Advanced Copper-Catalyzed Amide N-Demethylation for Scalable Pharmaceutical Intermediate Production
Advanced Copper-Catalyzed Amide N-Demethylation for Scalable Pharmaceutical Intermediate Production
The synthesis of secondary amides via the removal of N-methyl groups represents a persistent challenge in modern organic synthesis, particularly within the context of generating high-value pharmaceutical intermediates. Traditional methods often struggle with the inherent stability of the amide bond, where resonance between the carbonyl group and the nitrogen lone pair reduces nucleophilicity and hinders oxidative cleavage. Addressing this critical gap, patent CN111253275A discloses a groundbreaking methodology utilizing N-fluorobisbenzenesulfonamide (NFSI) in conjunction with divalent copper salts to achieve efficient N-demethylation. This technical breakthrough allows for the conversion of tertiary amides into secondary amides under remarkably mild thermal conditions ranging from 40 to 120°C. For R&D directors and process chemists, this innovation offers a viable pathway to access deprotected amide building blocks essential for drugs like BACE-1 inhibitors and PYK2 kinase inhibitors without resorting to harsh degradative conditions.
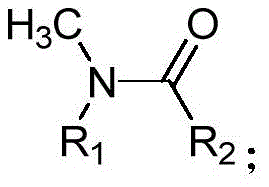
The structural transformation is generalized where R1 and R2 can be independently selected from aryl, alkyl, or alkenyl groups, providing significant flexibility for medicinal chemistry applications. The ability to selectively remove the methyl group while preserving the integrity of the carbonyl functionality and other sensitive substituents is paramount for maintaining the purity profiles required in high-purity pharmaceutical intermediates. This patent data underscores a shift towards more selective oxidative protocols that minimize side reactions, thereby simplifying downstream purification processes and enhancing overall process mass intensity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, N-demethylation strategies have been predominantly focused on amines rather than amides, largely due to the electronic differences between these functional groups. Conventional approaches for amines often rely on the formation of quaternary ammonium salts or N-oxides, followed by elimination or reduction steps. However, applying these same logic to amides frequently results in failure because the amide nitrogen is a poor electron donor. Furthermore, existing literature describes photochemical or biochemical methods that, while effective in specific biological contexts, lack the robustness and scalability required for industrial fine chemical manufacturing. Many traditional chemical oxidants, such as hydrogen peroxide, have been experimentally proven ineffective for amide N-demethylation, leading to zero conversion or extensive decomposition of the starting material. This limitation forces process developers to utilize multi-step protection-deprotection sequences that drastically increase cost and waste generation.
The Novel Approach
The methodology outlined in the provided patent data introduces a paradigm shift by leveraging the unique oxidative properties of NFSI activated by a copper catalyst. Unlike previous attempts that failed with standard oxidants, this system specifically targets the N-methyl bond on the amide nitrogen. The reaction operates effectively in a mixed solvent system of methanol and acetonitrile, which facilitates better dispersion of reactants compared to non-polar solvents like toluene or dichloromethane that yield negligible results. By heating the mixture to moderate temperatures, typically around 80°C, the system achieves conversion rates that are commercially viable. This approach eliminates the need for exotic reagents or extreme conditions, positioning it as a superior alternative for cost reduction in API manufacturing where operational simplicity translates directly to margin improvement.
Mechanistic Insights into Copper-Catalyzed Oxidative Demethylation
The core of this transformation lies in the synergistic interaction between the divalent copper salt and the NFSI oxidant. While the exact mechanistic cycle may involve radical intermediates or high-valent copper species, the empirical data confirms that the copper center is essential for activating the oxidant towards the sterically hindered amide nitrogen. The use of copper acetylacetonate (Cu(acac)2) has been identified as particularly effective, likely due to its solubility profile and ligand exchange kinetics in the methanol/acetonitrile medium. The reaction atmosphere also plays a subtle but important role; while the process can proceed in air, conducting the reaction under an inert nitrogen atmosphere consistently yields superior results, suggesting that uncontrolled oxidation of the solvent or substrate by atmospheric oxygen might compete with the desired catalytic cycle. This sensitivity highlights the importance of precise process control in a commercial setting.
