Scalable Manufacturing of Ethacridine Lactate via Direct Reductive Salt Formation
Scalable Manufacturing of Ethacridine Lactate via Direct Reductive Salt Formation
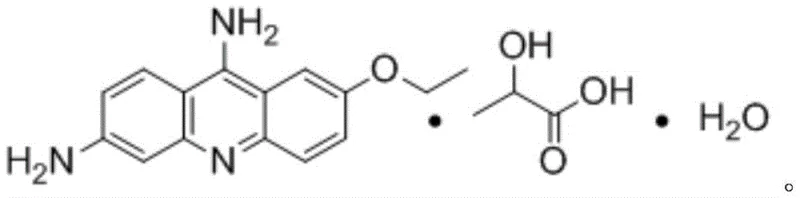
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic efficiency, particularly for established antiseptic agents like Ethacridine Lactate. A pivotal advancement in this domain is detailed in patent CN107954932B, which discloses a novel preparation process that fundamentally restructures the synthesis of this critical active pharmaceutical ingredient. Historically, the production of Ethacridine Lactate has been plagued by the complexities associated with isolating the intermediate 2-ethoxy-6,9-diaminoacridine, a step notorious for poor solubility and difficult purification. This new methodology circumvents these legacy hurdles by integrating the reduction and salt formation steps into a unified operation. By leveraging a direct reductive strategy using zinc powder and lactic acid within an alcoholic solvent system, the process achieves a seamless transformation from the nitro precursor to the final lactate salt. This innovation not only enhances the chemical elegance of the route but also aligns perfectly with modern green chemistry principles by minimizing solvent consumption and waste discharge.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in early German patents, typically rely on a disjointed two-stage sequence where the free base, 2-ethoxy-6,9-diaminoacridine, must be isolated prior to salt formation. This conventional approach presents severe engineering challenges, primarily stemming from the physicochemical properties of the intermediate. The free base exhibits notoriously difficult dissolution characteristics, necessitating the use of excessive volumes of organic solvents to maintain a workable slurry or solution during processing. Furthermore, the isolation step itself is fraught with inefficiencies; separating the solid base from the reaction matrix often requires complex filtration and washing protocols that lead to significant material loss. From an environmental and operational standpoint, this multi-step isolation generates substantial three-waste discharge, including contaminated solvent streams and solid residues that require costly treatment. The cumulative effect of these inefficiencies is a process that is economically burdensome and technically rigid, making it ill-suited for the demands of modern large-scale industrial production where throughput and cost-per-kilogram are paramount metrics.
The Novel Approach
In stark contrast, the process outlined in CN107954932B introduces a paradigm shift by eliminating the intermediate isolation entirely. The novel approach utilizes a "one-pot" or continuous feeding strategy where the reduction of the nitro group and the subsequent formation of the lactate salt occur in the same reaction vessel. By introducing lactic acid directly into the reduction mixture containing 2-ethoxy-6-nitro-9-aminoacridine and a reducing agent like zinc powder, the system facilitates the immediate conversion of the generated amine into its stable lactate salt. This strategic modification drastically simplifies the post-treatment operations; instead of complex extractions or multiple crystallizations, the process merely requires hot filtration to remove metal residues followed by cooling to precipitate the product. The result is a streamlined workflow that significantly shortens the reaction route and improves equipment utilization rates. Moreover, the ability to operate with reduced solvent volumes while maintaining high reaction efficiency demonstrates a sophisticated understanding of process intensification, directly addressing the solubility and purification bottlenecks that have long hindered the efficient manufacture of this compound.
Mechanistic Insights into Zinc-Mediated Reductive Salt Formation
The core chemical transformation driving this process is the reduction of the nitro group at the 6-position of the acridine ring to an amino group, mediated by a metal reducing agent in an acidic alcoholic medium. In this specific embodiment, zinc powder serves as the electron donor, undergoing oxidation to zinc ions while transferring electrons to the nitro substrate. The presence of lactic acid is dual-functional; it acts as the proton source necessary for the reduction mechanism and simultaneously serves as the counter-ion for the final salt formation. As the reduction proceeds under nitrogen protection at temperatures ranging from 40°C to 90°C, the transient diaminoacridine species is instantly protonated by the lactic acid. This immediate protonation prevents the accumulation of the free base, thereby avoiding the precipitation and aggregation issues that complicate traditional methods. The reaction kinetics are carefully balanced to ensure complete conversion of the nitro starting material while minimizing side reactions, such as over-reduction or degradation of the sensitive acridine core. The choice of ethanol or methanol as the solvent further supports this mechanism by providing a polar environment that stabilizes the ionic intermediates and facilitates the solubility of the reactants without requiring excessive volumes.
Impurity control in this system is inherently managed through the physical properties of the product and the simplicity of the workup. Since the reaction does not generate complex organic byproducts typical of multi-step syntheses, the primary impurities are inorganic salts (zinc lactate) and unreacted metal powders. These are effectively removed via the hot filtration step, which exploits the differential solubility of the organic product versus the inorganic residues in the hot alcoholic solution. The subsequent cooling crystallization is highly selective, allowing the bright yellow Ethacridine Lactate crystals to precipitate with high purity, often exceeding 98% without the need for further recrystallization. This high level of selectivity is crucial for pharmaceutical applications, where strict limits on heavy metals and organic impurities must be met. The mechanism ensures that the final product is not only chemically pure but also possesses the correct crystal habit and particle size distribution, which are critical for downstream formulation and bioavailability.
