Advanced Metal-Free Synthesis of 1,1-Dithio-1-Alkenes for Commercial Scale-Up
Advanced Metal-Free Synthesis of 1,1-Dithio-1-Alkenes for Commercial Scale-Up
The landscape of organic synthesis for functionalized alkenes is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective methodologies that do not compromise on yield or purity. A pivotal development in this domain is documented in patent CN102249962B, which discloses a highly efficient preparation method for 1,1-dithio-1-alkenes. These compounds serve as critical molecular building blocks in the synthesis of beta-lactam antibiotics, sugar derivatives, and polysulfide optical linear materials, making their production scalability a matter of strategic importance for the global fine chemical industry. The patented technology introduces a novel organocatalytic approach that utilizes 1,1-dibromo-1-alkenes as readily available starting materials, reacting them with thiols or mercaptans in the presence of the organic base 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU). Unlike traditional methods that rely on harsh conditions, this innovation operates effectively at room temperature in dimethyl sulfoxide (DMSO) solvent, representing a paradigm shift towards sustainable manufacturing practices for high-value intermediates.

For procurement managers and supply chain directors seeking a reliable pharmaceutical intermediate supplier, the implications of this metal-free protocol are profound. By removing the dependency on precious metals, the process not only lowers the raw material cost baseline but also mitigates the supply chain risks associated with fluctuating prices of palladium and nickel. The ability to synthesize these complex sulfur-containing olefins under mild conditions suggests a robust pathway for commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and delivery timelines for downstream API manufacturers. This report delves deep into the technical nuances of this invention, contrasting it with legacy technologies to highlight its potential for cost reduction in agrochemical intermediate manufacturing and broader fine chemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,1-dithio-1-alkenes has been plagued by significant technical and economic hurdles that limit their widespread industrial adoption. Traditional methodologies often necessitate the use of expensive transition metal catalysts, such as palladium or nickel, which introduce substantial cost burdens and complicate the purification process due to the stringent regulatory limits on heavy metal residues in pharmaceutical products. For instance, prior art describes the coupling of 1,1-dibromo-1-alkenes with alkyl indium sulfides under the joint action of palladium catalysts and expensive ligands like Xantphos, requiring elevated temperatures to drive the reaction to completion. Another representative method involves the reaction of 1,1-dibromo-1-alkenes with sodium alkyl sulfides under nickel catalysis at high temperatures around 120°C for extended periods. These harsh thermal conditions not only consume excessive energy but also severely limit substrate compatibility, often leading to decomposition of sensitive functional groups and the formation of complex impurity profiles that are difficult to separate.
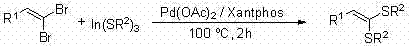
Furthermore, alternative palladium-catalyzed routes utilizing alkyl lithium sulfides have demonstrated poor efficiency, with reported conversions ranging merely from 10% to 20%, rendering them economically unviable for large-scale production. The reliance on stoichiometric amounts of organometallic reagents and the necessity for rigorous exclusion of moisture and oxygen in many of these protocols add layers of operational complexity and safety risks. For a reliable supplier aiming to deliver high-purity OLED material precursors or drug intermediates, these conventional methods present a bottleneck. The removal of trace metal catalysts often requires additional processing steps such as scavenging or recrystallization, which further erodes overall yield and increases the environmental footprint through the generation of hazardous waste streams containing heavy metals.
The Novel Approach
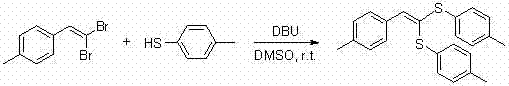
In stark contrast to these legacy techniques, the method disclosed in CN102249962B offers a streamlined, metal-free alternative that addresses the core inefficiencies of the prior art. By employing DBU as a non-nucleophilic organic base in DMSO, the reaction proceeds efficiently at room temperature (25°C), eliminating the need for energy-intensive heating systems and specialized high-pressure equipment. This mild condition is particularly advantageous for substrates containing thermally labile groups, thereby expanding the scope of accessible chemical space for medicinal chemists. The absence of transition metals means that the final product is inherently free from metal contamination, a critical quality attribute for high-purity pharmaceutical intermediates intended for human consumption. The operational simplicity is further enhanced by the use of commercially available and inexpensive reagents, facilitating a direct translation from laboratory bench to pilot plant and eventually to full-scale commercial production without the need for extensive process re-engineering.
Mechanistic Insights into DBU-Mediated Nucleophilic Substitution
The success of this novel synthetic route lies in the unique mechanistic pathway facilitated by the strong organic base DBU in a polar aprotic solvent. The reaction initiates with the deprotonation of the thiol or mercaptan by DBU, generating a highly nucleophilic thiolate anion in situ. This anion then attacks the electron-deficient carbon of the 1,1-dibromo-1-alkene substrate. Unlike metal-catalyzed cross-couplings that proceed through oxidative addition and reductive elimination cycles, this organocatalytic process likely follows a nucleophilic vinylic substitution (SNV) mechanism or an addition-elimination pathway. The first equivalent of thiolate displaces one bromine atom to form a vinyl sulfide intermediate, which retains sufficient electrophilicity at the alpha-position due to the remaining bromine and the electron-withdrawing nature of the double bond system. A second equivalent of thiolate subsequently attacks this intermediate, displacing the second bromine atom to yield the final 1,1-dithio-1-alkene product. The use of DMSO as the solvent is crucial, as its high polarity stabilizes the charged intermediates and enhances the nucleophilicity of the thiolate species, driving the reaction to completion rapidly even at ambient temperatures.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed pathways. The absence of metal centers precludes the formation of metal-complexed side products or homocoupling byproducts often seen in palladium chemistry. The primary impurities are likely to be unreacted starting materials or mono-substituted intermediates, which are generally easier to separate via standard chromatographic techniques using ethyl acetate and petroleum ether. The patent data indicates excellent tolerance for a wide range of electronic environments on the aromatic rings, with electron-withdrawing groups like nitro and chloro, as well as electron-donating groups like methyl and methoxy, all yielding high conversion rates. This robustness suggests that the transition state of the rate-determining step is not overly sensitive to steric or electronic perturbations, making the process highly predictable and reliable for diverse substrate libraries.
