Revolutionizing Chiral Alcohol Production with Engineered Short-Chain Dehydrogenase Mutants for Industrial Scale-Up
Introduction to Advanced Biocatalytic Reduction Technologies
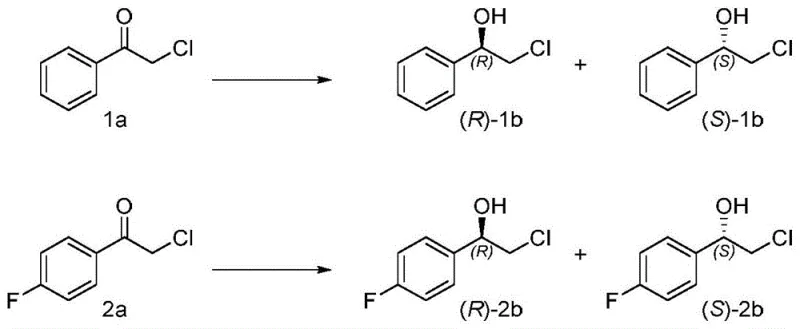
The landscape of chiral synthesis is undergoing a paradigm shift, driven by the urgent need for sustainable and highly selective manufacturing processes in the pharmaceutical and fine chemical industries. Patent CN108570460B represents a significant technological breakthrough in this domain, disclosing a series of engineered short-chain dehydrogenase (SDR) mutants derived from Bacillus megaterium and Bacillus subtilis. These biological catalysts are specifically designed to overcome the inherent limitations of natural enzymes, particularly their often rigid stereoselectivity which restricts their utility in synthesizing diverse chiral intermediates. By introducing precise point mutations at critical active site residues, such as position 187 in BmSDR5, the inventors have successfully reversed the enantioselectivity of the enzyme, enabling the production of optically active phenethyl alcohol derivatives that were previously difficult or impossible to access with high purity using wild-type biocatalysts. This innovation not only expands the toolbox available to process chemists but also aligns perfectly with the global push towards greener chemistry by replacing harsh chemical reagents with benign biological systems.
For procurement managers and supply chain directors, the implications of this technology are profound. The ability to access specific enantiomers directly through biocatalysis eliminates the need for costly and wasteful resolution steps, which traditionally discard up to 50% of the material. Furthermore, the robustness of these engineered enzymes, expressed efficiently in E. coli systems, suggests a scalable pathway for the commercial production of high-value chiral building blocks. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such proprietary biocatalytic routes is essential for securing a competitive advantage in the market. The patent details specific amino acid substitutions—such as replacing alanine with phenylalanine or tryptophan at key positions—that sterically reshape the enzyme's binding pocket, thereby dictating the orientation of the substrate during the hydride transfer step. This level of molecular precision ensures that the resulting chiral alcohols meet the stringent purity specifications required for active pharmaceutical ingredients (APIs), reducing the burden on downstream purification processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral alcohols, which serve as universal synthetic building blocks for numerous medicinal active ingredients, has relied on methods such as the chiral pool approach, asymmetric chemical synthesis using transition metal catalysts, or classical resolution techniques. While these methods have served the industry for decades, they are fraught with significant disadvantages that impact both cost and environmental sustainability. Chemical asymmetric reduction often necessitates the use of expensive and potentially toxic transition metal catalysts, such as ruthenium or rhodium complexes, which require rigorous removal steps to meet residual metal limits in pharmaceutical products. Moreover, these reactions frequently demand extreme operating conditions, including high pressures and temperatures, which increase energy consumption and operational risks. Classical resolution methods, although effective, are inherently inefficient because they theoretically yield a maximum of 50% of the desired enantiomer, generating an equivalent amount of unwanted isomer that must be disposed of or recycled, leading to substantial material waste and increased raw material costs. Additionally, the use of stoichiometric amounts of chiral auxiliaries or resolving agents further escalates the production cost and complicates the waste stream management, posing challenges for environmental compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes engineered short-chain dehydrogenase mutants to achieve asymmetric reduction under mild, ambient conditions, effectively circumventing the drawbacks of traditional chemical methods. This biocatalytic strategy leverages the exquisite specificity of enzymes to differentiate between prochiral faces of ketone substrates, delivering products with exceptionally high enantiomeric excess (ee) without the need for complex chiral ligands or harsh reagents. The core innovation lies in the protein engineering of SDRs to alter their stereopreference; for instance, mutating the wild-type BmSDR5 to include a phenylalanine at position 187 switches its selectivity from producing the (R)-enantiomer to predominantly yielding the (S)-enantiomer. This capability allows manufacturers to access both enantiomers of a target molecule simply by selecting the appropriate enzyme variant, providing unparalleled flexibility in process design. Furthermore, the integration of a cofactor regeneration system, typically involving glucose dehydrogenase, ensures that the expensive NADPH cofactor is recycled in situ, making the process economically viable on a large scale. This holistic biocatalytic platform offers a cleaner, safer, and more cost-effective route for cost reduction in chiral alcohol manufacturing, addressing the critical pain points of modern chemical production.
