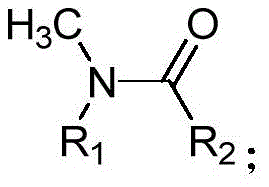
Advanced Copper-Catalyzed Amide N-Demethylation for Scalable Pharmaceutical Manufacturing
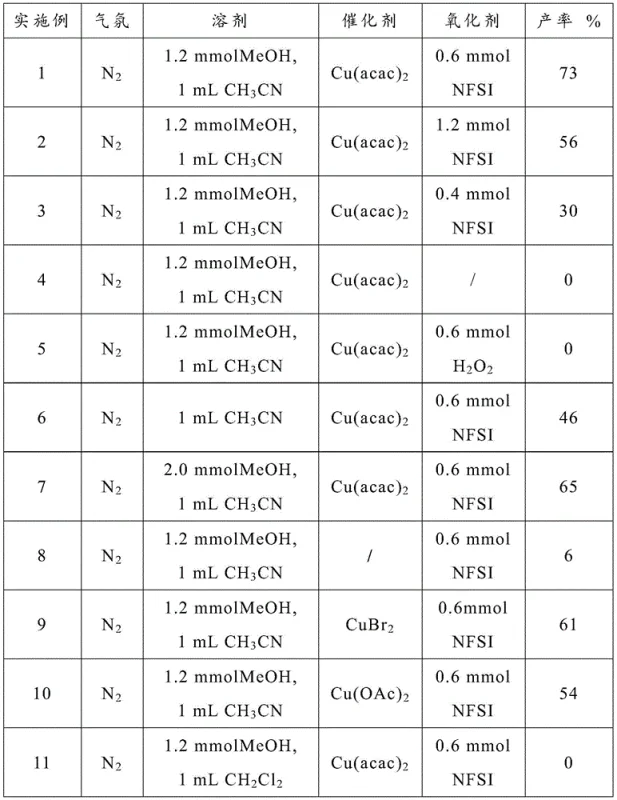
The landscape of organic synthesis constantly evolves to meet the rigorous demands of the pharmaceutical industry, particularly in the modification of complex molecular scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN111253275A, which discloses a novel and efficient method for the N-demethylation of amides. This transformation is notoriously difficult due to the resonance stabilization of the amide bond, which renders the nitrogen lone pair less nucleophilic and resistant to traditional dealkylation strategies. The patented technology utilizes a synergistic catalytic system involving N-fluorobisbenzenesulfonamide (NFSI) and a divalent copper salt, specifically copper acetylacetonate, to achieve this conversion under remarkably mild conditions. By operating at temperatures ranging from 40°C to 120°C, this method circumvents the harsh acidic or basic environments often required by legacy techniques, thereby preserving sensitive functional groups elsewhere in the molecule. This innovation represents a pivotal advancement for manufacturers seeking reliable pharmaceutical intermediates supplier partnerships that prioritize both chemical integrity and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the removal of N-methyl groups from amides has been a persistent bottleneck in medicinal chemistry and process development. Traditional approaches often rely on the formation of quaternary ammonium salts followed by elimination, or the generation of N-oxides, both of which typically necessitate extreme reaction conditions such as high temperatures, strong acids, or potent bases. These aggressive environments frequently lead to the degradation of the core molecular structure, resulting in complex impurity profiles that are costly and time-consuming to remove. Furthermore, many existing methods are limited in their substrate scope, failing to accommodate diverse aryl, alkyl, or alkenyl substituents without significant loss of yield. The reliance on stoichiometric amounts of hazardous reagents also poses substantial environmental and safety challenges, complicating waste disposal and increasing the overall carbon footprint of the manufacturing process. Consequently, there has been an urgent industry demand for a more selective and温和 (mild) methodology that can deliver high-purity products with minimal downstream processing.
