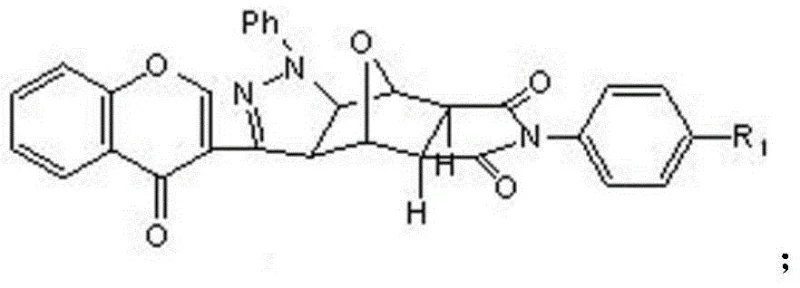
Advanced Synthesis and Commercial Scale-Up of Chromone-Containing Pyrazole Norcantharidin Derivatives
The pharmaceutical industry is constantly seeking novel molecular scaffolds that can offer improved therapeutic indices, particularly in the challenging field of oncology. Patent CN103554123A discloses a groundbreaking class of chromone structure-containing pyrazole norcantharidin derivatives, representing a significant evolution in the design of anti-tumor agents. This technology addresses the historical limitations of cantharidin, a natural product known for its potent biological activity but also for its severe toxicity and complex synthesis. By strategically modifying the norcantharidin backbone through the introduction of pyrazole and chromone moieties, this invention achieves a delicate balance between efficacy and safety. The core innovation lies in the 1,3-dipolar cycloaddition method, which allows for the precise construction of these complex heterocyclic systems. For R&D directors and procurement specialists, understanding the chemical robustness of this pathway is essential for securing a reliable supply of high-value pharmaceutical intermediates. The following analysis details the technical merits and commercial viability of this synthesis, highlighting its potential for cost reduction in anti-tumor drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
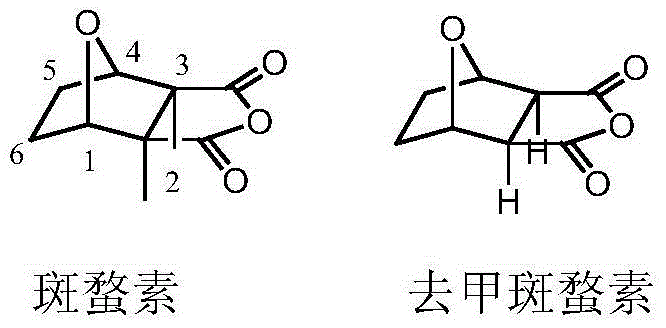
Traditional approaches to synthesizing cantharidin-based therapeutics have long been plagued by significant structural and toxicological challenges that hinder their widespread clinical adoption. The parent compound, cantharidin, possesses a rigid bicyclic structure that, while biologically active, is associated with severe irritation to the urinary system and other dose-limiting side effects. Furthermore, the synthetic routes to modify the cantharidin skeleton often involve harsh reaction conditions, multiple protection and deprotection steps, and the use of expensive or hazardous reagents that complicate the purification process. These conventional methods frequently result in low overall yields and generate substantial chemical waste, which poses both environmental and economic burdens on the manufacturing process. The inability to easily functionalize the C5 and C6 positions without compromising the stability of the anhydride ring has historically restricted the chemical diversity available to medicinal chemists. Consequently, the supply chain for these intermediates has been fragile, with high production costs translating into limited availability for downstream drug development projects.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a streamlined strategy that leverages the inherent reactivity of dehydronorcantharidin to build complexity efficiently. By removing the methyl groups found in natural cantharidin to form norcantharidin, the toxicity profile is drastically improved while retaining the core pharmacophore responsible for protein phosphatase inhibition. The innovation extends further by employing a 1,3-dipolar cycloaddition to fuse a pyrazole ring and a chromone structure directly onto the norcantharidin scaffold. This methodology not only simplifies the synthetic sequence by reducing the number of discrete steps but also enhances the structural diversity through the use of various substituted anilines and chromone hydrazones. The result is a series of derivatives that exhibit superior anti-tumor activity compared to the parent compound, with a much more favorable safety margin. This strategic modification opens new avenues for drug discovery, providing a robust platform for the development of next-generation oncology therapeutics that are both potent and safer for patients.
