Advanced Manufacturing of 2-Butyl-BBIT: A Cost-Effective Nitrile-Based Route for Global Supply Chains
The chemical industry is constantly seeking more efficient pathways for synthesizing high-value heterocyclic compounds, particularly those used as biocides and preservatives. Patent CN103145638A introduces a groundbreaking preparation method for 2-butyl-1,2-benzoisothiazolin-3-one (BBIT), a critical intermediate widely utilized in plastics, metalworking fluids, and coatings due to its potent antifungal properties. This innovation shifts the synthetic paradigm away from traditional disulfide precursors, utilizing o-methylthio benzonitrile as a cost-effective starting material. By employing a streamlined sequence of esterification, amidation, and cyclization, this technology addresses long-standing challenges regarding raw material availability and environmental compliance. For global procurement teams and R&D directors, understanding this novel route is essential for securing a stable supply of high-purity BBIT while minimizing production costs and ecological footprints in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of BBIT has relied heavily on 2,2'-dithiodibenzoic acid as the primary feedstock, a route fraught with significant economic and technical inefficiencies. Traditional methods often involve the conversion of this disulfide into acyl chlorides followed by harsh halogen cracking to break the sulfur-sulfur bond, a process that requires aggressive reagents and generates substantial hazardous waste. Alternatively, some existing protocols attempt to synthesize BBIT by alkylating the parent BIT molecule with n-butyl bromide; however, this approach is fundamentally flawed due to poor selectivity. As illustrated in the reaction scheme below, this alkylation inevitably leads to the formation of O-alkylated by-products alongside the desired N-alkylated target, resulting in low yields and extremely difficult purification processes that drive up operational expenses.
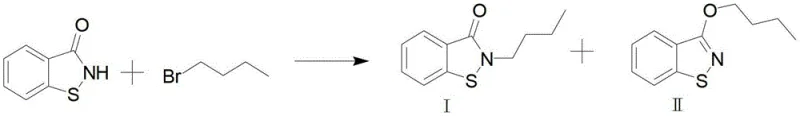
The Novel Approach
In stark contrast to these legacy methods, the patented technology leverages o-methylthio benzonitrile, a commercially abundant and inexpensive nitrile derivative, to construct the benzisothiazolinone scaffold de novo. This innovative pathway bypasses the need for disulfide bond cleavage entirely, instead relying on a controlled cyclization of an amide intermediate. The process operates under significantly milder conditions, typically ranging from 60°C to 95°C, and utilizes standard chlorinating agents like sulfuryl chloride or phosphorus trichloride to effect ring closure. By avoiding the use of expensive oxidants and complex separation steps associated with by-product removal, this method achieves total yields exceeding 90%, representing a substantial leap forward in process efficiency and economic viability for large-scale manufacturers.
Mechanistic Insights into Acid-Catalyzed Esterification and Cyclization
The core of this synthetic strategy lies in the sequential transformation of the nitrile group, beginning with an acid-catalyzed esterification in an alcohol solvent such as methanol or ethanol. In this initial phase, hydrogen chloride gas or sulfuric acid acts as the catalyst to convert the nitrile functionality into an imino ether ester intermediate, a reaction that proceeds smoothly at moderate temperatures between 50°C and 90°C. Following neutralization, the introduction of butylamine facilitates a nucleophilic attack on the ester carbonyl, driving the amidation reaction to form the critical 2-(methylthio)-N-butylbenzamide intermediate. This step is crucial as it installs the necessary butyl chain on the nitrogen atom prior to ring closure, ensuring the correct regiochemistry for the final product without the risk of O-alkylation side reactions.
The final and most critical stage involves the oxidative cyclization of the amide intermediate using a chlorinating agent in a biphasic system of organic solvent and water. Agents such as sulfuryl chloride (SO2Cl2) or phosphorus pentachloride react with the thioether sulfur to generate a reactive sulfenyl chloride species in situ. This electrophilic sulfur center then undergoes an intramolecular nucleophilic attack by the adjacent amide nitrogen, displacing the chloride and closing the five-membered isothiazolinone ring. The use of a biphasic workup allows for the efficient removal of inorganic salts and acidic by-products through simple washing with sodium carbonate and sodium thiosulfate solutions, ensuring the final isolation of BBIT as a high-purity yellow oil with minimal downstream processing requirements.