Mechanistic Insights into SDR-Catalyzed Asymmetric Reduction
The mechanistic elegance of short-chain dehydrogenases lies in their conserved catalytic machinery, which typically involves a catalytic tetrad (Ser-Tyr-Lys-Asn) that facilitates the transfer of a hydride ion from the nicotinamide ring of the NADPH cofactor to the carbonyl carbon of the substrate. In the context of the patented mutants, the alteration of stereoselectivity is achieved through strategic modifications of the substrate-binding pocket. For example, in the BmSDR5 mutant, the substitution of a smaller residue with a bulky aromatic amino acid like phenylalanine at position 187 introduces steric hindrance that forces the acetophenone derivative to bind in a flipped orientation compared to the wild-type enzyme. This reorientation ensures that the hydride attack occurs on the opposite face of the carbonyl group, thereby inverting the configuration of the resulting alcohol from (R) to (S). Such precise control over the transition state geometry is crucial for achieving high optical purity, as even minor deviations can lead to the formation of the wrong enantiomer, compromising the quality of the final API.
Beyond stereoselectivity, the engineered enzymes also demonstrate robust activity towards a variety of substituted acetophenone derivatives, as illustrated by the general structure in the patent where R1 can be a halogenated alkyl group and R2 can be various substituents on the benzene ring. The ability of these mutants to accommodate bulky or electron-withdrawing groups without significant loss of activity highlights the versatility of the protein engineering strategy employed. Impurity control is inherently superior in this enzymatic process because the enzyme's active site acts as a highly specific filter, rejecting substrates or transition states that do not fit the precise geometric and electronic requirements. This specificity minimizes the formation of side products such as over-reduced species or regio-isomers, which are common concerns in chemical catalysis. Consequently, the crude reaction mixture is often much cleaner, simplifying the work-up procedure and reducing the load on purification columns. For R&D directors, understanding these mechanistic details is vital for troubleshooting potential scale-up issues, such as substrate inhibition or cofactor depletion, and for designing fed-batch strategies that maximize space-time yield while maintaining the high selectivity promised by the mutant enzymes.
How to Synthesize Optically Active Phenethyl Alcohol Derivatives Efficiently
The practical implementation of this biocatalytic technology involves a streamlined workflow that begins with the cloning and expression of the specific SDR mutant gene in a suitable microbial host, typically E. coli. Once the recombinant strain is established, the enzyme can be produced via fermentation, lysed, and utilized either as a whole-cell biocatalyst or as a crude/purified enzyme preparation depending on the specific process requirements. The reaction itself is conducted in an aqueous buffer system, often supplemented with a water-miscible co-solvent to enhance the solubility of the hydrophobic acetophenone substrates. A critical component of the reaction mixture is the cofactor regeneration system, which couples the SDR-catalyzed reduction with the oxidation of glucose by glucose dehydrogenase, ensuring a continuous supply of NADPH. Detailed standardized synthesis steps, including specific buffer compositions, pH ranges (typically pH 5-9), temperature profiles, and substrate loading concentrations, are essential for reproducibility and are outlined in the comprehensive guide below.
- Clone the gene encoding the specific short-chain dehydrogenase mutant (e.g., BmSDR5-A187F) into an expression vector like pET-22b and transform into E. coli host cells.
- Express the recombinant enzyme in E. coli BL21 cultures induced by IPTG, followed by cell lysis to obtain the crude enzyme solution or purified protein.
- Conduct the asymmetric reduction reaction in a phosphate buffer system containing the acetophenone derivative substrate, NADP+ cofactor, and a cofactor regeneration system (e.g., glucose dehydrogenase/glucose) at mild temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of these engineered SDR mutants offers tangible strategic benefits that extend beyond mere technical feasibility. The transition from chemical to biocatalytic synthesis fundamentally alters the cost structure of chiral alcohol production by eliminating the need for precious metal catalysts and expensive chiral ligands, which are subject to volatile market prices and supply chain disruptions. Instead, the primary inputs become renewable carbohydrates (for cofactor regeneration) and standard fermentation media, which are abundant and cost-stable. This shift results in significant cost savings in manufacturing, as the process operates at ambient temperature and pressure, drastically reducing energy consumption compared to high-pressure hydrogenation or cryogenic chemical reductions. Moreover, the high selectivity of the enzymes means that raw material utilization is maximized, with minimal waste generation, further driving down the effective cost per kilogram of the final product. The elimination of heavy metal residues also simplifies regulatory compliance and reduces the costs associated with wastewater treatment and hazardous waste disposal.
