Advanced Synthesis of Carbazole-Containing Discotic Liquid Crystals for Commercial OLED Applications
The rapid evolution of organic electronics has intensified the demand for high-performance materials capable of efficient charge transport and light emission. Patent CN111825598A introduces a groundbreaking methodology for synthesizing carbazole-containing discotic liquid crystal compounds, specifically targeting the next generation of organic light-emitting diodes (OLEDs) and photovoltaic devices. This technology leverages a unique molecular architecture where a carbazole core is fused with triphenylene units, creating a robust pi-conjugated system that exhibits both excellent luminescence and self-assembling liquid crystalline properties. The ability of these molecules to form stable hexagonal columnar phases over a wide temperature range addresses critical stability issues found in conventional amorphous organic semiconductors. By integrating the hole-transport characteristics of carbazole with the columnar stacking of discotic liquid crystals, this invention offers a dual-functionality material that simplifies device architecture while enhancing performance metrics.
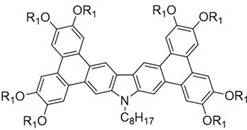
For R&D directors evaluating new material platforms, the structural versatility presented in this patent is particularly compelling. The general formulas (I) and (II) describe compounds where alkoxy chains (R1 and R2) can be tuned from C6 to C16, allowing for precise modulation of solubility and phase transition temperatures. This tunability is essential for optimizing the processing conditions required for thin-film deposition in display manufacturing. Furthermore, the extended pi-conjugation achieved through the fusion of aryl groups significantly strengthens intermolecular pi-pi interactions, which is the fundamental driver for the high charge carrier mobility observed in these materials. The patent data indicates that these compounds can be synthesized with high purity, a prerequisite for minimizing trap states in electronic devices that lead to efficiency roll-off. As a reliable electronic chemical supplier, understanding these structure-property relationships is key to selecting the right intermediates for high-end optoelectronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for extended pi-conjugated systems often rely on harsh thermal conditions or multiple steps involving unstable intermediates, which can lead to significant batch-to-batch variability and lower overall yields. In the context of liquid crystal materials, achieving the precise planar geometry required for columnar stacking is frequently hampered by steric hindrance during the coupling of bulky aromatic units. Conventional methods may also struggle with controlling the regioselectivity of substitution on the carbazole or triphenylene cores, resulting in complex mixtures of isomers that are difficult and costly to separate. Additionally, many existing processes utilize expensive transition metal catalysts in stoichiometric amounts or require rigorous anhydrous conditions that are challenging to maintain on a large industrial scale. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks for procurement managers seeking cost reduction in electronic chemical manufacturing.
The Novel Approach
The methodology outlined in CN111825598A overcomes these hurdles by employing a streamlined sequence of Suzuki coupling followed by mild oxidative cyclization. This approach utilizes readily available starting materials such as tetraalkoxybiphenyls and carbazole boronic esters, which are coupled efficiently using palladium catalysis under relatively moderate temperatures of 70°C. The subsequent ring-closing step uses ferric chloride (FeCl3) as an oxidant at room temperature, eliminating the need for energy-intensive heating and reducing the risk of thermal degradation of the sensitive organic framework. This novel pathway not only shortens the synthetic route but also enhances the overall atom economy by minimizing side reactions. For supply chain heads, this translates to a more robust and predictable production schedule, as the reliance on exotic reagents is minimized and the reaction conditions are forgiving enough to ensure consistent quality across large batches.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The core innovation of this technology lies in the intramolecular oxidative cyclization step, which transforms the flexible precursor into a rigid, planar discotic core. As illustrated in the reaction scheme below, the treatment of the carbazole-triphenylene precursor with FeCl3 in a solvent mixture of dichloromethane and nitromethane facilitates the removal of hydrogen atoms and the formation of new carbon-carbon bonds between the aromatic rings. This Scholl-type reaction mechanism proceeds via a radical cation intermediate generated by the single-electron oxidation capability of the iron(III) species. The presence of electron-donating alkoxy groups on the periphery stabilizes these cationic intermediates, directing the cyclization to occur specifically at the desired positions to form the fused bisphenanthro-carbazole system. This mechanistic pathway is crucial for establishing the extended conjugation length that defines the electronic properties of the final material.
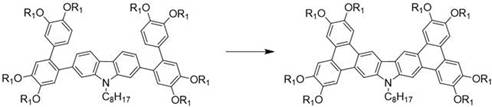
From an impurity control perspective, the choice of reaction conditions plays a pivotal role in ensuring the high purity required for electronic grade materials. The patent specifies the use of methanol to quench the reaction, which effectively deactivates the remaining Lewis acid and prevents over-oxidation or polymerization of the product. Subsequent purification via silica gel column chromatography using a toluene and petroleum ether eluent system allows for the precise separation of the target yellow-green solid from any unreacted starting materials or partially cyclized byproducts. The recrystallization step using ethanol and ethyl acetate further refines the product, removing trace metal residues and organic impurities that could act as charge traps in the final device. This rigorous purification protocol ensures that the resulting high-purity OLED material meets the stringent specifications demanded by top-tier display manufacturers, thereby reducing the risk of device failure due to material defects.
