Advanced Manufacturing of Tadalafil: A High-Efficiency D-Tryptophan Based Synthetic Strategy for Commercial Scale-Up
Advanced Manufacturing of Tadalafil: A High-Efficiency D-Tryptophan Based Synthetic Strategy for Commercial Scale-Up
The pharmaceutical landscape for erectile dysfunction treatments continues to evolve, driven by the demand for high-purity active pharmaceutical ingredients (APIs) produced via cost-effective and scalable routes. Patent CN103524502A introduces a robust preparation method for Tadalafil, a potent PDE5 inhibitor, utilizing D-tryptophan as the chiral pool starting material. This approach represents a significant technological leap over prior art, addressing critical pain points such as low overall yields, complex purification protocols, and the reliance on expensive chiral catalysts. By leveraging the inherent stereochemistry of D-tryptophan, the process ensures high optical purity of the final product while maintaining mild reaction conditions that are conducive to safe, large-scale operations. For procurement leaders and R&D directors seeking a reliable API intermediate supplier, understanding the nuances of this synthetic pathway is essential for securing a stable supply of high-quality Tadalafil.
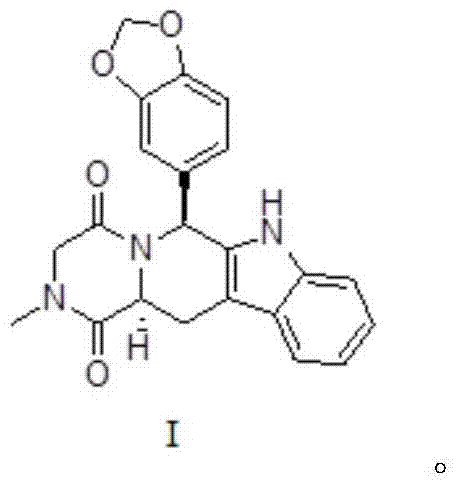
Tadalafil, chemically known as (6R,12aR)-6-(1,3-benzodioxol-5-yl)-2-methyl-2,3,6,7,12,12a-hexahydropyrazino[1',2':1,6]pyrido[3,4-b]indole-1,4-dione, is a cornerstone molecule in men's health therapeutics. The structural complexity of this molecule, featuring multiple chiral centers and fused ring systems, traditionally poses significant challenges for synthetic chemists aiming for commercial viability. The patent highlights a method that effectively navigates these complexities through a logical, step-wise construction of the molecular framework. This technical insight report delves deep into the mechanistic underpinnings and commercial implications of this novel preparation method, offering a comprehensive view for stakeholders involved in cost reduction in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tadalafil has been plagued by several inefficiencies that hinder optimal production economics and supply chain reliability. Many conventional routes rely on racemic synthesis followed by cumbersome chiral resolution steps, which inherently cap the maximum theoretical yield at 50% and generate substantial chemical waste. Furthermore, traditional methods often employ harsh reagents and extreme reaction conditions, such as cryogenic temperatures or high-pressure hydrogenation, which require specialized infrastructure and increase operational expenditures (OPEX). The presence of difficult-to-remove impurities in these older pathways frequently necessitates multiple recrystallization or chromatography steps, driving up solvent consumption and extending production lead times. These factors collectively contribute to a fragile supply chain where commercial scale-up of complex pharmaceutical intermediates becomes risky and cost-prohibitive.
The Novel Approach
In stark contrast, the method disclosed in CN103524502A utilizes a chiral pool strategy starting from D-tryptophan, effectively bypassing the need for resolution and ensuring high stereochemical fidelity from the outset. This novel approach streamlines the synthesis into four distinct, high-yielding steps that can be performed using standard reactor equipment found in most fine chemical facilities. The process operates under mild conditions, typically between 0°C and 100°C, utilizing common solvents like dichloromethane, isopropanol, and DMF. By eliminating the need for exotic catalysts or extreme physical parameters, this route significantly lowers the barrier to entry for manufacturing. The result is a process that not only delivers high-purity Tadalafil but also offers a simplified workflow that enhances throughput and reduces the environmental footprint associated with API production.
Mechanistic Insights into the D-Tryptophan Based Cascade
The core of this synthetic innovation lies in the efficient transformation of the amino acid backbone into the complex pyrazinoindole scaffold. The journey begins with the activation of D-tryptophan (Compound 1) using thionyl chloride (SOCl2) in the presence of a catalytic amount of DMF. This step converts the carboxylic acid into a highly reactive acid chloride intermediate (Compound 2), a crucial electrophile for the subsequent amide bond formation. The use of DMF as a catalyst facilitates the formation of the Vilsmeier-Haack reagent in situ, accelerating the chlorination process and ensuring near-quantitative conversion, as evidenced by the reported 99% yield in embodiment examples. This high efficiency at the very first step sets a positive tone for the overall mass balance of the process.
