Revolutionizing NMN Production: A Deep Dive into the Novel PNP-NRK Enzymatic Cascade
The global demand for Beta-Nicotinamide Mononucleotide (NMN), a critical precursor for NAD+ biosynthesis, has surged due to its potential applications in anti-aging therapeutics and metabolic health. However, scalable manufacturing has long been bottlenecked by the limitations of chemical synthesis and early-generation biocatalytic routes. Patent CN112795606A introduces a transformative enzymatic synthesis method that addresses these historical inefficiencies. By leveraging a dual-enzyme system comprising Purine Nucleoside Phosphorylase (PNP) and Nicotinamide Ribokinase (NRK), this technology enables the conversion of inexpensive adenosine and nicotinamide into high-purity NMN. The process is distinguished by its use of an irreversible final reaction step and an integrated ATP regeneration cycle, offering a robust pathway for reliable pharmaceutical intermediate supplier operations seeking to optimize both yield and cost structures in the nutraceutical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of NMN has relied heavily on chemical synthesis or first-generation enzymatic processes that suffer from significant thermodynamic and economic drawbacks. Chemical routes often involve complex protection and deprotection steps, requiring hazardous solvents and generating substantial toxic waste, which complicates regulatory approval for pharmaceutical grades. On the biocatalytic front, existing methods typically utilize Nicotinamide Phosphoribosyltransferase (NAMPT) to condense nicotinamide with PRPP (Phosphoribosyl Pyrophosphate). This approach is fundamentally flawed because the NAMPT-catalyzed reaction is reversible, creating an equilibrium that limits substrate conversion and necessitates expensive downstream purification to remove unreacted starting materials. Furthermore, PRPP is chemically unstable under physiological conditions, leading to spontaneous hydrolysis that wastes resources and lowers overall process efficiency. Additionally, these conventional bio-routes often consume stoichiometric amounts of ATP, a costly reagent, without an efficient regeneration mechanism, rendering the cost of goods sold prohibitively high for mass-market applications.
The Novel Approach
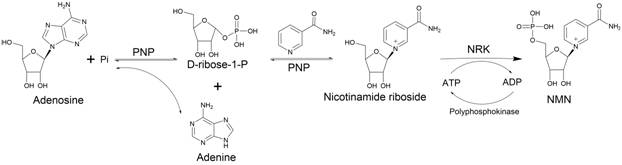
The methodology disclosed in the patent represents a paradigm shift by decoupling the ribose donation and phosphorylation steps into a highly efficient cascade. Instead of relying on unstable PRPP, the process utilizes PNP to cleave adenosine, generating D-ribose-1-phosphate and adenine. This ribose phosphate is then immediately utilized by the same PNP enzyme to ribosylate nicotinamide, forming nicotinamide riboside. Crucially, the final step employs NRK to phosphorylate this intermediate into NMN. Unlike the reversible NAMPT reaction, the NRK-catalyzed phosphorylation is effectively irreversible under the specified conditions, driving the reaction to near-completion with conversion rates reported between 90% and 99%. This novel approach not only stabilizes the reaction intermediates but also simplifies the purification workflow, as the byproduct adenine can be easily separated and potentially recycled, thereby enhancing the sustainability profile of the manufacturing process.
Mechanistic Insights into the PNP-NRK Catalytic Cascade
The core innovation lies in the synergistic interaction between the PNP and NRK enzymes within a single reaction vessel. The mechanism initiates with the phosphorolysis of adenosine by PNP in the presence of inorganic phosphate, yielding D-ribose-1-phosphate and free adenine. This step is reversible, but the equilibrium is pulled forward by the subsequent consumption of the ribose phosphate. The PNP enzyme then catalyzes the transglycosylation of the D-ribose-1-phosphate to nicotinamide, producing nicotinamide riboside. This intermediate serves as the specific substrate for the NRK enzyme. In the presence of magnesium or manganese ions, NRK transfers a phosphate group from ATP to the 5'-hydroxyl of the ribose moiety. The brilliance of this design is the thermodynamic drive; while the initial steps may approach equilibrium, the final phosphorylation by NRK acts as a sink, preventing the reverse reaction and ensuring high accumulation of the target NMN molecule. This cascade minimizes the formation of side products and ensures that the chiral integrity of the ribose sugar is maintained throughout the synthesis.
A critical component of this mechanistic framework is the ATP regeneration loop, which is essential for economic viability at scale. The phosphorylation step consumes ATP, converting it to ADP. In traditional setups, this would require the continuous addition of fresh ATP. However, this patent integrates polyphosphate kinase into the system. This auxiliary enzyme utilizes inexpensive polyphosphate chains to phosphorylate ADP back into ATP in situ. This creates a catalytic cycle where ATP acts as a cofactor rather than a stoichiometric reactant. Consequently, the process requires only a minimal initial charge of ATP, while the polyphosphate serves as the bulk phosphate donor. This regeneration capability significantly lowers the variable cost of production and reduces the ionic load in the reaction mixture, simplifying the subsequent crystallization and purification of the final high-purity NMN product.
How to Synthesize Beta-Nicotinamide Mononucleotide Efficiently
The implementation of this enzymatic route requires precise control over reaction parameters to maximize the activity of the recombinant enzymes. The patent outlines a procedure where host cells, such as E. coli expressing the specific PNP and NRK sequences, are cultured and lysed to provide the crude enzyme source, although purified or immobilized forms are also viable for continuous processing. The reaction is typically conducted in a buffered aqueous solution, such as Tris or PBS, maintained at a pH between 7.0 and 8.5 to optimize enzyme stability. Temperature control is equally vital, with optimal activity observed between 35°C and 45°C. To initiate the synthesis, adenosine and nicotinamide are introduced at equimolar or near-equimolar concentrations, alongside a catalytic amount of ATP and a molar excess of polyphosphate. The detailed standardized synthesis steps, including specific enzyme loading ratios and downstream purification protocols, are provided in the guide below.
