Scalable Metal-Free Production of 1,1-Dithio-1-Alkene Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN102249962A, which discloses a novel preparation method for 1,1-dithio-1-alkenes, a critical class of building blocks widely utilized in the synthesis of beta-lactam antibiotics, carbohydrate derivatives, and optical linear materials. Historically, the construction of the carbon-sulfur bonds in these molecules relied heavily on transition metal catalysis, presenting substantial bottlenecks in terms of cost, environmental impact, and downstream purification complexity. This patent introduces a groundbreaking organocatalytic approach that utilizes 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) as a promoter in dimethyl sulfoxide (DMSO), enabling the efficient conversion of readily available 1,1-dibromo-1-alkenes into valuable 1,1-dithio-1-alkenes under remarkably mild conditions. By operating at room temperature without any metal additives, this technology addresses the core pain points of modern pharmaceutical intermediate production, offering a pathway to high-purity compounds with drastically reduced operational overhead.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 1,1-dithio-1-alkenes was predominantly achieved through transition metal-catalyzed cross-coupling reactions, which imposed severe limitations on both economic efficiency and process safety. As illustrated in the prior art, traditional methods often employed nickel catalysts requiring harsh reaction temperatures of 120°C and extended reaction times up to 24 hours to achieve acceptable conversion.
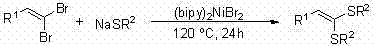
Furthermore, alternative protocols utilizing precious palladium catalysts, such as those involving Xantphos ligands or tetrakis(triphenylphosphine)palladium, necessitated elevated temperatures around 100°C and still suffered from inconsistent substrate scope or low transformation efficiencies ranging from 10% to 20%. These legacy processes not only incur high raw material costs due to the utilization of scarce noble metals but also generate significant waste streams containing heavy metal residues. For pharmaceutical manufacturers, the presence of residual nickel or palladium in the final active pharmaceutical ingredient (API) intermediate is a critical quality attribute that demands expensive and time-consuming purification steps, such as scavenger resin treatment or repeated recrystallization, to meet stringent regulatory limits typically measured in parts per million (ppm).
The Novel Approach
In stark contrast to these energy-intensive and metal-dependent methodologies, the invention disclosed in CN102249962A leverages a metal-free organocatalytic strategy that fundamentally reshapes the process economics. The core innovation lies in the use of DBU, a strong, non-nucleophilic organic base, which activates the thiol nucleophile and facilitates the substitution of bromine atoms on the 1,1-dibromo-1-alkene substrate at ambient temperature (25°C).

This novel approach eliminates the need for external heating sources, thereby slashing energy consumption and mitigating thermal safety risks associated with large-scale exothermic reactions. The reaction proceeds rapidly, often completing within minutes to hours, and tolerates a wide array of functional groups including halogens, nitro groups, and methoxy substituents on the aromatic rings. By removing the transition metal catalyst entirely, the process inherently produces a cleaner crude product profile, significantly simplifying the workup procedure to a standard aqueous extraction and column chromatography. This shift from heterogeneous or homogeneous metal catalysis to simple organocatalysis represents a paradigm shift for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios for sustainability and cost.
Mechanistic Insights into DBU-Promoted Nucleophilic Substitution
The mechanistic elegance of this transformation relies on the dual role of the reaction medium and the organic base in facilitating nucleophilic attack without metal coordination. In the proposed pathway, DBU acts as a proton sponge to deprotonate the thiol reactant (R-SH), generating a highly reactive thiolate anion in situ within the polar aprotic solvent DMSO. This thiolate species then attacks the electron-deficient vinyl carbon of the 1,1-dibromo-1-alkene, displacing the first bromide ion through a nucleophilic vinylic substitution mechanism. The presence of the second bromine atom further activates the double bond towards nucleophilic attack, allowing for the sequential introduction of the second sulfur moiety to form the geminal bis-thioether structure. Unlike palladium cycles that require oxidative addition and reductive elimination steps prone to catalyst deactivation or ligand dissociation, this ionic mechanism is robust and less sensitive to trace impurities in the starting materials.
From an impurity control perspective, the absence of metal catalysts is the single most significant advantage for R&D teams focusing on purity profiles. In metal-catalyzed routes, side reactions such as homocoupling of the thiol to form disulfides or debromination of the alkene can occur, often exacerbated by metal impurities. The DBU-mediated system minimizes these pathways by maintaining a controlled basic environment that favors the desired substitution. Furthermore, the lack of metal residues means that the final product does not require aggressive chelating treatments that might degrade sensitive functional groups. This results in a superior impurity spectrum where the primary contaminants are easily separable organic byproducts rather than intractable metal complexes, ensuring that the high-purity 1,1-dithio-1-alkene produced meets the rigorous specifications required for downstream drug synthesis.
