Advanced Biocatalytic Routes for Chiral Phenethyl Alcohol Derivatives and Commercial Scalability
Advanced Biocatalytic Routes for Chiral Phenethyl Alcohol Derivatives and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable methods for producing chiral building blocks, which are essential for the synthesis of active pharmaceutical ingredients. Patent CN113293152B introduces a significant advancement in this field by disclosing novel short-chain dehydrogenase mutants and their specific applications in the asymmetric reduction of acetophenone derivatives. This technology represents a pivotal shift from traditional chemical synthesis to biocatalytic processes, offering enhanced stereoselectivity and environmental benefits. For R&D directors and procurement specialists, understanding the implications of this patent is crucial for optimizing supply chains and reducing manufacturing costs. The ability to produce optically active phenethyl alcohol derivatives with high precision addresses a critical bottleneck in drug development, where purity and isomeric composition are paramount. This report provides a deep technical and commercial analysis of the patented technology, highlighting its potential to transform the production of high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral alcohols, such as the chiral pool method, asymmetric chemical synthesis, and resolution techniques, have long been the standard in the industry. However, these approaches often suffer from significant drawbacks that impact both economic efficiency and environmental sustainability. Chemical asymmetric synthesis frequently requires the use of expensive transition metal catalysts, which can leave behind toxic residues that are difficult and costly to remove to meet stringent pharmaceutical purity standards. Furthermore, resolution methods are inherently inefficient, as they theoretically discard at least half of the starting material, leading to poor atom economy and increased waste generation. The need for extreme operating conditions, such as high pressures or temperatures, also increases energy consumption and safety risks. These limitations create substantial barriers for manufacturers aiming to scale up production while maintaining cost competitiveness and regulatory compliance.
The Novel Approach
In contrast, the biocatalytic approach detailed in the patent utilizes engineered short-chain dehydrogenase mutants to achieve highly selective reductions under mild conditions. This method leverages the inherent specificity of enzymes to distinguish between enantiomers, thereby eliminating the need for complex separation steps associated with racemic mixtures. By mutating specific amino acid residues in the enzyme's active site, the inventors have successfully altered the stereoselectivity to favor the production of desired isomers, including those with anti-Prelog selectivity which are often challenging to obtain. This biological route operates at ambient temperatures and neutral pH levels, significantly reducing energy requirements and minimizing the formation of hazardous by-products. The integration of cofactor recycling systems further enhances the economic viability of the process by reducing the consumption of expensive nicotinamide cofactors. This novel approach not only improves yield and purity but also aligns with green chemistry principles, making it an attractive option for sustainable manufacturing.
Mechanistic Insights into SDR-Catalyzed Asymmetric Reduction
The core of this technology lies in the precise engineering of short-chain dehydrogenases (SDRs) derived from sources such as Bacillus megaterium and Bacillus subtilis. The patent describes specific mutations at key positions, such as position 187 in BmSDR5, where substituting with aromatic amino acids like phenylalanine can dramatically reverse the enzyme's stereoselectivity. This structure-guided protein engineering allows for the customization of the biocatalyst to fit the steric and electronic requirements of specific substrates. The catalytic mechanism involves the transfer of a hydride ion from the cofactor NADPH to the carbonyl carbon of the ketone substrate, facilitated by a conserved catalytic tetrad within the enzyme's active site. By modifying the amino acids surrounding this active site, the spatial orientation of the substrate is altered, dictating which face of the carbonyl group receives the hydride. This level of control is essential for producing single-enantiomer products with high optical purity, which is a critical quality attribute for many pharmaceutical intermediates.
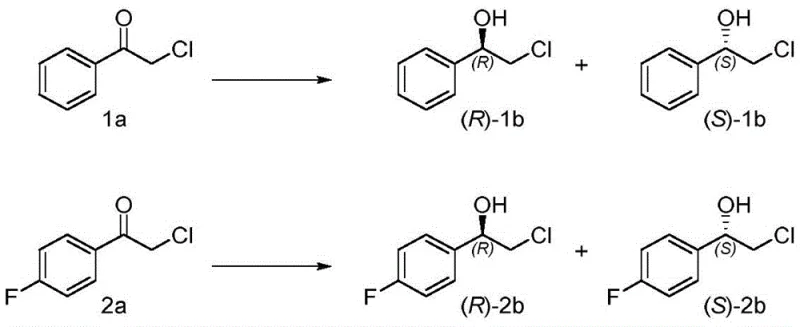
Furthermore, the impurity control mechanism in this biocatalytic system is inherently superior to chemical methods. Enzymes are highly specific, meaning they typically do not react with other functional groups present on the molecule, thereby reducing the formation of side products. The patent highlights that these mutants can effectively reduce various acetophenone derivatives, including those with halogen substitutions, without affecting the halogen atoms. This chemoselectivity is vital for complex molecule synthesis where protecting groups might otherwise be required. The ability to maintain high enantiomeric excess (ee) values across a range of substrates demonstrates the robustness of the engineered enzymes. For R&D teams, this means a more predictable and reliable synthesis route that minimizes the need for extensive downstream purification, ultimately streamlining the process development timeline and reducing the overall cost of goods.
