Advanced Manufacturing of High-Purity Phosphite Esters for Industrial Stabilization
Advanced Manufacturing of High-Purity Phosphite Esters for Industrial Stabilization
The chemical industry constantly seeks robust methodologies for synthesizing high-performance additives, particularly phosphite esters which serve as critical antioxidants and stabilizers in polymer matrices. Patent CN101684130A introduces a transformative preparation method that addresses longstanding challenges in the esterification of phosphorus halides with phenolic compounds. By utilizing dichloromethane as a primary reaction solvent, this innovation effectively mitigates the formation of high-viscosity intermediates that typically plague traditional synthesis routes. This technical breakthrough ensures that the resulting phosphite esters possess exceptionally low chloride ion content, a parameter vital for their application as ligands in sensitive transition metal catalysis. For R&D directors and procurement specialists, understanding this shift from conventional toluene-based systems to optimized dichloromethane protocols represents a significant opportunity for enhancing product reliability and supply chain efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for phosphite esters often rely on solvents like toluene in conjunction with tertiary amine acid binding agents, a combination that frequently leads to severe processing bottlenecks. As the reaction progresses, the precipitation of ammonium salts combined with the inherent properties of the organophosphorus intermediates causes a dramatic increase in mixture viscosity. This rheological change creates substantial difficulties in stirring and heat exchange, making temperature control precarious and potentially hazardous on a large scale. Furthermore, the presence of solid by-products necessitates complex filtration operations, which are not only time-consuming but also risk trapping valuable product within the filter cake. The removal of residual chloride ions from the final product is notoriously difficult in these systems, often requiring extensive washing that generates significant waste streams and lowers overall yield.
The Novel Approach
The methodology disclosed in the patent circumvents these issues by employing dichloromethane, a solvent that maintains the reaction intermediates in a true solution state throughout the process. This fundamental change in solvent physics prevents the sharp rise in viscosity, allowing for uniform mixing and efficient thermal management even during exothermic stages. The liquid-phase nature of the by-products facilitates a straightforward aqueous workup where the organic phase containing the product separates cleanly from the aqueous layer containing the salts. This simplification eliminates the need for filtering solid precipitates, thereby streamlining the purification workflow and reducing equipment downtime. Consequently, the final phosphite ester exhibits superior purity profiles with chloride levels consistently maintained below 0.01% wt, meeting the stringent requirements for high-end applications.
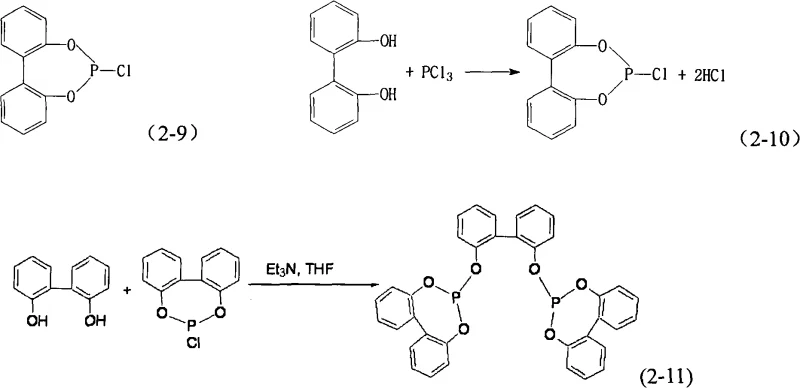
Mechanistic Insights into Dichloromethane-Mediated Esterification
The core of this synthetic advantage lies in the solvation dynamics of dichloromethane compared to aromatic solvents like toluene. In the initial stage, phosphorus trichloride reacts with a phenolic substrate to form a chloro phosphite ester intermediate, a step that is highly sensitive to steric hindrance and solvent polarity. When dichloromethane is used, the intermediate remains fully dissolved, preventing the localized concentration spikes that lead to oligomerization or decomposition. This homogeneous environment ensures that the subsequent nucleophilic attack by the second phenolic component proceeds with high regioselectivity and conversion rates. The mechanism avoids the formation of insoluble complexes that typically sequester the active phosphorus species, thereby maximizing the atom economy of the transformation.