The Novel Approach
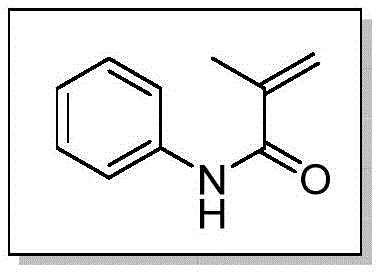
The methodology presented in CN111253275A offers a transformative solution by leveraging a copper-catalyzed oxidative pathway that operates efficiently under much gentler parameters. Unlike previous attempts that struggled with amide stability, this system utilizes NFSI as a specialized oxidant in conjunction with a catalytic amount of Cu(acac)2 to selectively cleave the N-methyl bond. Experimental data within the patent demonstrates exceptional versatility, successfully demethylating a wide array of substrates including N-methyl-N-phenylmethacrylamide and various benzamides with yields reaching up to 94% in optimized cases. The process tolerates a broad range of functional groups, allowing R&D teams to apply this technique late-stage in synthetic sequences without fear of compromising other sensitive moieties. Moreover, the reaction exhibits robustness across different atmospheric conditions; while nitrogen provides optimal results, the system remains functional even under air, significantly reducing the engineering controls required for scale-up. This adaptability makes it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing, where simplicity and reliability are paramount.
Mechanistic Insights into Cu-Catalyzed Oxidative Demethylation
The efficacy of this N-demethylation protocol stems from the unique interplay between the divalent copper catalyst and the NFSI oxidant. Mechanistically, the copper center likely facilitates the activation of the NFSI, generating a reactive nitrogen-centered radical species capable of abstracting a hydrogen atom from the N-methyl group. This initial abstraction creates an aminyl radical intermediate, which subsequently undergoes further oxidation and hydrolysis steps mediated by the methanol co-solvent to release formaldehyde and regenerate the secondary amide. The specificity of NFSI is crucial here; comparative experiments revealed that common oxidants like hydrogen peroxide were completely ineffective, yielding 0% conversion under identical conditions. This suggests that the fluorosulfonyl moiety of NFSI plays an indispensable role in modulating the redox potential of the system to match the high oxidation barrier of the amide nitrogen. Understanding this mechanism allows chemists to fine-tune reaction parameters, ensuring that the catalytic cycle proceeds with maximum turnover frequency while minimizing side reactions.
From an impurity control perspective, the mild nature of this catalytic system inherently limits the formation of degradation byproducts. Harsh thermal or acidic conditions often promote hydrolysis of the amide bond itself or polymerization of unsaturated side chains, but the 40-120°C window utilized here preserves structural integrity. The use of acetonitrile and methanol as the solvent system further enhances selectivity, as trials with dichloromethane or toluene resulted in no reaction, indicating that the polar protic/aprotic mixture is essential for stabilizing the transition states involved in the demethylation pathway. For quality assurance teams, this means a cleaner crude reaction profile, which simplifies the purification workflow. The ability to achieve high conversions with minimal byproduct generation directly translates to higher overall process efficiency and reduced solvent consumption during the isolation of the final high-purity pharmaceutical intermediates.
How to Synthesize N-Demethylated Amides Efficiently
Implementing this synthesis route requires precise adherence to the optimized conditions outlined in the patent to ensure reproducibility and high yield. The standard protocol involves charging a dry pressure vessel with the N-methyl amide substrate, NFSI, and copper acetylacetonate, followed by the addition of a specific ratio of methanol and acetonitrile. The mixture is then heated, typically to 80°C, and stirred for a period ranging from 6 to 36 hours depending on the steric bulk of the substrate. Reaction progress is monitored via TLC, and upon completion, the product is isolated through standard workup procedures involving concentration and silica gel chromatography. This streamlined workflow eliminates the need for exotic equipment or dangerous reagents, making it highly accessible for both laboratory discovery and pilot plant operations. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in immediate adoption.
- Combine the N-methyl amide substrate with N-fluorobisbenzenesulfonamide (NFSI) and copper acetylacetonate (Cu(acac)2) in a mixture of methanol and acetonitrile.
- Heat the reaction mixture to a temperature between 40°C and 120°C, preferably 80°C, under a nitrogen atmosphere for 6 to 36 hours.