How to Synthesize Phenethyl Alcohol Derivatives Efficiently
Implementing this biocatalytic route requires a systematic approach to enzyme expression and reaction optimization. The process begins with the cloning of the mutated gene sequences into suitable expression vectors, followed by transformation into E. coli host cells for protein production. Once the recombinant enzymes are expressed, they can be used in the form of whole cells or crude enzyme extracts, offering flexibility in process design. The reaction system typically involves a phosphate buffer to maintain pH stability, along with a cofactor regeneration system using glucose and glucose dehydrogenase to sustain the catalytic cycle. This setup ensures that the expensive cofactors are recycled in situ, making the process economically feasible for large-scale applications. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Clone and express specific short-chain dehydrogenase mutants (e.g., BmSDR5, BsSDR4) in E. coli host cells.
- Prepare the reaction system with phosphate buffer, cofactor NADP+, glucose, and glucose dehydrogenase for recycling.
- Add acetophenone derivative substrate and incubate at controlled temperature to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers compelling advantages that extend beyond technical performance. The shift from chemical to enzymatic synthesis fundamentally alters the cost structure and risk profile of manufacturing chiral intermediates. By eliminating the need for precious metal catalysts and harsh reagents, companies can significantly reduce raw material costs and mitigate supply chain vulnerabilities associated with these commodities. Additionally, the milder reaction conditions reduce the demand for specialized high-pressure equipment and extensive safety infrastructure, lowering capital expenditure requirements. The enhanced selectivity of the enzymes minimizes waste generation, which translates to lower disposal costs and a reduced environmental footprint. These factors collectively contribute to a more resilient and cost-effective supply chain, enabling companies to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage lead to substantial cost savings in the manufacturing process. Unlike traditional methods that require extensive purification to remove metal residues, this biocatalytic route produces high-purity products with minimal downstream processing. The use of renewable resources like glucose for cofactor regeneration further drives down operational expenses. These efficiencies allow for a more competitive pricing structure, providing a strategic advantage in the global market for pharmaceutical intermediates. The overall reduction in process complexity also lowers labor and maintenance costs associated with production facilities.
- Enhanced Supply Chain Reliability: Relying on biocatalysis reduces dependence on volatile markets for rare earth metals and hazardous chemicals, which are often subject to geopolitical tensions and supply disruptions. Enzymes can be produced consistently through fermentation, ensuring a stable and scalable supply of the catalyst. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream customers. Furthermore, the robustness of the engineered enzymes allows for storage and transport under standard conditions, simplifying logistics. By diversifying the sourcing of critical production inputs, companies can build a more resilient supply chain that is less susceptible to external shocks.
- Scalability and Environmental Compliance: The process is designed for scalability, with fermentation and biocatalytic reactions being well-established technologies in the industrial sector. The mild operating conditions and aqueous reaction media simplify the scale-up process, reducing the technical risks associated with moving from lab to plant. From an environmental perspective, the reduction in hazardous waste and energy consumption aligns with increasingly stringent global regulations on emissions and effluent discharge. This compliance not only avoids potential fines but also enhances the company's reputation as a sustainable manufacturer. The ability to scale up complex biocatalytic processes efficiently ensures that supply can meet growing demand without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial implementation of the patented short-chain dehydrogenase mutants. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation. Understanding these aspects is essential for stakeholders evaluating the integration of this technology into their existing manufacturing frameworks. The responses cover key areas such as enzyme performance, substrate compatibility, and process scalability, providing a comprehensive overview for decision-makers.
Q: How do these SDR mutants improve upon wild-type enzymes?
A: The mutants described in CN113293152B exhibit reversed or enhanced stereoselectivity compared to wild-types, allowing for the production of specific enantiomers that are difficult to access via conventional chemical synthesis.
Q: What are the substrate scopes for these biocatalysts?
A: The engineered enzymes effectively reduce various acetophenone derivatives, including those with halogen substitutions at the alpha or phenyl positions, providing broad utility for pharmaceutical intermediate synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of recombinant E. coli and cofactor recycling systems supports scalable production, offering a greener alternative to traditional metal-catalyzed reduction methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenethyl Alcohol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalytic technologies like those described in CN113293152B for the production of high-value chiral intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative enzymatic routes from the laboratory to commercial scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from development to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing the quality and consistency required for pharmaceutical applications. Our facility is equipped to handle complex biocatalytic processes, providing a secure and efficient pathway for your supply chain needs.
We invite you to collaborate with us to optimize your synthesis routes and achieve significant operational efficiencies. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific production requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Our experts are ready to discuss how we can support your goals for cost reduction in chiral alcohol manufacturing and reducing lead time for high-purity chiral intermediates. Let us help you navigate the complexities of modern chemical synthesis and secure a reliable supply of critical materials for your business.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