- Cost Reduction in Manufacturing: The biocatalytic route described in the patent eliminates the reliance on expensive transition metal catalysts and stoichiometric chiral auxiliaries, which are major cost drivers in traditional asymmetric synthesis. By utilizing recombinant enzymes produced via fermentation, the variable costs associated with catalysts are significantly reduced, and the process avoids the complex and costly steps required to remove trace metals from the final product to meet pharmaceutical standards. Additionally, the high atom economy of the enzymatic reaction, coupled with the efficiency of the cofactor regeneration system, ensures that reagents are utilized effectively, leading to substantial overall cost optimization without compromising on yield or quality.
- Enhanced Supply Chain Reliability: Relying on biocatalysis enhances supply chain resilience by diversifying the source of chirality from finite mineral resources to renewable biological systems. The enzymes are produced in robust E. coli strains that can be scaled up rapidly in standard fermenters, ensuring a consistent and reliable supply of the biocatalyst regardless of geopolitical fluctuations affecting metal mining or chemical synthesis feedstocks. Furthermore, the stability of the engineered mutants allows for the potential development of immobilized enzyme formulations, which can be stored for extended periods and reused for multiple batches, thereby reducing the frequency of enzyme production runs and smoothing out supply chain logistics for long-term manufacturing campaigns.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory shake flasks to industrial-scale bioreactors without the safety risks associated with high-pressure hydrogenation or pyrophoric reagents. The mild reaction conditions (aqueous medium, neutral pH, moderate temperature) simplify the engineering requirements for production facilities, allowing for the use of standard stainless steel equipment rather than specialized high-pressure vessels. From an environmental perspective, the process generates significantly less hazardous waste, as it avoids the use of organic solvents in large volumes and toxic metal salts, aligning with increasingly stringent global environmental regulations and corporate sustainability goals, thus mitigating regulatory risks and enhancing the company's green credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these short-chain dehydrogenase mutants in industrial settings. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, aiming to clarify the operational parameters and potential advantages for stakeholders considering this technology for their manufacturing pipelines. Understanding these details is crucial for making informed decisions about process adoption and integration.
Q: How do the SDR mutants improve upon wild-type enzymes for chiral synthesis?
A: The patented mutants, such as BmSDR5-A187F, exhibit reversed or significantly enhanced stereoselectivity compared to wild-type enzymes. While wild-types often follow Prelog's rule yielding one specific enantiomer, these engineered variants can produce the opposite enantiomer (anti-Prelog) with high enantiomeric excess (ee), which is critical for synthesizing specific drug intermediates that require the less common stereochemistry.
Q: What are the substrate scopes compatible with these biocatalysts?
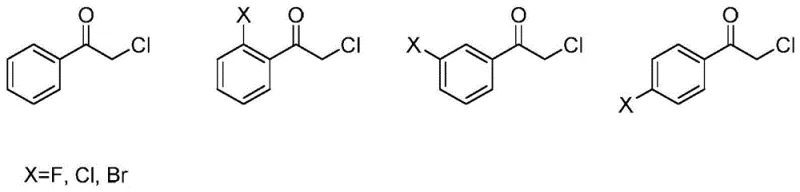
A: These short-chain dehydrogenase mutants are specifically designed to reduce acetophenone derivatives, including alpha-substituted derivatives like alpha-chloro acetophenone and 4-fluoro-alpha-chloro acetophenone. The enzyme active site accommodates various substituents on the benzene ring (hydrogen, halogens, nitro groups) and different alpha-groups (alkyl, halogenated alkyl), making them versatile for producing a range of chiral phenethyl alcohol building blocks.
Q: Is cofactor regeneration required for this biocatalytic process?
A: Yes, since short-chain dehydrogenases typically require NADPH or NADH as a cofactor for the reduction reaction, an efficient cofactor regeneration system is essential for industrial viability. The patent describes coupling the SDR mutant with glucose dehydrogenase (GDH) and glucose to continuously regenerate NADPH from NADP+, ensuring the reaction proceeds to high conversion without the need for stoichiometric amounts of expensive cofactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phenethyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic technologies disclosed in patents like CN108570460B for the production of high-value chiral intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust, cost-effective industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the high enantiomeric excess achieved by these engineered enzymes. We are committed to leveraging our expertise in protein engineering and fermentation technology to help our clients optimize these biocatalytic routes for maximum efficiency and yield.
We invite you to engage with our technical procurement team to discuss how these advanced SDR mutants can be integrated into your supply chain to achieve a Customized Cost-Saving Analysis tailored to your specific product portfolio. Whether you require specific COA data for validation or detailed route feasibility assessments to evaluate the transition from chemical to enzymatic synthesis, our experts are ready to provide the support you need. Partner with us to secure a sustainable and competitive supply of high-purity chiral building blocks for your next-generation pharmaceutical developments.
