Revolutionizing D-Tryptophan Production via Advanced Tryptophanase Mutants for Commercial Scale
Revolutionizing D-Tryptophan Production via Advanced Tryptophanase Mutants for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for producing optically active amino acids, particularly non-proteinogenic variants like D-tryptophan. A significant breakthrough in this domain is documented in patent CN110904086A, which discloses a novel tryptophanase mutant capable of catalyzing the condensation of D-serine and indole. This technology represents a paradigm shift from traditional chemical synthesis and inefficient resolution methods, offering a direct biocatalytic route that addresses critical pain points regarding purity, environmental impact, and yield. For R&D directors and procurement specialists, understanding the mechanistic underpinnings of this mutant enzyme is essential for evaluating its potential in large-scale manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of D-tryptophan has been plagued by significant inefficiencies inherent to both chemical and biological legacy processes. The chemical synthesis route typically involves complex multi-step reactions that generate substantial environmental pollution and often result in low overall yields due to the difficulty in controlling stereochemistry without expensive chiral auxiliaries. Alternatively, the biological resolution method, which relies on racemizing L-tryptophan into DL-tryptophan followed by the enzymatic degradation of the L-isomer, suffers from a theoretical maximum yield of only 50%. This intrinsic limitation means that half of the starting material is essentially wasted or requires energy-intensive recycling, drastically inflating the cost of goods sold (COGS). Furthermore, the hydantoinase pathway, while industrially applied, necessitates the use of chemically synthesized hydantoin substrates, introducing heavy environmental costs and complex downstream purification requirements to remove byproducts.
The Novel Approach
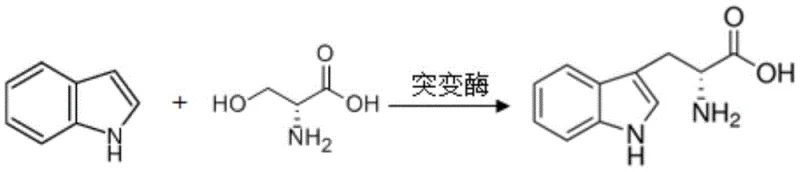
In stark contrast to these legacy methods, the technology described in CN110904086A introduces a groundbreaking enzymatic solution that bypasses the 50% yield ceiling of resolution strategies. By utilizing a specifically engineered tryptophanase mutant (SEQ ID NO: 3), manufacturers can directly synthesize D-tryptophan from readily available precursors: D-serine and indole. This condensation reaction is catalyzed with high specificity, eliminating the formation of the unwanted L-isomer entirely and thus removing the need for difficult chiral separations. The process operates under mild aqueous conditions, significantly reducing the reliance on hazardous organic solvents and heavy metal catalysts often found in chemical routes. This not only simplifies the downstream processing workflow but also aligns with modern green chemistry principles, offering a cleaner and more economically viable pathway for the commercial scale-up of complex amino acid intermediates.
Mechanistic Insights into Tryptophanase Mutant Catalysis
The core innovation lies in the precise protein engineering of the tryptophanase enzyme, transforming it from a degradative enzyme into a synthetic powerhouse. The wild-type tryptophanase (SEQ ID NO: 1), derived from Escherichia coli, naturally catalyzes the decomposition of L-tryptophan and is inherently inactive towards the synthesis of D-tryptophan from D-serine. Through error-prone PCR and high-throughput screening, six critical amino acid substitutions were identified: P111S, K115D, K156E, L345A, I348V, and F376G. These mutations collectively reshape the enzyme's active site architecture, altering its substrate binding pocket to accommodate D-serine effectively. The substitution of bulky or charged residues at these specific positions likely modifies the electrostatic environment and steric constraints, enabling the enzyme to stabilize the transition state for the C-C bond formation between the beta-carbon of D-serine and the C3 position of the indole ring.
From an impurity control perspective, this biocatalytic mechanism offers superior selectivity compared to chemical alternatives. Because the enzyme is highly specific for the D-isomer of serine and the indole substrate, the generation of side products such as regio-isomers or over-alkylated byproducts is minimized. The requirement for pyridoxal phosphate (PLP) as a cofactor ensures a controlled Schiff base mechanism, which prevents non-enzymatic background reactions that often lead to complex impurity profiles in chemical synthesis. For quality assurance teams, this means the crude reaction mixture contains significantly fewer structurally related impurities, thereby reducing the burden on purification columns and crystallization steps. This high fidelity in catalysis ensures that the final API intermediate meets stringent pharmacopeial standards with reduced risk of genotoxic impurities often associated with harsh chemical reagents.