How to Synthesize 1,1-Dithio-1-Alkenes Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to minimize operational overhead while maximizing throughput. The protocol involves a simple one-pot procedure where all reagents are combined in a standard round-bottom flask, removing the need for slow addition funnels or inert atmosphere gloveboxes typically required for air-sensitive organometallic chemistry. The reaction time is exceptionally short, often completing within 5 to 15 minutes, which allows for rapid turnover in batch processing. Workup involves a standard aqueous quench followed by extraction, drying, and solvent removal, yielding a crude product that can be purified to analytical grade purity using routine column chromatography. This ease of execution makes it an ideal candidate for both rapid parallel synthesis in drug discovery campaigns and tonnage production for commercial supply chains.
- Prepare the reaction mixture by adding dimethyl sulfoxide (DMSO), 1,1-dibromo-1-alkene, thiophenol or mercaptan, and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) into a round-bottom flask.
- Stir the reaction mixture at room temperature (25°C) for 5 to 15 minutes to ensure complete conversion without the need for external heating.
- Quench the reaction with distilled water, extract with ethyl acetate, wash the organic layer with saturated brine, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the transition to this metal-free methodology offers compelling strategic benefits that extend beyond simple reagent costs. The elimination of precious metal catalysts represents a direct removal of a volatile cost component from the bill of materials, insulating the manufacturing process from market fluctuations in palladium and nickel prices. Furthermore, the simplified purification train reduces the consumption of specialized scavenging resins and solvents, contributing to a leaner and more efficient production workflow. The mild reaction conditions also translate to lower energy utility costs, as there is no requirement for sustained heating or cooling cycles, aligning with modern sustainability goals and reducing the carbon footprint of the manufacturing site.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete avoidance of expensive transition metal catalysts and proprietary ligands such as Xantphos, which can constitute a significant portion of the raw material cost in traditional routes. By substituting these with inexpensive, commodity-grade organic bases like DBU and common solvents like DMSO, the variable cost per kilogram of the final product is drastically reduced. Additionally, the high efficiency and short reaction times minimize reactor occupancy time, increasing the overall asset utilization rate of the production facility and allowing for greater output volume without capital expansion.
- Enhanced Supply Chain Reliability: Dependence on specialized catalytic systems often introduces single points of failure in the supply chain, where shortages of specific ligands or metal salts can halt production entirely. This new method relies on widely available bulk chemicals that are sourced from multiple global suppliers, thereby diversifying the supply base and enhancing resilience against disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality or environmental conditions, ensuring consistent batch-to-batch performance and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling up exothermic metal-catalyzed reactions often presents significant engineering challenges related to heat dissipation and safety management. The ambient temperature operation of this DBU-mediated process greatly simplifies scale-up, as thermal runaway risks are negligible. From an environmental standpoint, the absence of heavy metals simplifies waste treatment protocols, as the effluent does not require complex metal recovery or hazardous waste disposal procedures. This facilitates easier compliance with increasingly stringent environmental regulations and reduces the administrative burden associated with hazardous material handling and reporting.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of adopting this technology, we have compiled answers to common inquiries regarding the process parameters and product specifications. These insights are derived directly from the experimental data provided in the patent literature, ensuring accuracy and relevance for process development scientists. Understanding these details is crucial for integrating this synthetic route into existing manufacturing frameworks and for assessing its fit within specific project timelines and quality requirements.
Q: What are the primary advantages of this DBU-mediated method over traditional palladium-catalyzed routes?
A: The DBU-mediated method eliminates the need for expensive transition metal catalysts like palladium and costly ligands such as Xantphos. Furthermore, it operates at room temperature (25°C) rather than requiring high temperatures (up to 120°C), significantly reducing energy consumption and simplifying process safety.
Q: What is the substrate scope for the R1 and R2 groups in this synthesis?
A: The method demonstrates excellent compatibility with various substituents. R1 can be hydrogen, p-methyl, p-isopropyl, p-methoxy, p-chloro, m-chloro, o-chloro, p-bromo, p-nitro, or naphthyl. R2 accommodates phenyl, p-methylphenyl, or benzyl groups, allowing for diverse functionalization suitable for drug discovery.
Q: How does this process impact the purity profile of the final pharmaceutical intermediate?
A: By avoiding heavy metal catalysts, the risk of metal residue contamination is completely removed. This simplifies the downstream purification process, often requiring only standard column chromatography with ethyl acetate and petroleum ether to achieve high-purity specifications required for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Dithio-1-alkene Supplier
The technological breakthroughs detailed in patent CN102249962B underscore the immense potential of 1,1-dithio-1-alkenes as versatile intermediates for the next generation of pharmaceuticals and functional materials. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methods into robust, industrial-scale processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to multi-ton supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,1-dithio-1-alkene delivered meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this metal-free synthesis can optimize your specific supply chain. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener route impacts your total cost of ownership. We encourage potential partners to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, taking the first step towards a more efficient and sustainable sourcing strategy for your critical chemical intermediates.
