Scalable Green Synthesis of Ethacridine Lactate Intermediate for Global Pharmaceutical Manufacturing
Scalable Green Synthesis of Ethacridine Lactate Intermediate for Global Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for producing critical active pharmaceutical ingredients and their precursors. Patent CN101560185A introduces a groundbreaking methodology for the preparation of ethacridine lactate intermediates, specifically targeting the synthesis of 2-ethoxy-6-nitro-9-aminoacridine hydrochloride. This compound serves as the pivotal building block for Ethacridine Lactate, widely known as Rivanol, a potent antiseptic and disinfectant used extensively in wound care and dermatological treatments. The structural integrity of the final API relies heavily on the purity of this acridine derivative, making the synthesis route a matter of critical quality assurance. 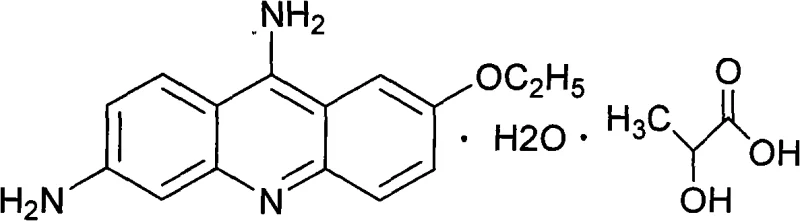 Traditional manufacturing processes have long been plagued by the use of hazardous reagents and complex purification steps that drive up costs and environmental liabilities. The innovation presented in this patent shifts the paradigm by employing a green chemistry approach that utilizes ammonium salts and potassium activators, effectively replacing the historically toxic phenol-based amination protocols. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while adhering to increasingly stringent global environmental regulations. The ability to produce this key intermediate with higher yields and reduced ecological footprint positions this technology as a preferred choice for modern contract development and manufacturing organizations.
Traditional manufacturing processes have long been plagued by the use of hazardous reagents and complex purification steps that drive up costs and environmental liabilities. The innovation presented in this patent shifts the paradigm by employing a green chemistry approach that utilizes ammonium salts and potassium activators, effectively replacing the historically toxic phenol-based amination protocols. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while adhering to increasingly stringent global environmental regulations. The ability to produce this key intermediate with higher yields and reduced ecological footprint positions this technology as a preferred choice for modern contract development and manufacturing organizations.
The core value of this intermediate lies in its specific substitution pattern on the acridine ring system, which dictates the biological activity of the final drug substance. 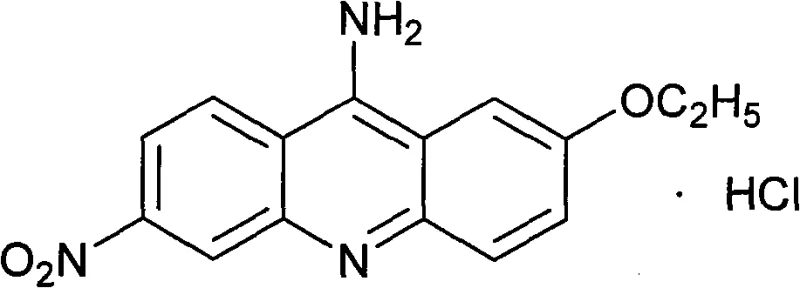 The molecule features a nitro group at the 6-position and an amino group at the 9-position, along with an ethoxy substituent, creating a unique electronic environment essential for its antimicrobial properties. Ensuring the correct regioselectivity during the synthesis is paramount, as isomers or incomplete reactions can lead to difficult-to-remove impurities that compromise the safety profile of the final medication. The patent details a robust protocol that ensures high regioselectivity through controlled reaction conditions and specific catalyst activation, providing a reliable source of high-purity pharmaceutical intermediates for downstream processing. By mastering this specific molecular architecture, manufacturers can guarantee the efficacy and safety of the final antiseptic product.
The molecule features a nitro group at the 6-position and an amino group at the 9-position, along with an ethoxy substituent, creating a unique electronic environment essential for its antimicrobial properties. Ensuring the correct regioselectivity during the synthesis is paramount, as isomers or incomplete reactions can lead to difficult-to-remove impurities that compromise the safety profile of the final medication. The patent details a robust protocol that ensures high regioselectivity through controlled reaction conditions and specific catalyst activation, providing a reliable source of high-purity pharmaceutical intermediates for downstream processing. By mastering this specific molecular architecture, manufacturers can guarantee the efficacy and safety of the final antiseptic product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-aminoacridine derivatives has relied heavily on the Bernthsen reaction or variations involving the amination of chloro-acridines using phenol and aqueous ammonia. These traditional routes present severe drawbacks that hinder modern commercial scale-up of complex pharmaceutical intermediates. The use of phenol as a solvent or promoter in the amination step introduces significant toxicity risks, requiring specialized handling equipment and extensive personal protective gear for operators. Furthermore, the post-reaction wastewater contains high concentrations of phenolic compounds, which are notoriously difficult and expensive to treat to meet environmental discharge standards. The reliance on ammonia water often results in lower reaction rates and necessitates high-pressure equipment to achieve acceptable conversion, adding capital expenditure to the manufacturing setup. Additionally, the separation of the desired amino product from phenolic byproducts often requires multiple recrystallization steps, leading to substantial yield losses and increased solvent consumption. These factors collectively inflate the cost of goods sold and create supply chain vulnerabilities due to regulatory scrutiny on phenol usage.