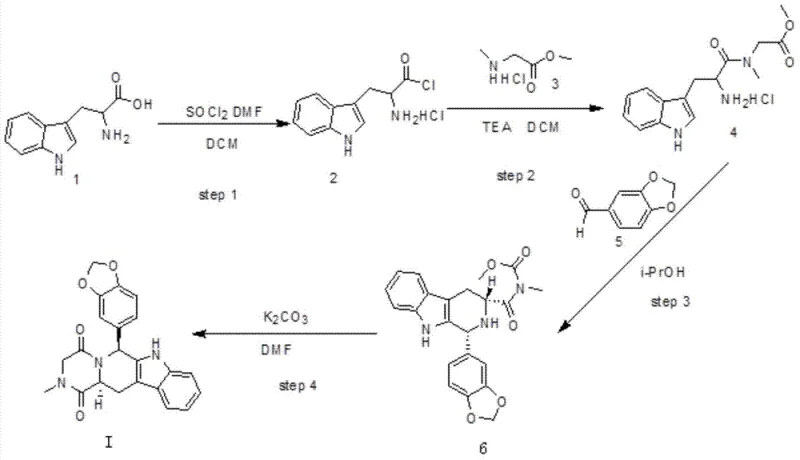
Following activation, the acid chloride undergoes nucleophilic attack by N-methylglycine methyl ester (sarcosine methyl ester, Compound 3) in the presence of a base such as triethylamine. This amidation step constructs the linear precursor (Compound 4), linking the tryptophan indole moiety with the glycine fragment. The subsequent Pictet-Spengler cyclization with piperonal (Compound 5) is the pivotal moment where the tetrahydro-beta-carboline ring system is formed. Conducted in isopropanol under reflux, this acid-catalyzed condensation creates the new C-C bond and establishes the critical stereocenter at the C1 position. Finally, the intramolecular cyclization of intermediate (Compound 6) using potassium carbonate closes the diketopiperazine ring, yielding the final Tadalafil structure (Compound I). This cascade demonstrates excellent atom economy and step convergence.
How to Synthesize Tadalafil Efficiently
Implementing this synthetic route requires precise control over reaction parameters to maximize yield and purity, particularly during the cyclization stages. The patent provides detailed embodiments that serve as a blueprint for process chemists, outlining specific molar ratios, solvent choices, and temperature profiles. For instance, the selection of isopropanol over ethanol for the Pictet-Spengler step was shown to dramatically improve yields from 47% to 89%, highlighting the sensitivity of the reaction to solvent properties. Understanding these nuances is vital for transferring the technology from the laboratory bench to pilot and production scales. The following guide summarizes the standardized operational procedure derived from the patent data.
- Activate D-tryptophan (Compound 1) using thionyl chloride and DMF in dichloromethane to form the acid chloride intermediate (Compound 2).
- Couple the acid chloride with N-methylglycine methyl ester (Compound 3) using triethylamine to generate the amide precursor (Compound 4).
- Perform Pictet-Spengler cyclization with piperonal (Compound 5) in isopropanol to form the tetrahydro-beta-carboline intermediate (Compound 6).
- Execute final intramolecular cyclization using potassium carbonate in DMF or THF to yield pure Tadalafil (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical merits of this patent translate directly into tangible business value. The shift towards a D-tryptophan based synthesis mitigates many of the risks associated with traditional manufacturing, offering a more predictable and cost-efficient supply model. By analyzing the process inputs and operational requirements, we can identify several key areas where this technology drives value for the organization, specifically regarding raw material stability, operational simplicity, and regulatory compliance.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the elimination of chiral resolution steps and the use of inexpensive, commodity-grade starting materials. D-tryptophan is a widely available fermentation product, ensuring stable pricing and availability compared to specialized chiral building blocks. Furthermore, the high yields reported in the patent embodiments, such as the 99% conversion in the activation step and 89% in the cyclization, minimize raw material waste. The avoidance of expensive transition metal catalysts removes the need for costly metal scavenging processes and rigorous residual metal testing, further lowering the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-proprietary reagents and solvents. Thionyl chloride, triethylamine, and potassium carbonate are bulk chemicals with established global supply networks, reducing the risk of bottlenecks. The process tolerance to mild reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. Additionally, the simplified purification protocol, which relies on standard crystallization and filtration rather than complex chromatography, accelerates batch turnover times. This efficiency allows manufacturers to respond more agilely to market demand fluctuations, ensuring reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route offers significant advantages for large-scale operations. The reaction conditions are gentle, operating at atmospheric pressure and moderate temperatures, which reduces energy consumption and safety hazards associated with high-pressure reactors. The solvent system, primarily consisting of dichloromethane, isopropanol, and DMF, is well-understood and manageable within standard waste treatment frameworks. The high selectivity of the reactions minimizes the formation of by-products, leading to a cleaner crude product and reducing the load on wastewater treatment facilities. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common questions regarding its implementation and performance. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the technology's capabilities.
Q: What are the key advantages of using D-tryptophan as a starting material for Tadalafil?
A: Using D-tryptophan offers significant advantages including high stereochemical purity which simplifies downstream purification, readily available commercial sourcing which stabilizes supply chains, and a streamlined reaction pathway that avoids complex chiral resolution steps often required in racemic syntheses.
Q: How does this patent address the issue of harsh reaction conditions in traditional Tadalafil synthesis?
A: The described method utilizes mild reaction temperatures ranging from 0°C to 100°C and employs common organic solvents like dichloromethane and isopropanol. This eliminates the need for specialized high-pressure equipment or cryogenic conditions, thereby reducing operational risks and energy consumption.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. It features high yields (up to 99% in the activation step), simple work-up procedures involving standard extraction and filtration, and avoids the generation of inseparable impurities, making it highly robust for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tadalafil Supplier
The synthesis of Tadalafil via the D-tryptophan route exemplifies the kind of process innovation that drives value in the modern pharmaceutical supply chain. At NINGBO INNO PHARMCHEM, we specialize in translating such complex synthetic pathways into reliable commercial realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Tadalafil meets the highest international standards for potency and safety.
We invite potential partners to engage with our technical procurement team to discuss how this optimized manufacturing route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a solid foundation of technical and commercial certainty.
