Revolutionizing Adapalene Production with Advanced N-Heterocyclic Carbene Palladium Catalysts
Revolutionizing Adapalene Production with Advanced N-Heterocyclic Carbene Palladium Catalysts
The pharmaceutical industry is constantly seeking more efficient pathways for the synthesis of high-value dermatological agents, and the recent disclosure in patent CN113262822A represents a significant leap forward in this domain. This patent details the development of a novel N-heterocyclic carbene (NHC) metal palladium complex catalyst, specifically engineered to streamline the production of adapalene, a critical retinoid used in acne treatment. Unlike traditional methods that rely on cumbersome multi-step sequences involving unstable organometallic reagents, this innovation leverages the unique electronic and steric properties of an acenaphthene-fused imidazolium framework to achieve superior catalytic performance. For R&D directors and process chemists, this technology offers a compelling alternative that simplifies the synthetic route while maintaining high purity standards. By integrating this advanced catalytic system, manufacturers can transition from legacy processes to a more robust, scalable, and economically viable production model for key pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of adapalene has been plagued by inefficiencies inherent to classical organometallic chemistry. The conventional route typically begins with a Friedel-Crafts alkylation to introduce the adamantyl group, followed by methylation to protect the phenol. The critical bottleneck arises in the subsequent coupling step, which necessitates the formation of a Grignard reagent from the bromoanisole derivative. Grignard reagents are notoriously sensitive to moisture and oxygen, requiring strictly anhydrous conditions and specialized handling equipment that drives up operational costs. Furthermore, the process often involves a transmetallation to an organozinc species before the final coupling with the naphthoate fragment can occur. This elongated sequence not only results in poor atom economy but also accumulates impurities at each stage, complicating downstream purification and reducing overall yield. The instability of these intermediates poses significant safety risks and supply chain vulnerabilities, making the traditional route less attractive for modern, high-volume manufacturing environments.
The Novel Approach
In stark contrast, the methodology outlined in CN113262822A introduces a streamlined Suzuki-Miyaura coupling strategy that bypasses the need for Grignard or zinc reagents entirely. By utilizing a specially designed cyclopalladated catalyst, the synthesis achieves the direct coupling of 2-(1-adamantyl)-4-bromoanisole with a naphthoate boronate ester in a single, efficient step. This approach drastically shortens the synthetic timeline and eliminates the hazards associated with pyrophoric reagents. The reaction proceeds under relatively mild thermal conditions, typically between 90°C and 110°C, using common solvents like toluene. This shift not only enhances safety but also significantly reduces the complexity of the reactor setup required for production. The ability to directly access the key ester precursor in high yield sets the stage for a simplified hydrolysis step to finalize the adapalene molecule, offering a clear pathway for cost reduction in API manufacturing.
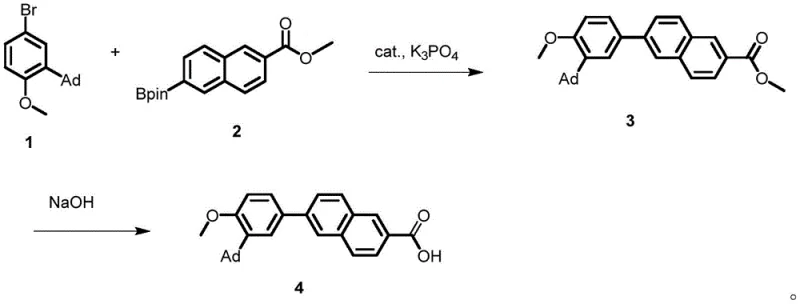
Mechanistic Insights into NHC-Pd Catalyzed Coupling
The exceptional performance of this catalyst stems from its sophisticated molecular architecture, which combines an acenaphthene backbone with a nitrogen heterocyclic carbene ligand. The acenaphthene moiety acts as a powerful electron-donating group, enriching the electron density at the palladium center. This electronic enrichment is crucial for facilitating the oxidative addition step of the catalytic cycle, particularly when dealing with sterically hindered substrates like the adamantyl-substituted bromoanisole. Additionally, the catalyst features bulky isopropyl phenyl groups on the nitrogen atoms, which create a protective steric environment around the metal center. This steric bulk is not merely structural; it plays a functional role in shielding the active site from deactivation by water or oxygen, thereby enhancing the catalyst's tolerance to ambient conditions compared to standard phosphine-based systems. The result is a catalytic species that maintains high activity over extended periods, ensuring consistent conversion rates throughout the reaction batch.
