Scalable Water-Based Synthesis of High-Purity Allyl Glycosides for Pharma Intermediates
The landscape of carbohydrate chemistry is undergoing a significant transformation driven by the demand for greener, more efficient synthetic routes for critical intermediates. Patent CN115043890A introduces a groundbreaking methodology for the synthesis of allyl glycosides, specifically targeting the production of high-purity beta-allyl pyranosides. This technology addresses long-standing challenges in the field by utilizing water as the sole reaction solvent, thereby eliminating the need for hazardous organic solvents and complex protection-deprotection sequences. For R&D directors and process chemists, this represents a pivotal shift towards atom-economical processes that maintain rigorous stereochemical control. The ability to generate allyl glycosides with a beta-to-alpha isomer ratio exceeding 85:15 under mild conditions opens new avenues for the rapid assembly of glycoconjugates and oligosaccharides, which are essential for modern drug discovery and development programs focusing on glycobiology.
Allyl glycosides serve as versatile building blocks in organic synthesis due to the reactive double bond present in the allyl group, which can undergo diverse transformations such as epoxidation, dihydroxylation, and click chemistry reactions. Traditionally, accessing these compounds required harsh conditions or multi-step protocols that compromised overall yield and purity. The disclosed invention leverages a direct nucleophilic substitution mechanism at the anomeric position of unprotected pyranoses. By optimizing the molar ratios of reactants and reaction time, the process achieves high conversion rates without the aid of phase transfer catalysts, which are often difficult to remove and can contaminate the final product. This technical advancement not only streamlines the workflow but also aligns with the increasing regulatory pressure on pharmaceutical manufacturers to adopt sustainable manufacturing practices that minimize environmental impact while maximizing output quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allyl glycosides has relied on three primary methodologies, each fraught with significant operational and economic drawbacks that hinder large-scale production. The Fischer glycosylation reaction, while simple, typically requires acid catalysis and alcohol solvents, often resulting in a mixture of pyranosides, furanosides, and ring-opened products with poor stereocontrol, predominantly favoring the alpha-anomer due to the anomeric effect. Alternatively, the Koenigs-Knorr reaction utilizes halogenated sugars activated by heavy metal salts such as silver carbonate or mercury oxide; while effective for stereocontrol, this method generates toxic heavy metal waste and requires expensive protected sugar starting materials, drastically increasing the cost of goods. Furthermore, the Helferich glycosylation employs Lewis acids like boron trifluoride etherate with peracetylated sugars, which limits substrate scope to simple structures and necessitates additional steps for acetylation and subsequent deacetylation. These traditional routes are not only labor-intensive but also involve the use of high-boiling point organic solvents that are energy-intensive to remove, creating a bottleneck in process efficiency and supply chain sustainability for fine chemical manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN115043890A offers a streamlined, single-pot solution that bypasses the need for sugar protection entirely. By conducting the reaction in an aqueous medium at mild temperatures ranging from 15°C to 35°C, the process significantly reduces energy consumption and operational hazards associated with volatile organic compounds. The key innovation lies in the precise control of reactant stoichiometry, where the molar ratio of pyranose to allyl bromide is maintained between 1:1.2 and 1:9, and the base is carefully dosed to promote selective alkylation at the anomeric hydroxyl group. This approach effectively suppresses poly-allylation side reactions, ensuring that the primary product is the desired mono-allylated glycoside. The elimination of acetylation and deacetylation steps shortens the synthetic timeline considerably, allowing for faster turnaround times from raw material to finished intermediate. Moreover, the use of water as a solvent simplifies the workup procedure, as inorganic salts and by-products remain in the aqueous phase while the product can be efficiently extracted, leading to a cleaner crude profile and reduced purification burdens downstream.
Mechanistic Insights into Aqueous Phase Alkylation
The mechanistic pathway of this water-based alkylation is distinct from traditional organic phase reactions, relying on kinetic control to achieve high stereoselectivity without neighboring group participation from C-2 esters. In the absence of a phase transfer catalyst, the reaction rate is inherently modulated by the poor solubility of allyl bromide in water, which creates a biphasic system that prevents runaway exotherms and uncontrolled side reactions. The alkaline reagent, typically sodium hydroxide or lithium hydroxide, deprotonates the anomeric hydroxyl group of the pyranose to generate a reactive alkoxide species in situ. This nucleophile then attacks the allyl bromide via an SN2 mechanism. Interestingly, the aqueous environment appears to stabilize the transition state leading to the beta-isomer, possibly through specific solvation effects or hydrogen bonding networks that are not present in organic solvents. This results in a product distribution where the beta-anomer is dominant, with ratios consistently observed above 85 percent, a level of selectivity that is difficult to achieve with unprotected sugars in non-aqueous media without extensive protecting group manipulation.
Furthermore, the optimization of reaction parameters plays a critical role in suppressing the formation of poly-hydroxy allylated by-products. By maintaining the pyranose concentration between 0.1 mol/L and 0.3 mol/L, the system ensures sufficient intermolecular contact while minimizing intermolecular aggregation that could lead to oligomerization. The reaction time, typically ranging from 4 to 24 hours depending on the specific sugar substrate, allows for complete conversion of the starting material while preventing the degradation of the sensitive allyl group under prolonged basic conditions. For instance, in the synthesis of allyl glucoside, a reaction time of 8 hours at 25°C was found to be optimal, balancing yield and selectivity. This fine-tuning of kinetic variables demonstrates a deep understanding of the reaction dynamics, enabling the production of high-purity intermediates suitable for sensitive downstream applications such as the synthesis of complex glycoconjugates or active pharmaceutical ingredients where impurity profiles are strictly regulated.
