Revolutionizing Oncology Intermediates: Scalable Synthesis of Novel Norcantharidin-Pyrazole Hybrids
Revolutionizing Oncology Intermediates: Scalable Synthesis of Novel Norcantharidin-Pyrazole Hybrids
Introduction: The Evolution of Cantharidin-Based Therapeutics
The landscape of oncology drug development is constantly shifting towards molecules that offer higher efficacy with minimized systemic toxicity, a challenge that has driven significant innovation in the field of natural product derivatives. Patent CN103554123A, published in early 2014, presents a groundbreaking approach to modifying the backbone of norcantharidin, a less toxic analog of the potent but dangerous cantharidin found in blister beetles. This intellectual property details the synthesis of a novel series of chromone structure-containing pyrazole norcantharidin derivatives, which represent a significant leap forward in the design of anti-tumor pharmaceutical intermediates. By strategically introducing a pyrazole ring at the C5 and C6 positions and fusing a chromone moiety, the inventors have created a chemical architecture that retains the leukocyte-increasing and immunosuppressive benefits of the parent compound while substantially mitigating its notorious side effects on the urinary system. This technical breakthrough provides a robust foundation for developing next-generation anticancer agents that are safer for patients and more viable for commercial manufacturing.
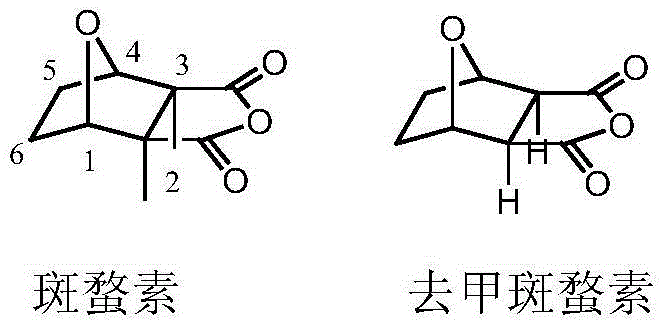
For procurement specialists and supply chain directors evaluating reliable pharmaceutical intermediates suppliers, understanding the structural nuances depicted in the patent is crucial for assessing long-term viability. The transition from the methyl-substituted cantharidin to the demethylated norcantharidin backbone is not merely a chemical triviality but a strategic maneuver to enhance the therapeutic index. The removal of the 2,3-dimethyl groups simplifies the synthetic pathway and reduces the metabolic burden on the patient, which translates to a more marketable end-product for pharmaceutical partners. As we delve deeper into the technical specifics, it becomes evident that this patent offers more than just a new molecule; it offers a scalable, cost-effective platform for producing high-value oncology intermediates that address the critical unmet needs of modern cancer therapy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the utilization of cantharidin and its direct derivatives in clinical settings has been severely hampered by their narrow therapeutic window and severe toxicity profiles, particularly regarding renal irritation and urinary system damage. Conventional synthesis methods often struggle to modify the rigid bicyclic structure of cantharidin without compromising its biological activity or requiring prohibitively expensive and hazardous reagents. Traditional approaches to backbone modification frequently involve multi-step protections and deprotections that lower overall yields and generate substantial chemical waste, thereby inflating the cost of goods sold (COGS) for the final active pharmaceutical ingredient. Furthermore, the lack of structural diversity in earlier generations of norcantharidin derivatives limited their ability to overcome drug resistance mechanisms in tumor cells, rendering them less effective against aggressive malignancies. These inherent limitations in conventional chemistry have created a bottleneck in the supply chain for high-purity oncology intermediates, necessitating a shift towards more innovative synthetic strategies.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent leverages a sophisticated 1,3-dipolar cycloaddition strategy to construct a hybrid molecular framework that merges the pharmacophore of norcantharidin with the bioactive properties of chromone. This method allows for the precise introduction of a pyrazole ring and a chromone structure onto the norcantharidin scaffold, creating a diverse library of derivatives with tunable electronic and steric properties. By utilizing 6-methyl substituted chromone phenylhydrazone as a key building block, the synthesis achieves a level of structural complexity that was previously difficult to attain with high regioselectivity. This innovation not only enhances the anti-tumor activity against various cell lines, including liver and stomach cancer, but also streamlines the production process by reducing the number of purification steps required. For a reliable pharmaceutical intermediates supplier, adopting this novel approach means offering clients a product with superior purity profiles and a more competitive cost structure derived from efficient chemistry.