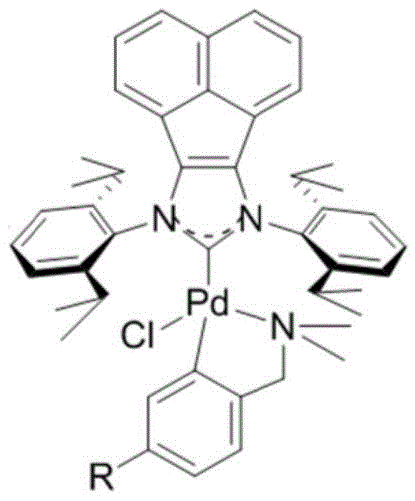
Furthermore, the synthesis of the catalyst itself is designed for practicality and scalability. As illustrated in the preparation scheme, the complex is formed by reacting acenaphthene imidazole hydrochloride with palladium chloride and specific benzylamine derivatives in the presence of a base like potassium carbonate. This cyclometallation process activates a phenyl carbon-hydrogen bond, locking the ligand into a rigid, stable ring structure that prevents ligand dissociation during the harsh conditions of the coupling reaction. The benzyl amino group further modulates the electronic properties, fine-tuning the catalyst for optimal turnover. This robust design ensures that the catalyst remains intact and active, minimizing the formation of palladium black or other inactive species that often contaminate the final product. For quality control teams, this stability translates to lower residual metal levels in the API, simplifying the purification process and ensuring compliance with stringent regulatory limits.
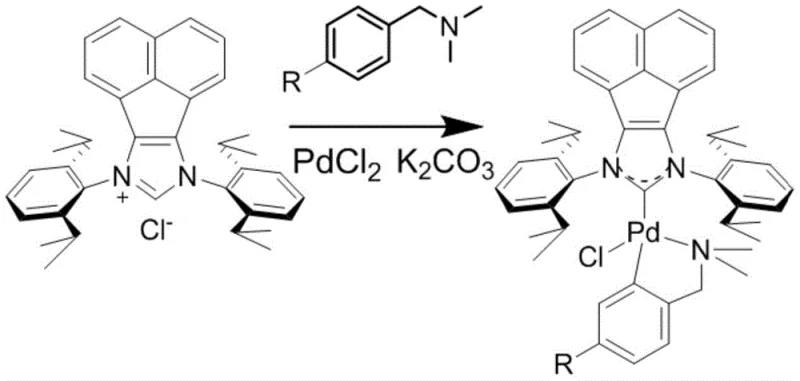
How to Synthesize Adapalene Efficiently
Implementing this novel catalytic system requires precise control over reaction parameters to maximize yield and purity. The process begins with the in-situ or ex-situ generation of the active palladium species, followed by the introduction of the aryl halide and boronate partners. The reaction is typically conducted under a nitrogen atmosphere to prevent any potential oxidation, although the catalyst's design offers improved resilience compared to predecessors. Following the coupling, the crude ester intermediate is isolated and subjected to basic hydrolysis to reveal the carboxylic acid functionality of adapalene. The detailed standardized synthesis steps below outline the specific stoichiometry, temperature profiles, and workup procedures required to replicate the high yields reported in the patent data, serving as a foundational guide for process engineers looking to adopt this technology.
- Prepare the N-heterocyclic carbene palladium complex catalyst by reacting acenaphthene imidazole hydrochloride with palladium chloride and N,N-dimethylbenzylamine derivatives in acetonitrile.