How to Synthesize Ethacridine Lactate Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal conditions to maximize yield and safety. The patent details two operational modes: a one-pot feeding method where all reagents are combined initially, and a continuous feeding method where the acid is added after the reduction phase. Both methods utilize a molar ratio of substrate to reducing agent to lactic acid optimized around 1:3.5:1.1 to ensure complete reaction without excessive reagent waste. The detailed standardized synthesis steps, including specific charging orders and temperature ramps validated for industrial reactors, are provided in the technical guide below.
- Charge 2-ethoxy-6-nitro-9-aminoacridine, zinc powder (reducing agent), and lactic acid into a reactor with ethanol solvent under nitrogen protection.
- Heat the mixture to 40-90°C and maintain reaction for 1-7 hours to ensure complete reduction of the nitro group.
- Filter the hot reaction mixture to remove metal residues, then cool the filtrate naturally to room temperature to precipitate bright yellow Ethacridine Lactate crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible operational improvements that extend far beyond simple chemical yield. The most significant advantage lies in the drastic reduction of raw material and utility costs driven by the elimination of the intermediate isolation step. By removing the need to dry, store, and re-dissolve the 2-ethoxy-6,9-diaminoacridine intermediate, manufacturers can achieve substantial savings in energy consumption and labor hours. Furthermore, the reduction in organic solvent usage is a critical cost driver; since the process operates efficiently with lower solvent-to-substrate ratios, the expenses associated with solvent purchase, recovery, and disposal are significantly curtailed. This leaner material footprint directly enhances the gross margin profile of the final API, making it a more competitive offering in the global marketplace.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis fundamentally alters the cost structure of Ethacridine Lactate production. By consolidating two distinct chemical operations into a single unit operation, the process eliminates the capital and operational expenditures associated with intermediate handling equipment. The avoidance of complex purification sequences means less downtime between batches and higher overall equipment effectiveness (OEE). Additionally, the use of commodity reagents like zinc powder and lactic acid, rather than exotic catalysts, ensures that raw material costs remain stable and predictable. This stability allows for more accurate long-term budgeting and pricing strategies, shielding the supply chain from volatility in specialized reagent markets.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that have multiple points of failure. This simplified route reduces the number of unit operations, thereby decreasing the statistical probability of batch failures or deviations. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent grades, further enhances reliability by reducing sensitivity to minor fluctuations in utility supplies. Moreover, the high yield consistency demonstrated in the patent examples ensures that production planning can be executed with greater confidence, minimizing the need for safety stock and reducing inventory carrying costs. This reliability is essential for maintaining uninterrupted supply to downstream formulators and healthcare providers.
- Scalability and Environmental Compliance: As regulatory pressure regarding environmental discharge intensifies, processes that minimize waste generation become increasingly valuable assets. This method's ability to reduce three-waste discharge by simplifying the workflow directly lowers the cost of environmental compliance and waste treatment. The reduced solvent load also eases the burden on solvent recovery systems, allowing for faster batch turnover. From a scalability perspective, the exothermic nature of the reduction is manageable within standard reactor configurations, and the absence of difficult-to-handle intermediates makes the transition from pilot scale to multi-ton commercial production seamless. This scalability ensures that suppliers can rapidly ramp up production to meet surges in demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific problem-solution dynamics presented in the patent documentation, focusing on the practical implications for industrial manufacturing and quality assurance.
Q: How does this new process improve upon traditional Ethacridine Lactate synthesis?
A: Traditional methods require isolating the free base intermediate (2-ethoxy-6,9-diaminoacridine) before salt formation, which involves difficult dissolution and complex purification. This patented process combines reduction and salt formation in a single pot, eliminating the isolation step and significantly reducing solvent usage and waste generation.
Q: What yields and purity levels can be expected from this method?
A: Industrial validation examples demonstrate total yields consistently exceeding 70%, with product purity reaching above 98%. Specific embodiments have achieved purities as high as 99.5%, making it suitable for high-grade pharmaceutical applications without extensive recrystallization.
Q: Is this process scalable for large-volume production?
A: Yes, the process is specifically designed for scalability. By simplifying the workflow to a one-pot or continuous feeding mode and reducing the volume of organic solvents required, it addresses the bottlenecks of heat transfer and mixing often found in multi-step batch processes, facilitating ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethacridine Lactate Supplier
The technical potential of this direct reductive salt formation route represents a significant opportunity for optimizing the supply chain of essential antiseptic APIs. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to market effectively. Our facility is equipped with rigorous QC labs and stringent purity specifications that ensure every batch of Ethacridine Lactate meets the highest international pharmacopoeia standards. We understand that transitioning to a new process requires a partner who can navigate the complexities of process validation and regulatory filing with precision and speed.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this optimized synthesis can enhance your product portfolio. By collaborating with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant with the evolving landscape of pharmaceutical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