- Upon completion, concentrate the mixture and purify the resulting demethylated amide using silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed N-demethylation technology offers tangible strategic benefits beyond mere chemical novelty. The shift away from precious metal catalysts or stoichiometric hazardous oxidants towards a base-metal copper system significantly lowers the raw material cost baseline. Copper salts are abundant, inexpensive, and widely available in the global market, insulating the supply chain from the volatility often seen with rare earth or noble metal prices. Furthermore, the operational simplicity of the reaction—requiring only standard heating and stirring without high-pressure hydrogenation or cryogenic cooling—reduces energy consumption and equipment wear. This simplicity facilitates faster batch turnover times, enhancing the overall throughput of the manufacturing facility and ensuring more reliable delivery schedules for downstream clients. The robustness of the method also minimizes the risk of batch failures due to sensitive reaction parameters, thereby securing supply continuity for critical drug substances.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the substitution of expensive reagents with cost-effective alternatives. By utilizing copper acetylacetonate instead of palladium or rhodium-based catalysts, the direct material cost per kilogram of product is drastically reduced. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the volume of solvents and stationary phases required for purification. This downstream efficiency compounds the savings, as less waste is generated and less time is spent on extensive recrystallization or chromatographic separation. The elimination of harsh acidic or basic workups also lowers the cost associated with neutralization agents and corrosion-resistant reactor maintenance, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the key reagents, NFSI and copper salts, which are commodity chemicals produced by multiple vendors globally. This multi-sourcing capability prevents bottlenecks that often occur when relying on single-source specialty reagents. Moreover, the reaction's tolerance to air atmosphere, as demonstrated in specific embodiments, reduces the dependency on high-purity nitrogen or argon supplies, simplifying logistics for facilities with limited gas infrastructure. The mild reaction conditions also enhance safety profiles, lowering insurance premiums and regulatory compliance burdens associated with handling hazardous materials. These factors collectively create a more stable and predictable supply environment, ensuring that production timelines are met consistently even during periods of market fluctuation.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is straightforward due to the absence of exothermic hazards or gas evolution that typically complicate scale-up. The liquid-phase homogeneous catalysis ensures good heat transfer and mixing, allowing for safe operation in large-scale reactors. From an environmental standpoint, the method aligns with green chemistry principles by avoiding toxic heavy metals and reducing solvent waste through higher yields. The use of methanol and acetonitrile, while requiring proper recovery systems, is standard in the industry and easier to manage than chlorinated solvents which were found ineffective in this specific chemistry. This environmental compatibility simplifies the permitting process for new production lines and supports corporate sustainability goals, making the technology attractive for long-term investment in commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this N-demethylation technology. These answers are derived directly from the experimental data and embodiments described in the patent documentation, providing clarity for R&D and process engineering teams evaluating this route. Understanding these nuances is critical for successful technology transfer and risk mitigation during the early stages of project planning. We encourage stakeholders to review these details to fully appreciate the operational flexibility and limitations of the system.
Q: Can this N-demethylation reaction be performed under air atmosphere?
A: Yes, while the patent indicates that an inert atmosphere like nitrogen yields better results, Example 14 demonstrates that the reaction can proceed under air atmosphere, offering flexibility for large-scale operations where strict inert conditions increase costs.
Q: Which solvents are compatible with this copper-catalyzed demethylation process?
A: The process specifically requires a solvent system containing methanol and/or acetonitrile. Experiments showed that using dichloromethane or toluene resulted in zero or extremely low yields, making the MeOH/MeCN mixture critical for success.
Q: Is hydrogen peroxide a viable alternative oxidant to NFSI in this method?
A: No, the patent explicitly states that substituting N-fluorobisbenzenesulfonamide (NFSI) with hydrogen peroxide (H2O2) fails to achieve N-demethylation, highlighting the unique oxidative capability of NFSI in this catalytic cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Demethylated Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the copper-catalyzed N-demethylation pathway described in CN111253275A and is prepared to integrate this technology into our custom synthesis portfolio. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-demethylated amide delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this method can optimize your budget. We encourage you to reach out today to discuss your requirements, obtain specific COA data for similar structures, and receive comprehensive route feasibility assessments. Let us help you accelerate your drug development timeline with reliable, high-quality intermediates produced through cutting-edge synthetic innovation.