How to Synthesize Carbazole Discotic Liquid Crystals Efficiently
The synthesis of these advanced functional materials follows a logical progression of bond-forming reactions designed to maximize yield and structural integrity. The process begins with the preparation of halogenated intermediates, followed by palladium-catalyzed cross-coupling to assemble the molecular skeleton, and concludes with the oxidative fusing of the rings. Each step is optimized for scalability, utilizing standard laboratory equipment and reagents that are commercially available in bulk quantities. The detailed standardized synthesis steps provided in the patent serve as a robust foundation for process engineers looking to transfer this technology from the bench to the pilot plant. By adhering to the specified molar ratios and reaction times, manufacturers can achieve reproducible results that align with the high-quality benchmarks set forth in the intellectual property.
- Perform bromination of tetraalkoxybiphenyl with Br2 in chloroform at room temperature to generate the brominated intermediate.
- Execute Suzuki coupling between the brominated intermediate and carbazole boronic ester using Pd(PPh3)4 catalyst at 70°C.
- Conduct intramolecular oxidative cyclization using FeCl3 in dichloromethane/nitromethane to form the final fused ring system.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthesis route offers substantial strategic benefits for organizations focused on optimizing their supply chain and reducing overall manufacturing expenditures. The elimination of complex multi-step sequences in favor of a direct coupling and cyclization strategy drastically simplifies the production workflow, leading to significant operational efficiencies. By utilizing common solvents like dichloromethane and THF, the process avoids the need for specialized or hazardous solvent handling infrastructure, which lowers capital investment requirements for new production lines. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to a lower carbon footprint and aligning with modern sustainability goals. These factors combine to create a cost-effective manufacturing model that enhances competitiveness in the global market for specialty electronic chemicals.
- Cost Reduction in Manufacturing: The use of catalytic amounts of palladium and stoichiometric iron chloride, both of which are relatively inexpensive compared to other noble metal catalysts, drives down the raw material costs significantly. The high yields reported in the patent examples, such as the 97% yield in the bromination step and 80% in the coupling step, minimize waste and maximize the output per batch, directly improving the cost per kilogram of the final product. Additionally, the straightforward workup procedures involving simple filtration and extraction reduce the labor hours and solvent volumes required for purification, further contributing to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: The starting materials, including various alkoxy-substituted biphenyls and carbazole derivatives, are commodity chemicals with established global supply networks, ensuring consistent availability and mitigating the risk of shortages. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, which enhances the reliability of supply and reduces the frequency of batch failures. This stability allows procurement managers to negotiate better long-term contracts and maintain leaner inventory levels without compromising on production schedules, ensuring a steady flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as demonstrated by the use of standard reactor sizes and conditions that can be easily translated from 100 ml lab scales to multi-ton industrial vessels. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using conventional wastewater treatment protocols, ensuring compliance with strict environmental regulations. The absence of highly toxic reagents or extreme pressure conditions simplifies the safety management protocols, making it easier to obtain the necessary permits for commercial scale-up of complex electronic chemicals and facilitating faster time-to-market for new product launches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole-based liquid crystal materials. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is critical for making informed decisions about material selection and process integration.
Q: What are the key advantages of the FeCl3-catalyzed cyclization method?
A: The FeCl3-catalyzed oxidative cyclization allows for the formation of large pi-conjugated systems under mild room temperature conditions, significantly reducing energy consumption compared to high-temperature thermal cyclization methods while maintaining high structural rigidity.
Q: How does the alkyl chain length affect the liquid crystal properties?
A: The patent specifies alkyl chains (R1) ranging from C6 to C16, which provide the necessary flexibility for self-assembly into hexagonal columnar phases while ensuring solubility in common organic solvents for processing.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the route utilizes standard reagents like Pd(PPh3)4 and FeCl3 with straightforward workup procedures involving extraction and column chromatography, making it highly adaptable for commercial scale-up of complex electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of electronic chemical manufacturing, including strict moisture control and inert atmosphere operations necessary for palladium-catalyzed reactions. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of carbazole discotic liquid crystals meets the exacting standards required for high-performance OLED applications. Our commitment to quality assurance guarantees that the materials you receive will perform consistently in your final devices, minimizing the risk of costly downstream failures.
We invite you to collaborate with our expert team to explore how this patented technology can be integrated into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your R&D and production goals with reliable, high-quality intermediates.
