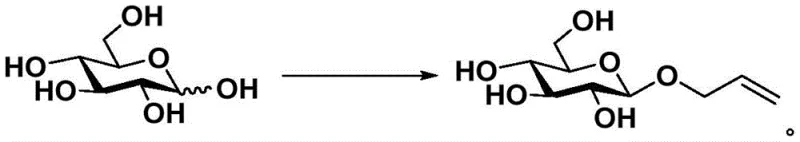 The structural integrity of the product is confirmed through rigorous NMR characterization, which validates the retention of the pyranose ring structure and the successful installation of the allyl group at the anomeric center. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the reaction parameters are robust and reproducible across different batches of raw materials, thereby supporting the commercial scale-up of complex pharmaceutical intermediates.
The structural integrity of the product is confirmed through rigorous NMR characterization, which validates the retention of the pyranose ring structure and the successful installation of the allyl group at the anomeric center. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the reaction parameters are robust and reproducible across different batches of raw materials, thereby supporting the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Allyl Pyranoside Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and stereoselectivity. The process begins with the preparation of an aqueous reaction mixture where the concentration of the pyranose substrate is maintained between 0.1 mol/L and 0.3 mol/L to ensure adequate solubility and interaction with the reagents. Operators must carefully monitor the addition of the alkaline reagent, such as sodium hydroxide or lithium hydroxide, to maintain the pH balance necessary for deprotonation without causing sugar degradation. The reaction time is flexible, ranging from 4 to 24 hours depending on the specific sugar substrate and temperature, allowing for process optimization based on real-time monitoring. Detailed standardized synthesis steps see the guide below.
- Mix pyranose, allyl bromide, and an alkaline reagent in water with specific molar ratios.
- Maintain the reaction temperature between 15°C and 35°C for 4 to 24 hours.
- Extract the reaction mixture, concentrate under reduced pressure, and purify via recrystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology offers substantial benefits that directly impact the bottom line and supply chain resilience for global chemical buyers. The elimination of acetylation and deacetylation steps significantly shortens the synthetic route, reducing the total number of unit operations and the associated labor and equipment costs required for production. By removing the dependency on expensive heavy metal catalysts like silver and mercury, the process not only lowers raw material expenses but also mitigates the regulatory burden associated with the disposal of toxic metal waste, leading to substantial cost savings in waste management and environmental compliance. The use of water as a solvent further enhances the economic profile by replacing costly organic solvents and reducing the energy consumption linked to solvent recovery and distillation, contributing to a lower overall cost of goods sold. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, improving the overall material efficiency and ensuring a more predictable supply of high-quality intermediates.
- Cost Reduction in Manufacturing: The streamlined process eliminates multiple protection and deprotection stages, which traditionally account for a significant portion of manufacturing time and resource allocation. By avoiding the use of precious metal catalysts and volatile organic compounds, the operational expenditure is drastically reduced, allowing for more competitive pricing structures in the global market. The simplified workup procedure, involving basic extraction and crystallization, reduces the need for specialized purification equipment, further lowering capital investment requirements for production facilities. These cumulative efficiencies translate into a leaner manufacturing model that is highly responsive to market demands while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as water, sodium hydroxide, and readily available allyl bromide ensures a stable and secure supply chain that is less susceptible to geopolitical disruptions or raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and times, provides operational flexibility that can accommodate variations in production schedules without compromising product quality. This reliability is critical for long-term supply agreements, as it guarantees consistent delivery timelines and reduces the risk of production delays that could impact downstream drug development programs. Furthermore, the scalability of the aqueous process facilitates seamless technology transfer from laboratory to pilot and commercial scales, ensuring continuity of supply as demand grows.
- Scalability and Environmental Compliance: Adopting this green chemistry approach aligns perfectly with increasingly stringent environmental regulations, positioning the manufacturer as a leader in sustainable chemical production. The reduction in hazardous waste generation and the use of non-toxic solvents simplify the permitting process for new production lines and reduce the liability associated with environmental incidents. The process is inherently scalable, as the heat transfer properties of water allow for safe operation in large reactors, enabling the production of multi-ton quantities without the safety risks associated with flammable organic solvents. This combination of scalability and compliance makes the technology an ideal choice for companies seeking to expand their capacity while adhering to corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and capabilities of this synthesis method, providing clarity for potential partners and technical evaluators. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing manufacturing workflows or for evaluating the quality of the supplied intermediates. We encourage technical teams to review these points to gain a comprehensive understanding of the process advantages.
Q: Why is water used as the solvent instead of organic solvents?
A: Water is used to eliminate the need for high-boiling organic solvents, reducing energy consumption during removal and avoiding toxic waste, while specific reagent ratios kinetically enhance the reaction rate despite solubility challenges.
Q: How is high beta-selectivity achieved without protecting groups?
A: By precisely controlling the molar ratios of pyranose to allyl bromide and the alkaline reagent, the reaction kinetics favor the formation of the beta-isomer, achieving a ratio greater than 85:15 without acetylation steps.
Q: What are the purification methods for the crude product?
A: The crude product can be purified effectively through recrystallization using alcohols like methanol or ethanol, or via column chromatography using ethyl acetate and methanol mixtures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Glycoside Supplier
At NINGBO INNO PHARMCHEM, we are committed to translating cutting-edge academic research into commercially viable solutions that empower our clients to innovate faster and more efficiently. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and risk-free. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of allyl glycoside meets the highest industry standards for pharmaceutical applications. Our dedication to quality and consistency makes us a trusted partner for global enterprises seeking to secure their supply chains with high-performance chemical intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our greener, more efficient production method. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process, ensuring that you have all the necessary information to move forward with confidence. Contact us today to explore a partnership that combines technical excellence with commercial value.