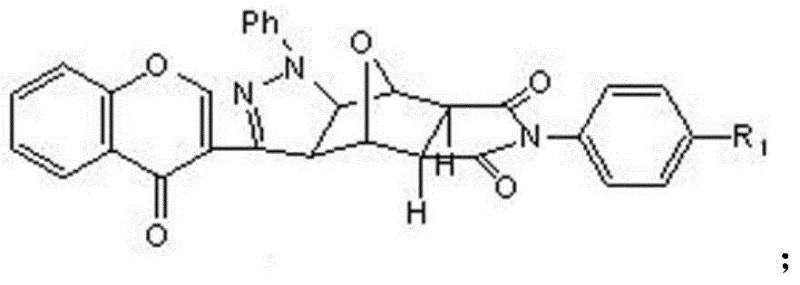
Mechanistic Insights into 1,3-Dipolar Cycloaddition and Structural Fusion
The core of this technological advancement lies in the intricate mechanistic pathway of the 1,3-dipolar cycloaddition, which serves as the pivotal step for fusing the heterocyclic systems. The reaction begins with the generation of a nitrile imine dipole from the chromone phenylhydrazone precursor, which is subsequently trapped by the dipolarophile present in the norcantharidin derivative. This concerted pericyclic reaction proceeds through a highly ordered transition state that ensures the formation of the pyrazoline ring with specific stereochemistry, critical for the biological interaction with tumor targets. The presence of the chloramine-T oxidant facilitates the subsequent aromatization or stabilization of the intermediate, locking the chromone and pyrazole moieties into a rigid, planar configuration that enhances DNA intercalation or enzyme inhibition capabilities. Understanding this mechanism is vital for R&D directors focused on impurity control, as deviations in temperature or stoichiometry during this phase could lead to regioisomers that complicate downstream purification and compromise the safety profile of the final drug substance.
Furthermore, the impurity control mechanism inherent in this synthetic route is designed to minimize the formation of toxic by-products that often plague cantharidin synthesis. The use of specific solvents like ethanol and dimethylformamide, combined with controlled temperature gradients during the recrystallization phases, ensures that unreacted starting materials and side products are effectively separated from the target molecule. The structural rigidity imparted by the fused ring system also contributes to the chemical stability of the derivative, reducing the risk of degradation during storage and transport, which is a key concern for supply chain heads managing global logistics. By mastering these mechanistic details, manufacturers can guarantee the delivery of high-purity norcantharidin derivatives that meet the stringent quality standards required by regulatory bodies in major pharmaceutical markets. This level of control over the chemical process underscores the feasibility of commercial scale-up of complex heterocyclic compounds for industrial applications.
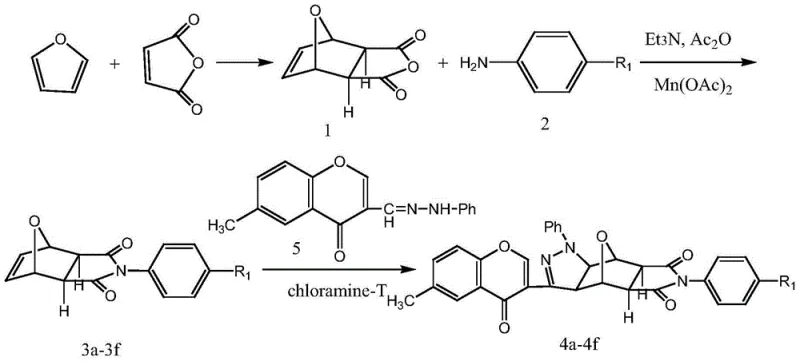
How to Synthesize Chromone-Containing Pyrazole Norcantharidin Derivatives Efficiently
The practical execution of this synthesis involves a carefully orchestrated sequence of reactions that transform simple commodity chemicals into high-value therapeutic intermediates. The process initiates with the Diels-Alder cycloaddition between furan and maleic anhydride to form the norcantharidin core, followed by amidation with substituted anilines to introduce nitrogen functionality. The final and most critical stage involves the coupling of this intermediate with the chromone hydrazone under reflux conditions, a step that demands precise monitoring to ensure complete conversion and optimal yield. Detailed standardized synthesis steps are essential for maintaining batch-to-batch consistency, which is why we have outlined the specific operational parameters below for technical reference.