Impurity control is another critical aspect governed by the choice of solvent and the specific workup protocol enabled by this system. The high solubility of the triethylamine hydrochloride salt in the aqueous phase during the quenching step allows for its near-quantitative removal from the organic layer. In contrast, conventional methods often leave trace amounts of chloride trapped within the viscous organic matrix, which can act as a poison for downstream catalytic applications such as hydroformylation. The ability to reduce chloride content to less than 0.01% wt through simple phase separation underscores the efficacy of this approach in producing electronic-grade or catalyst-grade intermediates. This level of purity is essential for preventing the deactivation of expensive rhodium or palladium catalysts in industrial processes.
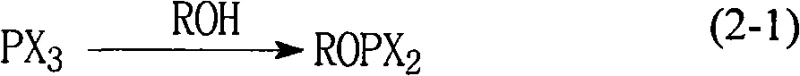
How to Synthesize High-Purity Phosphite Esters Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for generating complex bis-phosphite structures with minimal operational friction. The process begins with the dissolution of the chloro phosphite ester precursor in dichloromethane, followed by the separate preparation of the phenolic coupling partner in a triethylamine mixture. These two streams are combined under controlled thermal conditions, typically ranging from -40°C to 20°C, to manage the exotherm while ensuring complete conversion. The detailed standardized synthesis steps see the guide below for specific molar ratios and timing.
- Dissolve the chloro phosphite ester intermediate in dichloromethane to ensure a homogeneous solution state.
- Prepare a solution of the phenol or diol substrate in triethylamine or a triethylamine-dichloromethane mixture.
- Mix the solutions at controlled temperatures (-40 to 20°C), stir, and perform an aqueous workup to separate the organic phase containing the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this dichloromethane-based synthesis route translates directly into tangible operational efficiencies and cost optimizations. The elimination of solid filtration steps removes a major bottleneck in batch processing, allowing for faster turnover times and higher throughput in existing reactor infrastructure. By avoiding the use of excessive solid acid scavengers or ion-exchange resins, the process significantly reduces the volume of solid waste generated, lowering disposal costs and environmental compliance burdens. The simplified purification workflow also means less solvent is required for washing filter cakes, contributing to a reduction in overall raw material consumption. These factors combine to create a more resilient supply chain capable of delivering high-quality intermediates with greater consistency.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive filtration equipment and the associated labor costs of handling solid precipitates. By maintaining the reaction in a liquid phase, energy consumption for heating and stirring is optimized, leading to lower utility costs per kilogram of product. The high yield and purity reduce the need for reprocessing or secondary purification steps, further driving down the cost of goods sold. Additionally, the ability to use crude chloro phosphite intermediates without prior purification saves significant time and resources in the upstream synthesis stage.
- Enhanced Supply Chain Reliability: The robustness of the reaction against viscosity changes ensures that production schedules are met without unexpected delays caused by equipment fouling or mixing failures. The use of common, commercially available solvents like dichloromethane and triethylamine secures the supply of raw materials against market volatility. Consistent product quality with low chloride content reduces the risk of batch rejection by downstream customers, fostering stronger long-term partnerships. This reliability is crucial for maintaining continuous operations in industries where downtime can result in substantial financial losses.
- Scalability and Environmental Compliance: The absence of high-viscosity phases makes the technology inherently scalable from pilot plant to full commercial production without significant engineering modifications. The efficient phase separation minimizes the generation of wastewater contaminated with organic solids, simplifying effluent treatment processes. Reduced solvent usage for washing and purification aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. The process design supports the safe handling of reactive phosphorus species, enhancing overall plant safety profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel phosphite ester preparation method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. They serve to clarify the operational advantages and quality improvements achievable through this specific synthetic route.
Q: Why is dichloromethane preferred over toluene in this phosphite ester synthesis?
A: Dichloromethane provides superior solubility for the reaction intermediates, preventing the formation of high-viscosity suspensions that occur in toluene. This ensures efficient mixing and heat transfer, which is critical for maintaining reaction control and product quality.
Q: How does this method improve the purity of the final phosphite ester?
A: The process allows for the effective removal of chloride ions through a simple aqueous wash, achieving chloride content levels below 0.01% wt. Additionally, the absence of solid salt by-products eliminates the need for complex filtration steps that often trap impurities.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly scalable because it avoids the formation of viscous materials that complicate stirring and heat exchange in large reactors. The simplified workup procedure involving phase separation rather than solid filtration significantly enhances operational efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphite Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity phosphite esters play in advancing polymer stability and catalytic efficiency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We employ stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for sensitive applications. Our commitment to technical excellence allows us to deliver products that enhance the performance of your final formulations while minimizing supply chain risks.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this optimized process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to support your project goals. Let us partner with you to drive innovation and efficiency in your chemical manufacturing operations.