The Novel Approach
The methodology disclosed in CN101560185A offers a transformative solution by replacing the hazardous phenol and ammonia water system with a combination of solid ammonium salts and potassium activators in an alcoholic medium. This novel approach leverages the nucleophilic potential of ammonium ions, which are activated by potassium salts such as potassium sulfate or potassium carbonate to facilitate the displacement of the chlorine atom at the 9-position of the acridine ring. By conducting the reaction in organic alcohol solvents like ethanol or isopropanol, the process achieves a homogeneous reaction environment that enhances mass transfer and reaction kinetics without the need for extreme pressures. The elimination of phenol not only removes a major toxicological hazard but also simplifies the downstream purification process, as the byproducts are inorganic salts that are easily removed by aqueous washing. This shift enables cost reduction in pharmaceutical intermediates manufacturing by lowering raw material costs, reducing waste treatment expenses, and improving the overall atom economy of the synthesis. The result is a cleaner, safer, and more economically viable pathway that aligns perfectly with the principles of green chemistry.
Mechanistic Insights into Ammonium Salt-Mediated Nucleophilic Substitution
The heart of this innovative synthesis lies in the mechanistic details of the nucleophilic aromatic substitution where the chlorine atom is replaced by an amino group. In the conventional view, the electron-withdrawing nitro group at the 6-position activates the 9-position for nucleophilic attack, but the leaving group ability of chloride in this rigid acridine system often requires harsh conditions. The introduction of potassium salts acts as a phase transfer or activation catalyst, likely coordinating with the nitrogen of the ammonium salt to increase its nucleophilicity or stabilizing the transition state through cation-pi interactions with the aromatic system. The reaction proceeds through a Meisenheimer-like complex where the ammonium species attacks the C-9 carbon, followed by the elimination of hydrogen chloride. The use of alcohols as solvents is critical here, as they solvate the ionic intermediates effectively while remaining stable under the reflux conditions of 100°C to 200°C. This careful balance of solvent polarity and thermal energy ensures that the reaction proceeds to completion without degrading the sensitive nitro group or the ethoxy side chain. Understanding this mechanism allows process chemists to fine-tune the molar ratios of ammonium salt to substrate, typically optimizing around a 1:3 ratio to drive the equilibrium forward efficiently.
Impurity control is another critical aspect where this mechanism offers distinct advantages over traditional methods. In phenol-based routes, O-alkylation or ether formation can occur as side reactions, leading to impurities that are structurally similar to the product and hard to separate. By utilizing ammonium salts in an alcohol medium, the reaction specificity is directed almost exclusively towards N-arylation, minimizing the formation of ether byproducts. Furthermore, the subsequent salt formation step with hydrochloric acid is performed under controlled addition rates (1-3 ml/min) to ensure the precipitation of the hydrochloride salt in a crystalline form that excludes soluble organic impurities. The patent specifies a pH adjustment to 7-9 after the initial cyclization with phosphorus oxychloride, which is crucial for quenching excess POCl3 and preventing the formation of phosphorylated impurities that could carry through to the final step. This multi-stage control strategy ensures that the final intermediate meets the stringent purity specifications required for pharmaceutical applications, reducing the burden on analytical quality control labs.
How to Synthesize 2-Ethoxy-6-nitro-9-aminoacridine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing the target intermediate with high efficiency and reproducibility. The process begins with the cyclization of 2-p-ethoxyanilino-4-nitrobenzoic acid using phosphorus oxychloride, a standard dehydrating agent that closes the central ring of the acridine system. Following this, the crude chloro-intermediate is subjected to the amination step where the choice of ammonium salt and activator determines the success of the transformation. Detailed operational parameters regarding temperature ramps, stirring speeds, and addition rates are critical to maximizing yield and minimizing side reactions. For a comprehensive guide on the exact experimental procedures and safety precautions, please refer to the standardized synthesis steps provided below.