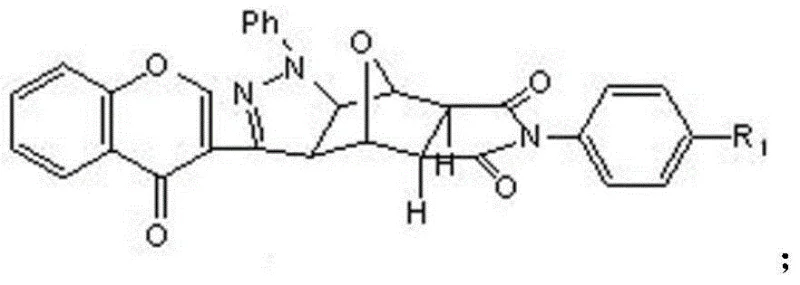
Mechanistic Insights into 1,3-Dipolar Cycloaddition and Structural Modification
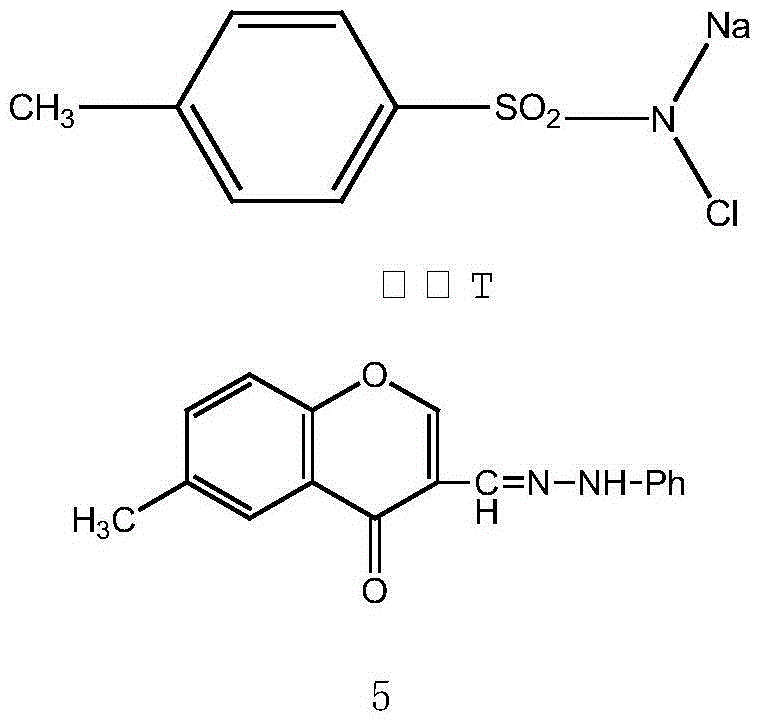
The heart of this synthetic innovation is the 1,3-dipolar cycloaddition reaction, a powerful tool in organic synthesis for constructing five-membered heterocyclic rings with high regioselectivity and stereocontrol. In this specific application, the reaction involves the generation of a nitrile imine dipole in situ from the 6-methyl substituted chromone phenylhydrazone using chloramine-T as the oxidant. This dipole then reacts with the electron-deficient double bond of the N-substituted dehydronorcantharidin imide to form the pyrazoline ring system. The mechanism is highly efficient because it forms two new carbon-nitrogen bonds and one carbon-carbon bond in a single concerted step, significantly reducing the atom economy waste typically associated with stepwise ring closures. The presence of the chromone moiety adds further electronic stabilization to the transition state, facilitating the reaction under relatively mild reflux conditions in ethanol. This mechanistic elegance ensures that the final products are formed with high purity, minimizing the formation of regioisomers that would otherwise require difficult and costly chromatographic separations.
Impurity control is a critical aspect of this process, particularly given the intended application in pharmaceutical manufacturing where strict regulatory standards must be met. The use of recrystallization from methanol as the final purification step is a testament to the high crystallinity and purity achievable through this route. By carefully controlling the stoichiometry of the chloramine-T oxidant and the reaction temperature, the formation of over-oxidized byproducts or hydrolysis of the anhydride ring is effectively suppressed. The structural rigidity imparted by the fused pyrazole and chromone rings also contributes to the stability of the final derivative, reducing the likelihood of degradation during storage or subsequent formulation. For quality control teams, this means that the impurity profile is predictable and manageable, with the major impurities being easily removable through standard workup procedures. This level of control is essential for scaling the process from gram-scale laboratory synthesis to multi-kilogram commercial production without compromising on the quality attributes required for clinical trials.
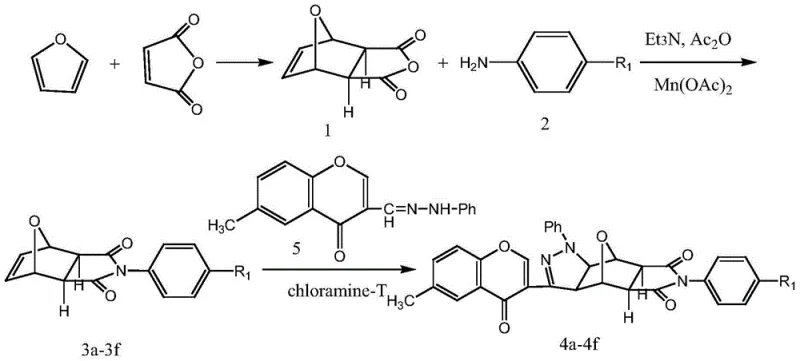
How to Synthesize Chromone-Containing Pyrazole Norcantharidin Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical three-stage sequence that begins with the preparation of the norcantharidin core and culminates in the cycloaddition step. The process is designed to be operationally simple, utilizing common solvents like ethanol, acetone, and ethyl acetate, which are readily available and easy to recover on a large scale. The initial Diels-Alder reaction between furan and maleic anhydride sets the stereochemical foundation for the entire molecule, establishing the bridged bicyclic system that is crucial for biological activity. Subsequent N-substitution allows for the introduction of diverse aryl groups, enabling the fine-tuning of physicochemical properties such as solubility and metabolic stability. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are provided in the guide below to ensure reproducibility and consistency across different production batches.