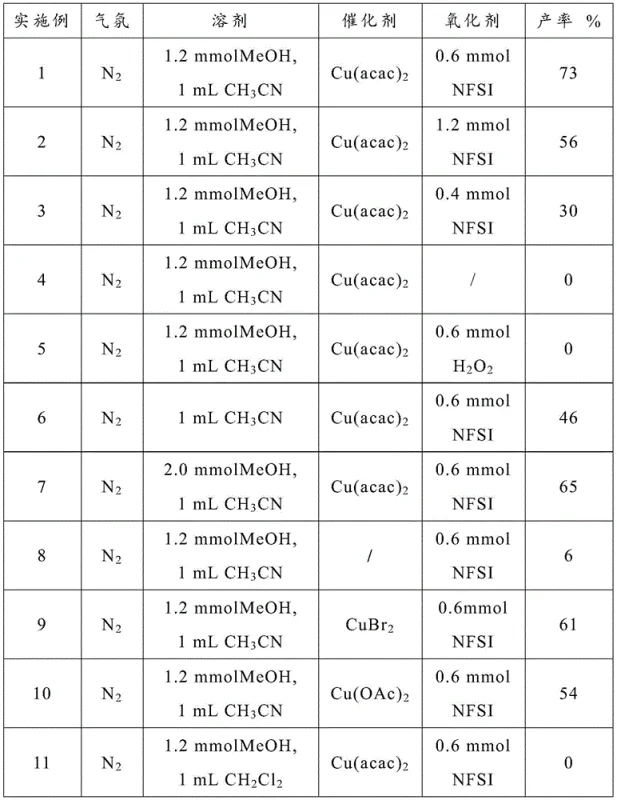
Optimization studies reveal that the stoichiometry of the oxidant is a critical parameter. A molar ratio of substrate to NFSI between 1:1 and 1:3 is effective, with a ratio of 1:1.5 demonstrating optimal performance. Deviating from this balance, such as using insufficient oxidant or substituting NFSI with hydrogen peroxide, leads to a complete collapse of the reaction yield. Furthermore, the solvent composition is not merely a medium for dissolution but an active participant in the reaction efficiency. The data explicitly shows that replacing acetonitrile with dichloromethane or toluene results in 0% yield, emphasizing that the polarity and coordinating ability of acetonitrile are indispensable for stabilizing the transition states involved in the C-N bond cleavage. This level of mechanistic understanding allows for rigorous impurity control, ensuring that the final pharmaceutical intermediates meet stringent regulatory specifications.
How to Synthesize N-Demethylated Amides Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to ensure reproducibility and safety. The process begins with the careful charging of reagents into a dry pressure vessel equipped with agitation, followed by the addition of the specific solvent mixture. The thermal profile must be maintained within the 40-120°C window, with 80°C serving as a robust baseline for most substrates. Reaction monitoring via thin-layer chromatography (TLC) is recommended to determine the precise endpoint, which typically occurs between 6 to 36 hours depending on the steric bulk of the R1 and R2 groups. Following the reaction, a standard workup involving ethyl acetate extraction and silica gel column chromatography using petroleum ether and ethyl acetate mixtures allows for the isolation of the pure secondary amide. Detailed standardized synthesis steps are provided in the guide below.
- Charge a dry pressure vessel with the N-methyl amide substrate, N-fluorobisbenzenesulfonamide (NFSI), and a divalent copper salt catalyst such as copper acetylacetonate.
- Add a solvent mixture comprising methanol and acetonitrile, ensuring a molar ratio of amide to methanol between 3: 1 and 5:1 for optimal dispersion.
- Heat the reaction mixture to a temperature range of 40-120°C under an inert nitrogen atmosphere for 6 to 36 hours, followed by silica gel column purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this copper-catalyzed N-demethylation protocol offers substantial strategic benefits over legacy synthetic routes. The primary advantage lies in the accessibility and cost profile of the reagents. Copper salts such as copper acetylacetonate, copper bromide, and copper acetate are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency often associated with precious metal catalysts like palladium or rhodium. Similarly, NFSI, while a specialized reagent, is a stable solid that is easier to handle and transport than gaseous oxidants or unstable liquid peroxides. This availability ensures enhanced supply chain reliability, allowing procurement managers to secure raw materials with shorter lead times and more predictable pricing structures.
