Scalable Metal-Free Synthesis of 3,4-Disubstituted Pyrroles for Advanced Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery. Patent CN114133349A, published in early 2022, introduces a groundbreaking preparation method for 3,4-disubstituted pyrrole derivatives that fundamentally shifts the paradigm from traditional transition metal catalysis to a greener, organocatalytic approach. This innovation utilizes tetrabutylammonium iodide (TBAI) as a cheap and efficient catalyst in conjunction with polymethylsiloxane (PMHS) as a hydrogen source, operating effectively under an oxygen atmosphere. For R&D directors and procurement specialists alike, this technology represents a significant leap forward, offering a route that avoids the complexities of heavy metal removal while maintaining high yields and broad substrate scope. The ability to synthesize these valuable pyrrole cores without relying on precious metals like Ruthenium, Copper, or Nickel addresses a major pain point in API intermediate manufacturing, promising both economic and environmental benefits for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-disubstituted pyrrole derivatives has heavily relied on transition metal catalysts such as Ruthenium, Copper, Nickel, or Silver, often requiring harsh acidic or basic conditions to drive the cyclization reactions. These conventional methodologies present substantial drawbacks for commercial manufacturing, primarily due to the high cost of the catalysts and the stringent regulatory limits on residual heavy metals in pharmaceutical products. The necessity for extensive downstream processing to scavenge trace metals not only increases the overall production cost but also complicates the supply chain by requiring specialized reagents and additional unit operations. Furthermore, many traditional routes suffer from limited functional group tolerance, where sensitive moieties might be degraded under the aggressive acidic or basic conditions required, leading to lower overall yields and difficult purification challenges. These factors collectively limit the widespread application of such methods in the rapid and cost-sensitive environment of modern drug development.
The Novel Approach
In stark contrast to the metal-dependent legacy processes, the novel method disclosed in the patent employs a metal-free system driven by TBAI and a silane reductant, creating a much milder and more environmentally benign reaction profile. By utilizing TBAI, an inexpensive phase-transfer catalyst that also serves as a source of iodine radicals, the process initiates a radical cyclization cascade that efficiently constructs the pyrrole ring without the need for toxic transition metals. This approach not only drastically simplifies the work-up procedure by eliminating metal scavenging steps but also enhances the safety profile of the manufacturing process by avoiding strong acids or bases. The reaction proceeds smoothly in common organic solvents like isopropanol at moderate temperatures, demonstrating exceptional compatibility with a wide array of functional groups including halogens, esters, and ketones. This versatility makes the new route highly attractive for the synthesis of complex pharmaceutical intermediates where structural diversity is key.

Mechanistic Insights into TBAI-Catalyzed Radical Cyclization
The mechanistic pathway of this transformation is believed to involve a radical-mediated cyclization initiated by the interaction between TBAI and molecular oxygen. Under the reaction conditions, TBAI likely generates iodine radicals which abstract hydrogen or interact with the bromoallyl moiety of the substrate to form carbon-centered radicals. These reactive intermediates then undergo intramolecular addition to the alkene double bond, closing the five-membered pyrrole ring. The presence of oxygen is crucial as it helps regenerate the active iodine species and drives the oxidative aromatization of the dihydropyrrole intermediate to the final aromatic pyrrole product. Simultaneously, the silane reagent, specifically PMHS, acts as a terminal reductant to quench radical species and facilitate the overall redox balance of the reaction. This delicate interplay between the iodine catalyst, oxygen oxidant, and silane reductant ensures a high turnover number and minimizes the formation of polymeric byproducts or oligomers that often plague radical reactions.
From an impurity control perspective, the absence of transition metals inherently reduces the risk of metal-catalyzed side reactions such as homocoupling or over-reduction, which are common pitfalls in traditional methods. The use of PMHS, a polymeric siloxane, offers a controlled release of hydride equivalents, preventing the violent exotherms sometimes associated with low molecular weight silanes like triethylsilane. This controlled reactivity contributes to a cleaner reaction profile, as evidenced by the high isolated yields reported across various substrates. For quality control teams, this means a simpler impurity profile that is easier to characterize and control, ensuring that the final API intermediate meets the stringent purity specifications required by global regulatory agencies. The robustness of this mechanism against varying electronic properties of the substrate further underscores its reliability for diverse synthetic campaigns.