- Cyclization of 2-p-ethoxyanilino-4-nitrobenzoic acid with phosphorus oxychloride at 80-110°C to form the chloro-intermediate.
- Nucleophilic amination using ammonium sulfate and potassium sulfate in an alcoholic solvent at 100-200°C to replace the chlorine atom.
- Salt formation by refluxing the amino-acridine in water followed by controlled addition of hydrochloric acid to precipitate the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this green synthesis route offers tangible strategic benefits that extend beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the raw material portfolio, shifting away from regulated, high-cost commodities like phenol to commodity chemicals like ammonium sulfate and potassium salts which are abundant and price-stable. This substitution mitigates the risk of supply disruptions caused by environmental crackdowns on phenol producers and reduces the volatility associated with petrochemical-derived feedstocks. Moreover, the simplified waste profile means that manufacturing facilities do not need to invest in specialized phenol incineration or advanced oxidation wastewater treatment units, leading to substantial capital and operational expenditure savings. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures and ensuring consistent delivery schedules to downstream API manufacturers. These factors collectively enhance the resilience of the supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic phenol with low-cost ammonium salts directly lowers the bill of materials for every kilogram of intermediate produced. Additionally, the elimination of phenol removes the need for complex solvent recovery systems designed to handle high-boiling, toxic aromatics, thereby reducing energy consumption and utility costs. The higher yields reported in the patent examples further contribute to cost efficiency by maximizing the output from a fixed amount of starting benzoic acid derivative. When combined with the reduced costs of waste disposal and environmental compliance, the total cost of ownership for this manufacturing process is significantly lower than legacy methods. This economic advantage allows suppliers to offer more competitive pricing without compromising on margin, creating a win-win scenario for both the manufacturer and the buyer.
- Enhanced Supply Chain Reliability: Sourcing ammonium salts and potassium activators is far less risky than sourcing phenol, as these inorganic salts are produced globally in massive quantities for the fertilizer and industrial sectors. This abundance ensures that raw material shortages are unlikely to impact production schedules, providing a stable foundation for long-term supply agreements. Furthermore, the one-pot variation of this process reduces the number of unit operations and intermediate storage requirements, shortening the overall lead time from raw material intake to finished goods. The simplified logistics of handling non-hazardous solids instead of corrosive liquids also streamlines warehouse operations and reduces transportation hazards. Consequently, partners can rely on a more predictable and agile supply chain capable of responding quickly to fluctuations in market demand for antiseptic agents.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with modern ESG (Environmental, Social, and Governance) goals, making it easier to obtain regulatory approvals for facility expansions or new plant constructions. The absence of phenolic effluents simplifies the permitting process and reduces the ongoing monitoring burden imposed by environmental agencies. From a scalability perspective, the reaction exotherms are manageable, and the use of common solvents like ethanol facilitates easy scale-up from pilot plant to commercial metric-ton scale without requiring exotic reactor materials. The process generates inorganic salts as the primary waste, which are easier to treat or dispose of compared to complex organic sludge. This environmental compatibility future-proofs the manufacturing asset against tightening global regulations on chemical emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific improvements and data points highlighted in the patent documentation to provide clarity on the feasibility and benefits of the new route. Understanding these details is crucial for technical teams evaluating the transfer of this process to their own manufacturing sites or for procurement teams assessing the quality of the supply base.
Q: What are the primary advantages of the ammonium salt amination method over traditional phenol-based routes?
A: The new method replaces toxic phenol and ammonia water with inexpensive and safer ammonium salts (like ammonium sulfate) and potassium activators. This eliminates the generation of phenolic wastewater, significantly reducing environmental treatment costs and improving operator safety while maintaining high reaction yields.
Q: How does the one-pot reaction method impact production efficiency?
A: The one-pot method combines the cyclization, amination, and salt formation steps into a streamlined sequence without isolating the unstable chloro-intermediate. This reduces solvent consumption, minimizes material transfer losses, and shortens the overall production cycle time compared to multi-step isolation processes.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial reagents like phosphorus oxychloride and ethanol, and operates at manageable temperatures (80-200°C). The elimination of hazardous phenol simplifies waste management compliance, making it highly scalable for metric-ton production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ethoxy-6-nitro-9-aminoacridine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, green synthesis technologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering this key ethacridine lactate intermediate with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our capability to implement the ammonium salt-mediated amination process allows us to offer a product that is not only chemically superior but also environmentally responsible, catering to the sustainability goals of our multinational clients.
We invite you to collaborate with us to leverage this cutting-edge technology for your antiseptic and dermatological product lines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this green route for your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain security and reduce your overall production costs. Let us be your partner in driving efficiency and quality in the production of essential pharmaceutical intermediates.