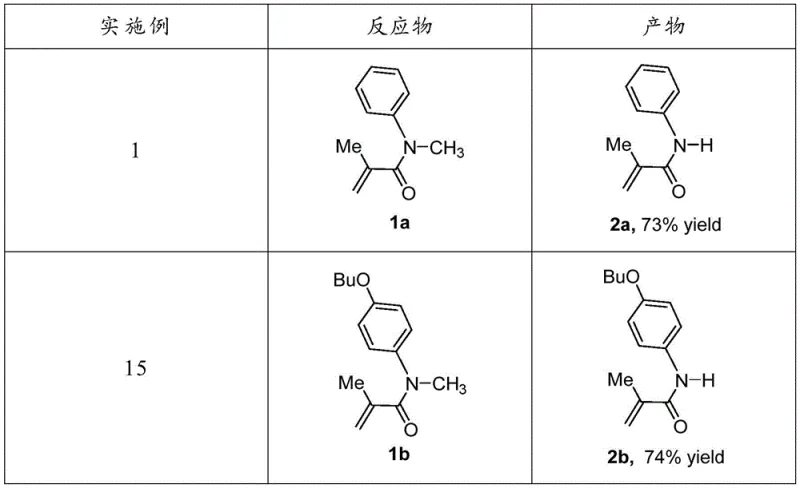
The economic impact extends to the manufacturing floor through simplified processing requirements. The reaction tolerates a wide range of functional groups, as evidenced by the successful conversion of substrates containing alkenes, cyclopropanes, and bulky tert-butyl groups without the need for additional protecting groups. This chemoselectivity reduces the total number of synthetic steps required to reach the target intermediate, directly correlating to cost reduction in pharmaceutical intermediates manufacturing. Fewer steps mean less solvent consumption, lower energy usage for heating and cooling, and reduced waste disposal costs. Moreover, the ability to run the reaction in air, albeit with slightly lower yields than under nitrogen, provides a fallback option that reduces the complexity of reactor setup and inert gas consumption for less sensitive batches. These factors collectively contribute to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts in favor of abundant copper salts significantly lowers the raw material cost per kilogram of product. Additionally, the high selectivity of the NFSI oxidation minimizes the formation of byproducts, reducing the burden on purification units and increasing the overall yield of the valuable secondary amide. This efficiency translates to substantial cost savings when scaling from laboratory grams to commercial tonnage.
- Enhanced Supply Chain Reliability: By utilizing reagents that are commercially available off-the-shelf, manufacturers can avoid the long lead times associated with custom-synthesized catalysts. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This stability is crucial for maintaining continuous production schedules in a fast-paced pharmaceutical environment.
- Scalability and Environmental Compliance: The mild reaction temperatures and the use of standard organic solvents like acetonitrile and methanol make this process highly scalable in existing stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. Furthermore, the avoidance of toxic heavy metals simplifies the environmental compliance landscape, reducing the cost and complexity of wastewater treatment and residue management.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this N-demethylation technology. These answers are derived directly from the experimental data and embodiments described in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for process engineers evaluating the feasibility of integrating this method into their current production workflows.
Q: Why is N-fluorobisbenzenesulfonamide (NFSI) critical for this demethylation?
A: Unlike common oxidants like hydrogen peroxide which fail to achieve N-demethylation on amides due to the conjugation stability of the amide bond, NFSI acts as a specific electrophilic fluorinating agent that facilitates the cleavage of the N-C bond under copper catalysis.
Q: What represents the optimal solvent system for this transformation?
A: Experimental data indicates that a mixture of methanol and acetonitrile provides superior yields compared to single solvents or non-polar options like toluene and dichloromethane, which result in negligible conversion.
Q: Does this method tolerate diverse functional groups on the amide substrate?
A: Yes, the protocol demonstrates broad substrate scope, successfully processing aryl, alkyl, and alkenyl groups at both the nitrogen and carbonyl positions, including sensitive moieties like cyclopropane and tert-butyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Demethylated Amides Supplier
As the demand for complex amide intermediates continues to grow in the development of next-generation therapeutics, having a manufacturing partner with deep technical expertise is vital. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-demethylated amide meets the highest industry standards. We understand the critical nature of these intermediates in the synthesis of active pharmaceutical ingredients and are committed to delivering quality that supports your regulatory filings.
We invite you to engage with our technical procurement team to discuss how this advanced copper-catalyzed technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how optimizing your synthetic route with our methods can improve your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver reliable solutions for your complex chemical challenges.