- Perform the Suzuki coupling reaction between 2-(1-adamantyl)-4-bromoanisole and the naphthoate boronate ester using the novel catalyst and potassium phosphate in toluene at 90-110°C.
- Hydrolyze the resulting methyl ester intermediate using sodium hydroxide solution, followed by acidification to isolate the final adapalene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-palladium catalyst technology presents a multitude of strategic benefits that extend beyond simple chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for magnesium turnings, alkyl halides for Grignard formation, and zinc salts, the supply chain becomes less vulnerable to the price volatility and availability issues often associated with reactive metals. Moreover, the removal of these hazardous reagents reduces the burden on waste management systems and lowers the costs related to safety compliance and disposal. The streamlined process also means fewer unit operations are required, which directly correlates to reduced energy consumption and shorter batch cycle times. These factors collectively contribute to a more resilient and cost-effective manufacturing operation, allowing companies to respond more agilely to market demands for dermatological treatments.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic route is profound, primarily driven by the elimination of expensive and hazardous organometallic reagents. Traditional Grignard processes require rigorous drying of solvents and reactors, which consumes significant energy and time; the new method operates under much milder conditions, substantially lowering utility costs. Additionally, the high catalytic activity allows for the use of lower catalyst loadings while maintaining excellent conversion, reducing the consumption of precious palladium. The simplified downstream processing, resulting from fewer side reactions and impurities, further decreases the cost of goods sold by minimizing chromatography or recrystallization steps. Overall, the process optimization leads to significant cost savings without compromising the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved due to the stability and commercial availability of the starting materials. Unlike Grignard reagents which must be prepared fresh and cannot be stored, the boronate esters and aryl halides used in this Suzuki coupling are stable, shelf-stable commodities that can be sourced from multiple reliable suppliers globally. This flexibility reduces the risk of production stoppages caused by the degradation of sensitive reagents. Furthermore, the catalyst itself can potentially be immobilized on supports like nano-silicon dioxide, as suggested in the patent, which opens the door for heterogeneous catalysis. This would allow for catalyst recovery and reuse, further stabilizing the supply of the catalytic system and reducing dependency on continuous fresh catalyst synthesis.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the robust nature of the reaction conditions. The tolerance to moisture and oxygen means that the stringent engineering controls required for pyrophoric materials are relaxed, making it easier to adapt existing multipurpose reactors for this synthesis. From an environmental perspective, the atom economy of the Suzuki coupling is superior to the Grignard-Zinc route, generating less inorganic salt waste. The use of toluene as a solvent, while requiring proper management, is standard in the industry and easier to recover and recycle compared to the ether solvents often used in Grignard reactions. These factors make the process more sustainable and aligned with modern green chemistry principles, easing the regulatory approval process for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented catalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating the feasibility of this route. Understanding these nuances is critical for making informed decisions about process adoption and technology transfer.
Q: How does the new Pd-NHC catalyst improve upon traditional Grignard methods?
A: The novel catalyst eliminates the need for unstable Grignard and organozinc reagents, allowing for a direct one-step coupling under milder conditions with higher atom economy.
Q: What are the stability advantages of this cyclopalladated complex?
A: The acenaphthene backbone provides strong electron donation, while the bulky isopropyl phenyl groups offer steric hindrance that enhances resistance to water and oxygen interference.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap chloride substrates, operates at moderate temperatures (90-110°C), and avoids strictly anhydrous conditions, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adapalene Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the NHC-palladium complex described in CN113262822A. As a leading CDMO partner, we possess the technical expertise to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle complex organometallic chemistry with the highest safety standards, ensuring that the transition from traditional methods to this novel catalytic route is seamless. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and reliability. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of adapalene intermediate meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your dermatological product lines. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes, demonstrating exactly how this technology can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage in the market through superior process chemistry and supply chain excellence.