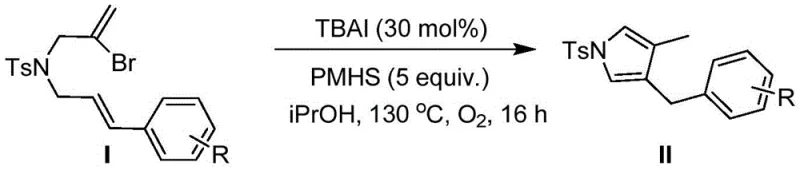
How to Synthesize 3-Benzyl-4-methyl-1-tosyl-1H-pyrrole Efficiently
To implement this synthesis in a laboratory or pilot plant setting, the process begins with the precise weighing of N-(2-bromoallyl)-N-cinnamyl-4-methylbenzenesulfonamide as the starting material. The reaction is typically conducted in a sealed pressure vessel to maintain the oxygen atmosphere required for the catalytic cycle. Isopropanol is selected as the solvent of choice due to its ability to dissolve both the organic substrate and the ionic catalyst while remaining stable under the oxidative conditions. The addition of TBAI at a loading of 30 mol% and PMHS at 5 equivalents ensures that the reaction kinetics are optimized for maximum conversion. Detailed standardized synthesis steps follow below to guide the technical team through the execution of this high-yielding transformation.
- Charge a dry pressure tube with the sulfonamide substrate, TBAI catalyst (30 mol%), and PMHS hydrogen source in isopropanol solvent.
- Heat the reaction mixture to 130°C under an oxygen atmosphere for 16 hours to facilitate radical cyclization.
- Quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography to isolate the target pyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TBAI-catalyzed methodology offers profound strategic advantages that extend beyond simple chemistry. The shift away from precious metal catalysts directly translates to a significant reduction in raw material costs, as TBAI is a commodity chemical available in bulk quantities at a fraction of the price of Ruthenium or Palladium complexes. Furthermore, the elimination of metal scavengers and the associated filtration steps streamlines the manufacturing workflow, reducing the consumption of auxiliary materials and shortening the overall batch cycle time. This efficiency gain allows for better utilization of reactor capacity and faster time-to-market for new drug candidates, providing a competitive edge in the fast-paced pharmaceutical landscape.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive transition metal catalysts with TBAI cannot be overstated, as it removes a major cost driver from the bill of materials. Additionally, the simplified work-up procedure, which avoids complex metal removal protocols, reduces labor costs and waste disposal fees associated with hazardous metal-contaminated streams. The use of PMHS, a stable and inexpensive polymeric siloxane, further contributes to cost savings compared to more volatile and pricey hydride sources. Overall, the process economics are vastly improved, making the production of these pyrrole derivatives commercially viable even for price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can often be a bottleneck due to geopolitical factors and limited supplier bases, whereas TBAI and PMHS are widely produced by numerous chemical manufacturers globally. This abundance ensures a stable and resilient supply chain, minimizing the risk of production delays caused by raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further enhancing supply chain reliability. Companies can confidently scale up production knowing that the critical inputs are readily available and not subject to the volatility of the precious metals market.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and uses isopropanol, a green solvent with a favorable safety profile, making it highly amenable to scale-up in standard stainless steel reactors. The absence of heavy metals simplifies environmental compliance, as wastewater treatment becomes less burdensome without the need for specialized metal precipitation units. This aligns perfectly with the industry's growing emphasis on green chemistry and sustainability, allowing manufacturers to reduce their environmental footprint while maintaining high productivity. The process generates minimal hazardous waste, supporting corporate sustainability goals and reducing the long-term liability associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel pyrrole synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential adopters. Understanding these nuances is critical for R&D teams planning to integrate this method into their existing synthetic portfolios.
Q: What are the primary advantages of using TBAI over transition metal catalysts for pyrrole synthesis?
A: The use of Tetrabutylammonium Iodide (TBAI) eliminates the need for expensive and toxic transition metals like Ruthenium or Copper. This significantly simplifies the purification process by removing the requirement for rigorous heavy metal scavenging steps, thereby reducing production costs and ensuring higher purity profiles suitable for pharmaceutical applications.
Q: Can this synthesis method accommodate diverse functional groups on the aromatic ring?
A: Yes, the patented methodology demonstrates excellent substrate tolerance. It successfully accommodates a wide range of substituents including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, trifluoromethyl, acetyl, and ester functionalities, yielding the corresponding pyrrole derivatives in good to excellent yields.
Q: Is the reaction scalable for commercial manufacturing of API intermediates?
A: The reaction utilizes common industrial solvents like isopropanol and operates at moderate temperatures (130°C) under oxygen pressure, which are conditions readily adaptable to large-scale reactors. The absence of sensitive metal catalysts and the use of robust reagents like PMHS suggest high potential for successful commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Disubstituted Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free catalytic systems like the one described in CN114133349A for the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless and efficient. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to guarantee that every batch meets the highest international standards for API intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of organocatalytic processes, providing a safe and compliant environment for your most sensitive projects.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your timeline and optimize your budget. Let us be your partner in turning innovative chemistry into commercial success.