How to Synthesize D-Tryptophan Efficiently
Implementing this biocatalytic route requires a robust fermentation and bioconversion protocol to maximize the expression of the mutant enzyme and its subsequent catalytic efficiency. The process begins with the cultivation of recombinant E. coli strains harboring the mutant gene, followed by induction to produce the biocatalyst. The subsequent bioconversion step is straightforward, utilizing whole cells or cell lysates in a buffered aqueous system. This simplicity allows for easy integration into existing fermentation facilities without the need for specialized high-pressure reactors or exotic solvent handling systems. The detailed standardized synthesis steps for optimizing yield and productivity are outlined in the technical guide below.
- Ferment the recombinant E. coli BL21(DE3) strain expressing the mutant enzyme (SEQ ID NO: 3) in TB medium at 37°C until OD600 reaches 0.6-0.8, then induce with IPTG.
- Harvest the wet cells via centrifugation and resuspend them in a reaction buffer containing 0.1M potassium phosphate (pH 8.3), 1.0mM D-serine, 10mM indole, and 0.2mM PLP.
- Maintain the reaction mixture at 36-38°C with stirring for 24 hours to achieve approximately 41% conversion of D-serine to D-tryptophan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic advantages beyond mere technical feasibility. The shift from chemical synthesis to biocatalysis fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the need for expensive chiral resolving agents and hazardous chemical reagents, the raw material costs are significantly optimized. Furthermore, the ability to use whole-cell biocatalysts reduces the capital expenditure required for enzyme purification equipment, as the cells themselves act as natural immobilized enzymes. This simplification of the manufacturing process enhances supply chain resilience by reducing the number of critical unit operations and potential failure points, ensuring a more reliable supply of high-purity D-tryptophan for downstream drug synthesis.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and organic solvents drastically reduces waste disposal costs and raw material expenses. Unlike resolution methods that inherently discard 50% of the substrate, this direct synthesis route maximizes atom economy, leading to substantial cost savings per kilogram of product. The mild reaction conditions also lower energy consumption for heating and cooling, contributing to a leaner manufacturing budget without compromising on output quality.
- Enhanced Supply Chain Reliability: The reliance on fermentable substrates like D-serine and indole, which are commercially available in bulk, mitigates the risk of raw material shortages often associated with specialized chemical intermediates. The robustness of the E. coli expression system ensures consistent batch-to-batch production capacity, allowing for scalable manufacturing that can respond rapidly to fluctuations in market demand. This stability is crucial for maintaining continuous production schedules for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system simplifies scale-up from laboratory to industrial fermenters, avoiding the safety hazards linked to exothermic chemical reactions in organic solvents. Additionally, the biodegradable nature of the biological waste stream facilitates easier compliance with increasingly strict environmental regulations. This eco-friendly profile not only reduces regulatory hurdles but also enhances the corporate sustainability image, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tryptophanase mutant technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this biocatalytic route into their existing production portfolios.
Q: How does the mutant enzyme differ from wild-type tryptophanase?
A: The wild-type tryptophanase (SEQ ID NO: 1) is inactive towards D-serine condensation. The mutant (SEQ ID NO: 3), featuring six specific point mutations (P111S, K115D, K156E, L345A, I348V, F376G), fundamentally alters substrate selectivity to catalyze D-tryptophan synthesis.
Q: What are the optimal reaction conditions for this biocatalytic process?
A: The process operates under mild physiological conditions, specifically at a temperature range of 36-38°C and a pH of 8.3 using a potassium phosphate buffer, requiring pyridoxal phosphate (PLP) as a cofactor.
Q: Why is this enzymatic route superior to chemical synthesis for D-tryptophan?
A: Unlike chemical synthesis which involves complex steps and heavy pollution, or resolution methods which waste 50% of the L-isomer, this direct condensation route offers higher atom economy and eliminates the need for hazardous solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Tryptophan Supplier
The enzymatic synthesis of D-tryptophan described in patent CN110904086A exemplifies the kind of innovative process chemistry that drives modern pharmaceutical manufacturing forward. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory technologies are successfully translated into robust industrial processes. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of D-tryptophan meets the exacting standards required for API synthesis, providing our partners with a secure and high-quality supply source.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in biocatalysis and process optimization, we can help you transition to this more efficient manufacturing route. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced capabilities can enhance your supply chain efficiency and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