- Prepare the reaction system by mixing adenosine and phosphate substrates with Purine Nucleoside Phosphorylase (PNP) to generate D-ribose-1-phosphate and adenine.
- Introduce nicotinamide to the system where PNP catalyzes the formation of nicotinamide riboside intermediate from the ribose-1-phosphate.
- Add Nicotinamide Ribokinase (NRK) and a catalytic amount of ATP, supported by polyphosphate kinase for ATP regeneration, to irreversibly phosphorylate the intermediate into final NMN product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this specific enzymatic platform offers tangible strategic benefits beyond mere technical feasibility. The primary value driver is the drastic reduction in raw material costs associated with the ATP regeneration system. By eliminating the need for stoichiometric ATP, the process removes one of the most expensive line items from the bill of materials. Furthermore, the use of adenosine as a starting material leverages a widely available and cost-effective commodity chemical, unlike nicotinamide riboside which is itself a high-value finished product in other contexts. This shift in substrate strategy ensures a more stable and predictable supply chain, insulating production from the volatility of niche precursor markets. The ability to recover adenine as a high-purity byproduct further enhances the economic model, potentially allowing for the internal reuse of this base in other synthesis pathways or its sale as a co-product.
- Cost Reduction in Manufacturing: The integration of polyphosphate kinase for ATP recycling fundamentally alters the cost structure of NMN production. Since ATP is no longer consumed in a 1:1 ratio with the product, the expense per kilogram of NMN is significantly lowered. Additionally, the high conversion rate of 90% to 99% minimizes the loss of valuable starting materials like nicotinamide and adenosine. The process operates in an aqueous environment without the need for organic solvents or heavy metal catalysts, which reduces the costs associated with solvent recovery, waste disposal, and environmental compliance. The elimination of complex protection groups required in chemical synthesis further streamlines the operation, reducing both material and labor costs involved in multi-step transformations.
- Enhanced Supply Chain Reliability: Reliance on unstable intermediates like PRPP in older methods often leads to batch-to-batch variability and supply disruptions. This new method utilizes stable substrates (adenosine and nicotinamide) that are commercially available in bulk quantities from multiple global suppliers. The robustness of the PNP and NRK enzymes, which can be immobilized for repeated use as demonstrated in the patent examples, ensures consistent production capacity. This stability allows for better long-term planning and contract fulfillment, reducing the risk of stockouts for downstream formulators. The simplified reaction setup also means that manufacturing can be scaled up with less specialized equipment, broadening the base of potential contract manufacturing organizations capable of executing the process.
- Scalability and Environmental Compliance: The biocatalytic nature of this synthesis aligns perfectly with modern green chemistry principles, a critical factor for regulatory approval in major markets like the EU and North America. The process generates minimal hazardous waste, primarily consisting of biodegradable enzyme proteins and benign phosphate salts. This reduces the burden on wastewater treatment facilities and lowers the environmental fees associated with chemical manufacturing. Scalability is facilitated by the ability to use whole-cell catalysis or immobilized enzymes, which are ideal for large-scale bioreactors. The high purity of the crude product (>99% in some examples) reduces the load on downstream chromatography columns, extending their lifespan and reducing the frequency of resin replacement, which is a significant operational expense in large-scale purification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented enzymatic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning pilot runs and procurement teams negotiating raw material contracts.
Q: How does the new enzymatic method improve substrate conversion compared to traditional PRPP routes?
A: Unlike traditional methods relying on Nicotinamide Phosphoribosyltransferase which catalyze a reversible reaction limiting yield, this novel PNP-NRK cascade drives the final phosphorylation step irreversibly. This thermodynamic advantage, combined with the stability of D-ribose-1-phosphate intermediates over unstable PRPP, allows substrate conversion rates to reach between 90% and 99%.
Q: What is the economic advantage of the ATP regeneration system described in the patent?
A: Traditional biocatalytic routes often require stoichiometric amounts of expensive ATP. This process utilizes a polyphosphate kinase system to regenerate ATP from ADP in situ. This means only a catalytic amount of ATP is initially required, drastically reducing the raw material cost per kilogram of NMN produced while maintaining high reaction kinetics.
Q: Can the byproduct adenine be utilized to further offset production costs?
A: Yes, the phosphorolysis of adenosine by PNP generates adenine as a stoichiometric byproduct. The patent explicitly notes that this adenine can be recovered and recycled. In an industrial setting, recovering high-purity adenine adds a secondary revenue stream or reduces the net cost of the adenosine starting material, enhancing the overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nicotinamide Mononucleotide Supplier
The technological advancements detailed in patent CN112795606A underscore the rapid evolution of NMN manufacturing towards more sustainable and cost-effective biocatalytic solutions. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such complex enzymatic pathways into commercial reality. Our facility is equipped with state-of-the-art fermentation and downstream processing capabilities, ensuring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of NMN meets the highest pharmacopoeial standards, ensuring safety and efficacy for your end consumers.
We invite you to collaborate with us to leverage this next-generation synthesis route for your product lines. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this enzymatic method can optimize your margin structure. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a stable, high-quality supply of NMN that drives your business forward in the competitive nutraceutical marketplace.
