Advanced Synthesis of Beta-Nicotinamide Mononucleotide for Commercial Pharmaceutical Manufacturing
The pharmaceutical and nutraceutical industries are constantly seeking robust manufacturing pathways for critical bioactive molecules, and Patent CN112724180B represents a significant advancement in the synthesis of Beta-Nicotinamide Mononucleotide (NMN). As a key precursor to NAD+, NMN plays a vital role in cellular energy metabolism and anti-aging research, making its efficient production a priority for global supply chains. This patent discloses a novel chemical preparation method that addresses longstanding challenges in yield, purity, and operational complexity. By utilizing a specific sequence of glycosidation condensation, deprotection, phosphorylation, and deacylation reactions, the invention offers a streamlined alternative to traditional enzymatic or less selective chemical routes. For procurement leaders and R&D directors, understanding the technical nuances of this approach is essential for evaluating potential partnerships with a reliable Pharmaceutical Intermediates supplier capable of delivering high-quality materials at scale.
The significance of this technology extends beyond mere academic interest; it directly impacts the feasibility of commercial scale-up of complex Pharmaceutical Intermediates. Traditional methods often suffer from poor stereoselectivity or require expensive starting materials that hinder cost-effective mass production. In contrast, the methodology outlined in CN112724180B leverages readily available raw materials and optimizes reaction conditions to ensure consistent quality. This report provides a deep technical analysis of the patent's claims, focusing on how the novel intermediate structures and reaction parameters contribute to a more stable and efficient manufacturing process. By adopting such innovative synthetic strategies, manufacturers can achieve substantial cost savings and improve their competitive positioning in the high-purity NMN market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of NMN has been dominated by enzymatic methods and earlier chemical routes, both of which present significant drawbacks for large-scale industrial application. Enzymatic synthesis, while highly specific, often requires expensive substrates like D-5-phosphate ribose or ATP, and the reliance on biocatalysts introduces complexities in enzyme stability, recycling, and removal. Furthermore, the downstream processing for enzymatic routes can be cumbersome, requiring extensive purification to eliminate protein residues and biological impurities. On the chemical side, prior art methods frequently struggle with poor regioselectivity during phosphorylation, leading to a mixture of polyphosphorylated and ectopic phosphorylated impurities that are structurally similar to the target molecule and extremely difficult to separate. These purification challenges not only drive up production costs but also limit the overall yield and purity achievable in a commercial setting.
The Novel Approach
The method disclosed in Patent CN112724180B overcomes these hurdles through a rationally designed chemical pathway that prioritizes selectivity and ease of purification. By introducing novel intermediate structures with specific protecting groups, the invention ensures that reactions occur only at the desired functional sites. For instance, the strategic protection of the primary hydroxyl group in the early stages allows for controlled glycosidation, while subsequent selective deprotection exposes the exact position needed for phosphorylation. This level of control drastically reduces the formation of unwanted byproducts, simplifying the workup procedure to basic washing or standard chromatography. Consequently, this approach facilitates cost reduction in Nutraceutical Ingredients manufacturing by minimizing material loss and reducing the need for complex, multi-step purification processes that plague conventional techniques.
Mechanistic Insights into TMSOTf-Catalyzed Glycosidation and Selective Phosphorylation
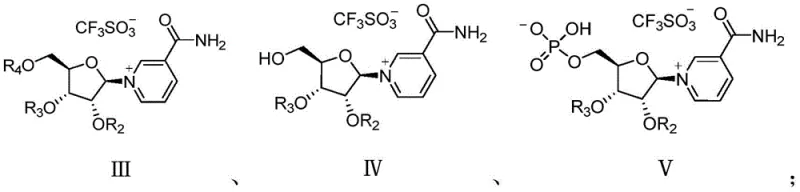
The core of this innovative synthesis lies in the precise manipulation of protecting groups and catalytic conditions to achieve high stereoselectivity and regioselectivity. The process begins with the condensation of a protected ribose derivative (Formula II) with a silylated nicotinamide (Formula VI) in the presence of a Lewis acid catalyst such as trimethylsilyl trifluoromethanesulfonate (TMSOTf). This step forms the critical glycosidic bond, yielding the quaternary ammonium salt intermediate (Formula III). The choice of catalyst and solvent, preferably acetonitrile at temperatures ranging from -10°C to 45°C, is crucial for maximizing the beta-configuration selectivity. Following this, a selective deprotection step removes the bulky silyl group from the primary hydroxyl position without affecting the acyl protections on the secondary hydroxyls. This generates intermediate Formula IV, which is uniquely poised for the subsequent phosphorylation step.
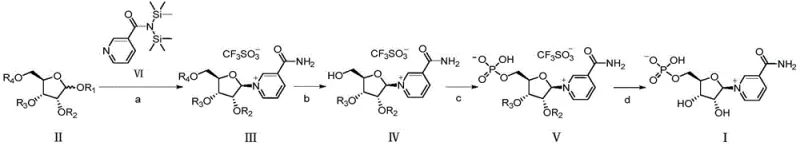
The phosphorylation of intermediate Formula IV is where the true elegance of this patent shines. By having only the primary hydroxyl group free, the reaction with phosphorus oxychloride proceeds with exceptional specificity, avoiding the formation of the troublesome polyphosphorylated impurities seen in older methods. The resulting phosphate intermediate (Formula V) retains the acyl protecting groups, which stabilize the molecule during the phosphorylation event. Finally, a mild deacylation step removes these remaining groups to reveal the final Beta-Nicotinamide Mononucleotide (Formula I). The structural integrity of the novel intermediates, particularly Formulas III, IV, and V, is key to this success, as they allow for easy monitoring and purification at each stage. This mechanistic clarity provides R&D teams with confidence in the reproducibility and robustness of the process for high-purity NMN production.