How to Synthesize 1,1-Dithio-1-Alkene Efficiently
The practical implementation of this synthesis is straightforward and designed for immediate adoption in process chemistry laboratories. The protocol involves dissolving the 1,1-dibromo-1-alkene substrate and the chosen thiol in DMSO, followed by the addition of DBU at room temperature. The reaction mixture is stirred for a short duration, typically between 5 to 15 minutes as demonstrated in the patent examples, after which it is quenched with water. The product is then extracted into an organic phase, washed, dried, and purified. This simplicity allows for rapid screening of different substrate combinations to optimize yields for specific target molecules.
- Dissolve 1,1-dibromo-1-alkene and thiol reactant in dimethyl sulfoxide (DMSO) solvent within a round-bottomed flask.
- Add the organic base 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) to the mixture and stir at room temperature (25°C) for 5 to 15 minutes.
- Quench the reaction with distilled water, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free technology translates directly into tangible bottom-line improvements and risk mitigation. The elimination of precious metal catalysts such as palladium and nickel removes a major source of cost volatility, as the prices of these commodities are subject to significant market fluctuations. Moreover, the supply chain for high-purity organocatalysts like DBU is far more stable and diversified compared to specialized transition metal complexes, ensuring consistent availability of raw materials for continuous manufacturing operations.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated ligands drastically reduces the bill of materials for every batch produced. Beyond the direct savings on reagents, the most profound cost benefit arises from the simplification of the purification train. Without the need for specialized heavy metal scavengers, activated carbon treatments, or extensive recrystallization cycles to meet ppm-level metal specifications, the overall processing time and solvent consumption are significantly reduced. This streamlined workflow lowers the cost of goods sold (COGS) and improves the overall margin profile for high-purity pharmaceutical intermediates manufactured via this route.
- Enhanced Supply Chain Reliability: The mild reaction conditions (room temperature) and short reaction times enhance the agility of the manufacturing process. Facilities can achieve faster turnaround times between batches, increasing the effective capacity of existing reactor trains without the need for capital expenditure on new equipment. Additionally, the robustness of the reaction against varying substrate qualities reduces the risk of batch failures due to raw material inconsistencies. This reliability is crucial for maintaining uninterrupted supply lines to downstream API manufacturers, reducing the lead time for high-purity intermediates and strengthening the resilience of the global pharmaceutical supply chain.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, particularly when dealing with high temperatures and toxic metals. This novel method operates at ambient temperature, eliminating the energy burden of heating large reactors and reducing the carbon footprint of the manufacturing process. The absence of heavy metals also simplifies waste stream management, as the effluent does not require specialized treatment for metal recovery or disposal. This alignment with green chemistry principles facilitates easier regulatory approval and environmental compliance, making the commercial scale-up of complex olefin derivatives more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DBU-catalyzed synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational benefits for potential partners.
Q: What are the advantages of using DBU over transition metal catalysts for this synthesis?
A: Using DBU eliminates the need for expensive and toxic transition metals like palladium or nickel. This removes the requirement for costly heavy metal scavenging steps and rigorous ICP-MS testing, significantly simplifying the purification process and ensuring higher purity suitable for pharmaceutical applications.
Q: Can this room-temperature method be scaled for industrial production?
A: Yes, the mild reaction conditions (25°C) and short reaction times make this process highly scalable. Unlike traditional methods requiring high temperatures (120°C) and long durations, this exothermic-friendly profile reduces energy consumption and thermal safety risks during commercial scale-up.
Q: What types of substrates are compatible with this 1,1-dithio-1-alkene synthesis?
A: The method demonstrates broad substrate tolerance, successfully reacting various substituted 1,1-dibromo-1-alkenes (including those with electron-withdrawing groups like nitro and halogens) with diverse thiols such as thiophenol and benzyl mercaptan to yield high-purity products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Dithio-1-Alkene Supplier
The technological potential of this metal-free synthesis route is immense, offering a clear path to higher quality and lower cost for critical chemical building blocks. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the DBU-catalyzed method are seamlessly translated from the laboratory bench to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,1-dithio-1-alkene delivered meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking organizations to collaborate with us to leverage this advanced chemistry for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how this greener, more efficient synthesis can optimize your manufacturing costs and secure your supply of high-value intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