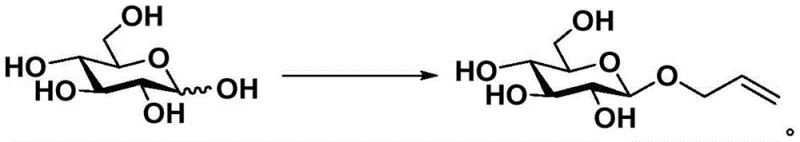
How to Synthesize Allyl Glycoside Efficiently
The implementation of this synthesis protocol requires careful attention to the addition order and mixing efficiency to maximize the interfacial area between the aqueous and organic phases. The process begins with the dissolution of the pyranose substrate in water, followed by the addition of the alkaline reagent to generate the nucleophilic species before introducing the allyl bromide. Detailed standardized operating procedures regarding temperature ramping, stirring speeds, and quenching protocols are essential for reproducibility on a kilogram scale. For process engineers and laboratory technicians looking to replicate these results, the following guide outlines the critical unit operations involved in transforming raw sugars into valuable allyl glycoside intermediates with minimal waste generation.
- Mix pyranose, allyl bromide, and an alkaline reagent (NaOH or LiOH) in water with a molar ratio of pyranose to allyl bromide between 1: 1.2 and 1:9.
- Maintain the reaction temperature between 15°C and 35°C for a duration of 4 to 24 hours to ensure high beta-selectivity.
- Perform extraction with ethyl acetate or dichloromethane, followed by vacuum concentration and purification via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this water-based synthesis technology offers substantial strategic advantages that directly impact the bottom line and operational resilience. The most significant benefit is the drastic reduction in raw material costs associated with protecting group chemistry; by eliminating the need for acetic anhydride, pyridine, and the subsequent reagents required for deprotection, manufacturers can realize significant savings on bill of materials. Additionally, the replacement of expensive, high-boiling organic solvents like dimethylformamide or dimethyl sulfoxide with water removes the costly and energy-intensive step of high-vacuum distillation for solvent recovery, thereby lowering utility costs and reducing the carbon footprint of the manufacturing facility. This simplification of the process flow also translates to shorter production cycles, allowing suppliers to respond more rapidly to fluctuating market demands and reducing the inventory holding costs for both the manufacturer and the client.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps fundamentally alters the cost structure of allyl glycoside production. Traditional routes often involve four to six discrete chemical steps to install and remove protecting groups, each incurring labor, reagent, and waste disposal costs. By condensing this into a single direct alkylation step, the overall process mass intensity is significantly lowered. Furthermore, the avoidance of heavy metal catalysts such as silver or mercury salts removes the need for specialized waste treatment protocols and expensive metal scavenging resins, which are often required to meet stringent residual metal specifications in pharmaceutical intermediates. This streamlined approach ensures that the cost of goods sold remains competitive even as raw material prices fluctuate, providing a stable pricing model for long-term supply agreements.
- Enhanced Supply Chain Reliability: Utilizing water as the primary solvent mitigates risks associated with the supply volatility of petrochemical-derived organic solvents. Water is universally available, non-flammable, and exempt from many of the transportation regulations that govern hazardous chemicals, simplifying logistics and storage requirements. The robustness of the reaction conditions, which tolerate a range of temperatures between 15°C and 35°C, means that production is less susceptible to minor fluctuations in facility cooling or heating capacity, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply lines for critical drug substances, reducing the risk of production delays that could impact downstream clinical trials or commercial launches. The simplicity of the workup, involving standard extraction and crystallization techniques, also means that the process can be easily transferred between different manufacturing sites without extensive re-validation.
- Scalability and Environmental Compliance: The green chemistry credentials of this process facilitate easier regulatory approval and environmental compliance. By generating primarily aqueous waste streams containing inorganic salts, the burden on wastewater treatment facilities is markedly reduced compared to processes generating halogenated or aromatic organic waste. The absence of volatile organic compounds (VOCs) improves workplace safety and reduces the need for complex emission control systems. From a scalability standpoint, the exotherm of the reaction is naturally moderated by the high heat capacity of water, making it safer to scale from gram-scale laboratory experiments to multi-ton commercial production without the risk of thermal runaway. This inherent safety profile allows for larger batch sizes, improving equipment utilization rates and further driving down the unit cost of production for high-volume applications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on selectivity, substrate scope, and purification strategies. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does this water-based method improve beta-selectivity compared to traditional methods?
A: By strictly controlling the stoichiometric ratios of pyranose to allyl bromide (1:1.2~9) and alkaline reagent (1:1.05~2) without phase transfer catalysts, the reaction kinetics favor the formation of the beta-isomer, achieving a beta-to-alpha ratio greater than 85:15.
Q: What are the environmental benefits of using water as a solvent in this synthesis?
A: Using water eliminates the need for high-boiling point polar organic solvents like DMF or DMSO, significantly reducing energy consumption during solvent removal and avoiding the generation of toxic organic waste streams associated with traditional glycosylation protocols.
Q: Does this process require protection and deprotection steps for the hydroxyl groups?
A: No, this novel approach avoids the conventional acetylation and deacetylation steps entirely, simplifying the synthetic route and directly yielding the target allyl pyranoside from unprotected starting sugars.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Glycoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this water-based allylation technology for the next generation of glycoscience applications. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of anomeric ratios via NMR spectroscopy to guarantee the high beta-selectivity promised by this novel method. We are committed to delivering high-purity allyl glycosides that meet the exacting standards of the global pharmaceutical industry, supporting your R&D efforts with reliable, high-quality intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your project volume and purity requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to validate the performance of our allyl glycoside intermediates in your downstream processes. Let us collaborate to accelerate your development timelines and bring your novel glycoconjugate therapeutics to market faster.