How to Synthesize Beta-Nicotinamide Mononucleotide Efficiently
Implementing this synthesis route requires careful attention to reaction parameters and reagent quality to ensure optimal yields and purity profiles. The patent outlines a clear four-step sequence that transforms simple starting materials into the complex NMN structure through a series of controlled chemical transformations. Operators must maintain strict control over temperature and molar ratios, particularly during the glycosidation and phosphorylation steps, to prevent side reactions. The use of specific solvents like acetonitrile and tetrahydrofuran is recommended to solubilize intermediates effectively while maintaining reaction stability. Detailed standard operating procedures regarding reagent addition rates and quenching methods are essential for safety and consistency. For a comprehensive guide on the standardized synthesis steps, please refer to the technical documentation provided below.
- Condensation Reaction: React protected ribose derivative (Formula II) with silylated nicotinamide (Formula VI) using TMSOTf catalyst to form the glycosidic bond (Formula III).
- Deprotection: Remove the primary hydroxyl protecting group from Formula III using a fluoride source to yield Formula IV, exposing the site for phosphorylation.
- Phosphorylation: React Formula IV with phosphorus oxychloride to selectively phosphorylate the primary hydroxyl group, forming the phosphate ester intermediate (Formula V).
- Final Deprotection: Remove acyl protecting groups from Formula V using alkaline conditions to obtain the final Beta-Nicotinamide Mononucleotide (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers compelling advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for NMN. The primary benefit lies in the significant simplification of the manufacturing process, which directly correlates to improved operational efficiency and reduced production timelines. By eliminating the need for expensive enzymes and complex bioprocessing equipment, manufacturers can lower their capital expenditure and operational overhead. Furthermore, the use of readily available chemical raw materials mitigates the risk of supply disruptions often associated with specialized biological reagents. This stability is crucial for maintaining continuous production schedules and meeting the growing global demand for NAD+ precursors without facing the volatility typical of enzyme-dependent supply chains.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the elimination of costly biocatalysts and the reduction of purification steps. Traditional enzymatic methods often incur high expenses related to enzyme procurement, immobilization, and disposal, alongside significant losses during downstream processing to remove biological impurities. In contrast, this chemical method utilizes standard reagents and allows for simpler workup procedures, such as aqueous washing, to remove salts and byproducts. The high selectivity of the phosphorylation step means less material is wasted on unusable impurities, leading to better overall mass balance. These factors combine to deliver substantial cost savings, making the final product more competitive in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on stable, commercially available chemical starting materials rather than perishable or hard-to-source biological components. Enzymes can suffer from batch-to-batch variability and require cold chain logistics, whereas the reagents used in this patent, such as protected ribose derivatives and phosphorus oxychloride, are stable and widely produced. This shift reduces the lead time for high-purity Nutraceutical Ingredients by streamlining the procurement process and minimizing the risk of production halts due to raw material shortages. Additionally, the robustness of the chemical process allows for more flexible manufacturing planning, enabling suppliers to respond more quickly to fluctuations in market demand.
- Scalability and Environmental Compliance: Scaling chemical processes is generally more straightforward than scaling biocatalytic ones, as it does not require the maintenance of sterile conditions or complex fermentation infrastructure. The reaction conditions described in the patent, such as moderate temperatures and standard pressure, are easily adaptable to large-scale reactors used in fine chemical manufacturing. Moreover, the simplified purification process reduces the volume of organic solvents and waste generated per kilogram of product. The ability to remove impurities through washing rather than extensive chromatography lowers the environmental footprint of the manufacturing process. This aligns with increasing regulatory pressures for greener chemistry practices and ensures long-term compliance with environmental standards in major pharmaceutical markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NMN synthesis technology. They are derived from the specific technical advantages and problem-solving capabilities detailed in Patent CN112724180B. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this method into their existing production frameworks. The answers reflect the patent's emphasis on purity, selectivity, and operational simplicity, providing clarity on how this approach differentiates itself from legacy methods. For further technical specifications or custom feasibility studies, direct consultation with our engineering team is recommended.
Q: How does this new chemical synthesis method improve impurity control compared to enzymatic routes?
A: Unlike enzymatic methods that often require complex downstream processing to remove proteins and enzyme residues, this chemical route utilizes distinct protection and deprotection strategies. The intermediates formed are structurally unique and allow for easy purification via standard washing or chromatography, significantly reducing the burden of removing water-soluble salts and polyphosphorylated byproducts common in older chemical methods.
Q: What are the supply chain advantages of using this specific NMN preparation method?
A: This method relies on readily available chemical raw materials rather than expensive, biologically derived enzymes or costly precursors like Coenzyme I. The operational simplicity, including mild reaction temperatures and standard solvent systems, facilitates easier commercial scale-up. This translates to enhanced supply chain reliability and reduced dependency on specialized biocatalyst suppliers.
Q: Is the phosphorylation step in this patent selective enough for high-purity production?
A: Yes, the patent specifically highlights high phosphorylation selectivity. By strategically protecting secondary hydroxyl groups while leaving the primary hydroxyl exposed in intermediate Formula IV, the reaction with phosphorus oxychloride targets only the desired position. This minimizes the formation of ectopic phosphorylated impurities, which are notoriously difficult to separate in conventional synthesis routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nicotinamide Mononucleotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthesis technologies like the one described in Patent CN112724180B for the future of NMN production. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards. We understand that consistency is key for pharmaceutical and nutraceutical clients, and our infrastructure is designed to deliver high-purity NMN with reliable impurity profiles that meet the demanding requirements of global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. By partnering with us, you gain access to not just a supplier, but a strategic ally capable of navigating the complexities of fine chemical manufacturing. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain efficiency and product competitiveness in the rapidly evolving NAD+ market.