- Synthesize dehydronorcantharidin imide element via Diels-Alder reaction between maleic anhydride and furan under room temperature conditions.
- Prepare N-substituted dehydronorcantharidin imide by reacting the imide element with aniline derivatives using manganese acetate and triethylamine catalysts.
- Execute the final 1,3-dipolar cycloaddition by refluxing the N-substituted imide with 6-methyl substituted chromone phenylhydrazone and chloramine-T.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement managers seeking cost reduction in anti-tumor drug manufacturing without compromising on quality. The elimination of expensive transition metal catalysts in favor of more abundant reagents like chloramine-T and manganese acetate significantly lowers the raw material costs and simplifies the waste treatment process, leading to substantial cost savings in the overall production budget. Additionally, the improved solubility and crystallinity of the final derivatives facilitate easier isolation and drying, which reduces energy consumption and processing time in the manufacturing plant. These efficiencies translate directly into a more competitive pricing structure for the end client, allowing pharmaceutical companies to allocate resources towards further clinical development rather than excessive production overheads.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway minimizes the number of unit operations required, thereby reducing labor costs and equipment occupancy time which drives down the overall manufacturing expense. By avoiding the use of precious metal catalysts that require complex removal and recovery steps, the process eliminates a significant source of variable cost and potential contamination risk. The high atom economy of the cycloaddition reaction ensures that a greater proportion of the starting mass is converted into the desired product, reducing the volume of chemical waste that needs to be disposed of in compliance with environmental regulations. Consequently, this leads to a leaner, more economical production model that enhances the margin potential for all stakeholders in the supply chain.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals such as furan, maleic anhydride, and substituted anilines ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or raw material shortages. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures or high-pressure vessels, allows for production in a wider range of manufacturing facilities, increasing the redundancy and flexibility of the supply network. This accessibility of raw materials and simplicity of processing conditions significantly mitigates the risk of production delays, ensuring consistent availability of high-purity norcantharidin derivatives for downstream drug formulation. Reducing lead time for high-purity oncology intermediates becomes achievable when the underlying chemistry is this forgiving and scalable.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that can be easily translated from laboratory glassware to industrial-sized reactors without significant loss of efficiency or selectivity. The use of common organic solvents like ethanol and acetone simplifies the solvent recovery and recycling processes, aligning the manufacturing practice with green chemistry principles and strict environmental compliance standards. The reduction in toxic by-products and the absence of heavy metal residues simplify the effluent treatment requirements, lowering the environmental footprint of the production facility. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing partner, making it a preferred choice for ethically conscious pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel derivatives, based on the detailed specifications found within the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of integrating this technology into your existing drug development pipeline. We encourage technical teams to review these points closely to understand the full scope of the opportunities presented by this chemistry.
Q: How does the pyrazole-chromone modification affect the toxicity profile compared to native cantharidin?
A: The introduction of the pyrazole ring at the C5 and C6 sites, combined with the chromone structure, significantly reduces the urinary system irritation and general toxicity associated with native cantharidin while maintaining or enhancing anti-tumor potency.
Q: What are the critical reaction conditions for the 1,3-dipolar cycloaddition step?
A: The key cycloaddition step requires refluxing in ethanol solvent for approximately 12 hours with chloramine-T as the oxidant to ensure the successful formation of the pyrazole ring fusion.
Q: Is this synthesis route suitable for large-scale commercial production of oncology intermediates?
A: Yes, the route utilizes readily available starting materials like maleic anhydride and furan, and avoids extreme pressure or cryogenic conditions, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Norcantharidin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chromone-containing pyrazole norcantharidin derivatives in the fight against cancer, and we are uniquely positioned to bring this technology from the lab to the clinic. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of intermediate we produce meets the highest international standards, providing you with the confidence to advance your clinical trials without supply chain interruptions.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthetic route for your oncology portfolio. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your time-to-market and optimize your manufacturing economics.