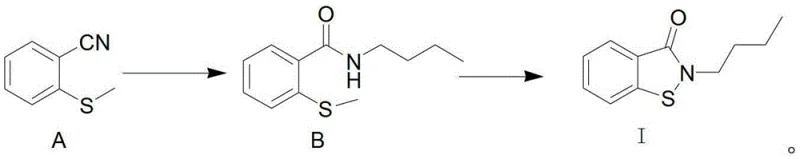
How to Synthesize 2-Butyl-1,2-benzoisothiazolin-3-one Efficiently
The implementation of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the dissolution of o-methylthio benzonitrile in an alcohol solvent, followed by saturation with hydrogen chloride gas to initiate esterification. Once the ester is formed, the system is neutralized, and butylamine is added to drive the amidation to completion. The resulting amide is then subjected to cyclization conditions using a selected chlorinating agent in a solvent like toluene. For detailed operational parameters, stoichiometry, and specific workup procedures validated by experimental data, please refer to the standardized synthesis guide provided below.
- React o-methylthio benzonitrile with alcohol under acid catalysis to form the corresponding ester intermediate.
- Perform amidation by reacting the ester with butylamine to obtain the amide compound.
- Execute ring-closure reaction using a chlorinating agent in an organic solvent to yield the final BBIT product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nitrile-based synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. By shifting the feedstock dependency from specialized disulfide acids to commodity nitriles, manufacturers can significantly decouple their production costs from the volatile pricing of niche sulfur chemicals. Furthermore, the elimination of complex purification steps required to remove O-alkylated impurities drastically reduces solvent consumption and energy usage during distillation. This streamlined workflow not only accelerates batch turnover times but also simplifies the regulatory burden associated with waste disposal, positioning companies to meet increasingly stringent environmental standards without compromising output volume.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the substitution of high-cost starting materials with readily available o-methylthio benzonitrile. Traditional routes relying on 2,2'-dithiodibenzoic acid incur substantial raw material expenses, whereas the nitrile precursor is produced on a massive industrial scale for other applications, ensuring a stable and low-cost supply. Additionally, the high selectivity of the cyclization step means that valuable reagents are not wasted on forming inseparable by-products, leading to a drastic improvement in overall atom economy and a significant reduction in the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source or geographically concentrated suppliers for exotic intermediates. This new method mitigates such risks by utilizing a raw material portfolio that is globally sourced and chemically robust. The reaction conditions are forgiving and do not require specialized high-pressure equipment or cryogenic cooling, allowing for production flexibility across diverse manufacturing sites. This adaptability ensures that production schedules remain uninterrupted even during periods of logistical strain, providing a reliable source of high-purity agrochemical and pharmaceutical intermediates for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a major advancement over legacy technologies that utilize heavy metal oxidants or generate toxic sulfur waste streams. The use of standard chlorinating agents and the ability to recycle solvents like toluene or chlorobenzene aligns perfectly with green chemistry principles. The absence of difficult-to-treat by-products simplifies wastewater treatment protocols, reducing the capital expenditure required for effluent management. Consequently, scaling this process from pilot plant to multi-ton commercial production is straightforward, enabling manufacturers to rapidly expand capacity to meet growing global demand for effective biocidal agents.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries regarding the patent specifications and process capabilities. These insights are derived directly from the experimental embodiments and comparative data presented in the intellectual property documentation, focusing on practical implementation details. Understanding these nuances is vital for assessing the potential return on investment and the technical readiness level of this synthesis route for immediate industrial deployment.
Q: What are the primary advantages of the nitrile-based route over traditional disulfide methods?
A: The nitrile-based route eliminates the need for expensive and hard-to-source 2,2'-dithiodibenzoic acid. It avoids harsh halogen cracking of disulfide bonds and removes the necessity for heavy metal oxidants, resulting in a cleaner process with significantly reduced environmental impact and lower raw material costs.
Q: How does this method address the impurity issues found in BIT alkylation?
A: Traditional alkylation of BIT with n-butyl bromide often produces significant amounts of O-alkylated by-products which are difficult to separate. The patented cyclization method constructs the heterocyclic ring directly from the amide precursor, bypassing the alkylation step entirely and ensuring higher regioselectivity and purity of the N-butyl product.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials like o-methylthio benzonitrile and common solvents such as toluene or chlorobenzene. The reaction conditions are mild (typically 60-95°C) and do not require extreme pressures or temperatures, making it highly scalable for commercial manufacturing with total yields exceeding 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Butyl-1,2-benzoisothiazolin-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic methodologies is key to maintaining competitiveness in the global fine chemicals market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of BBIT performs consistently in your final formulations, whether for industrial preservation or agricultural protection.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your specific application needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized supply chain can enhance your operational efficiency and reduce your total cost of ownership for this critical chemical intermediate.