- Synthesize dehydronorcantharidin via Diels-Alder reaction between furan and maleic anhydride at room temperature.
- Perform N-substitution using aniline derivatives and manganese acetate to form N-substituted dehydronorcantharidin imide.
- Execute 1,3-dipolar cycloaddition with 6-methyl chromone phenylhydrazone and chloramine-T under reflux to finalize the derivative structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages that directly address the pain points of procurement managers and supply chain directors in the pharmaceutical industry. The primary benefit lies in the significant cost reduction in anti-tumor drug manufacturing achieved through the use of inexpensive and abundant starting materials. Furan and maleic anhydride are commodity chemicals produced on a massive global scale, ensuring a stable and secure supply base that is not subject to the volatility often seen with exotic natural product extracts. Furthermore, the elimination of transition metal catalysts in the key bond-forming steps removes the need for expensive metal scavenging processes and rigorous testing for residual metals, which are major cost drivers in API production. The simplified purification protocol, relying on crystallization rather than complex chromatography, further drives down the cost of goods sold, making these derivatives economically viable for large-scale therapeutic applications.
- Cost Reduction in Manufacturing: The process achieves cost efficiency by avoiding the use of precious metal catalysts and reducing the number of synthetic steps required to reach the final target. By utilizing a convergent synthesis strategy where the chromone and norcantharidin fragments are coupled late in the sequence, the overall yield is maximized, and the loss of valuable intermediates is minimized. This efficiency translates directly into lower raw material costs and reduced waste disposal fees, providing a competitive edge in the pricing of the final pharmaceutical intermediate. Additionally, the use of ethanol as the primary solvent for the final step aligns with green chemistry principles, reducing the environmental compliance costs associated with hazardous solvent handling and disposal.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like furan, maleic anhydride, and aniline derivatives ensures that the supply chain is robust and resilient against disruptions. Unlike processes that depend on single-source natural extracts or specialized reagents with long lead times, this synthetic route can be rapidly scaled up using existing infrastructure in most fine chemical manufacturing facilities. The stability of the intermediates also allows for the strategic stocking of key precursors, enabling manufacturers to respond quickly to fluctuations in demand from downstream drug developers. This reliability is crucial for maintaining continuous production schedules and meeting the tight deadlines often imposed by clinical trial timelines.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require high-pressure equipment or cryogenic temperatures, making the process inherently safer and easier to scale from pilot plant to commercial tonnage. The absence of heavy metals and the use of recyclable solvents simplify the waste treatment process, ensuring compliance with increasingly stringent environmental regulations. This environmental friendliness not only reduces the risk of regulatory shutdowns but also enhances the corporate social responsibility profile of the manufacturing partner. The ability to produce these complex derivatives in large quantities without compromising on safety or quality makes this technology an ideal candidate for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these norcantharidin derivatives. These answers are derived directly from the patent data and practical manufacturing experience, providing clarity on the feasibility and benefits of this technology. Understanding these details is vital for stakeholders evaluating the potential of this route for their specific drug development pipelines. The responses cover aspects ranging from structural activity relationships to process scalability, ensuring a comprehensive overview of the technology's value proposition.
Q: How does the chromone structure modification affect the toxicity of norcantharidin?
A: The introduction of the chromone and pyrazole structures at the C5 and C6 positions significantly reduces the urinary system toxicity associated with traditional cantharidin while maintaining or enhancing anti-tumor activity.
Q: What are the key reagents required for the 1,3-dipolar cycloaddition step?
A: The critical reagents include N-substituted dehydronorcantharidin imide, 6-methyl substituted chromone phenylhydrazone, and chloramine-T as the oxidant in an ethanol solvent system.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available starting materials like furan and maleic anhydride and avoids complex transition metal catalysts, making it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chromone-Containing Pyrazole Norcantharidin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex patent chemistry into reliable commercial reality. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand that the synthesis of chromone-containing pyrazole norcantharidin derivatives requires meticulous attention to detail, particularly in the control of stereochemistry and impurity profiles. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch we produce. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global oncology market.
We invite you to collaborate with us to leverage this innovative technology for your next-generation anti-tumor drug projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to our deep technical expertise and robust supply chain capabilities. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how we can support your journey from discovery to commercial success with reliable, high-quality chemical solutions.