Scalable Biocatalytic Production of High-Purity Chiral Trifluoroethanol Intermediates for Global Pharma
Scalable Biocatalytic Production of High-Purity Chiral Trifluoroethanol Intermediates for Global Pharma
The pharmaceutical industry is constantly seeking more efficient, sustainable, and cost-effective routes for synthesizing chiral building blocks, particularly fluorinated intermediates which are ubiquitous in modern drug design. A significant breakthrough in this domain is detailed in Chinese Patent CN113462728A, which discloses a novel method for preparing (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol using the yeast strain Cyberlindnera saturnus ZJPH1807. This biocatalytic approach represents a paradigm shift from traditional chemical synthesis, offering a pathway that combines high stereoselectivity with environmentally benign processing conditions. The core innovation lies in the utilization of whole resting cells as biocatalysts, which not only simplifies the operational workflow but also ensures robust cofactor regeneration without the need for exogenous addition of expensive nicotinamide coenzymes.
This specific chiral alcohol serves as a critical precursor for potent therapeutic agents, including Cathepsin K inhibitors such as Orobacate (MK-0822) and Tryptophan Hydroxylase inhibitors like LX-1031. 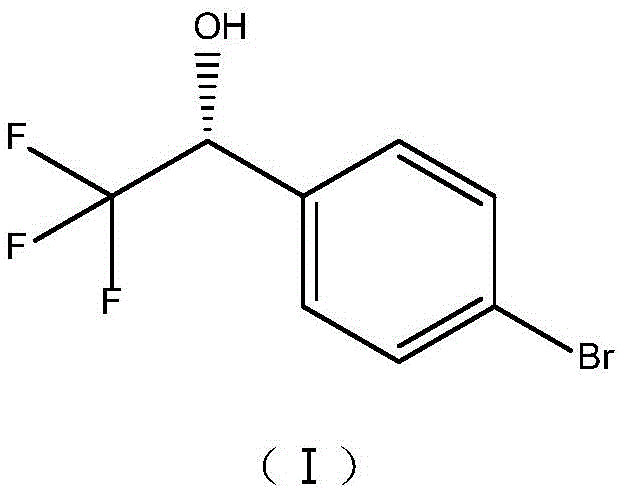 The ability to produce this intermediate with high optical purity is paramount, as the biological activity of the final APIs is strictly dependent on the stereochemistry of this building block. By leveraging the unique metabolic capabilities of the ZJPH1807 strain, manufacturers can achieve enantiomeric excess values exceeding 94% under optimized conditions, thereby securing a reliable supply of high-quality material for downstream coupling reactions.
The ability to produce this intermediate with high optical purity is paramount, as the biological activity of the final APIs is strictly dependent on the stereochemistry of this building block. By leveraging the unique metabolic capabilities of the ZJPH1807 strain, manufacturers can achieve enantiomeric excess values exceeding 94% under optimized conditions, thereby securing a reliable supply of high-quality material for downstream coupling reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral trifluoroethanols has relied heavily on chemical asymmetric reduction or resolution techniques, both of which present substantial drawbacks for large-scale manufacturing. Chemical methods often necessitate the use of precious metal catalysts, such as ruthenium or rhodium complexes, which are not only prohibitively expensive but also pose significant challenges regarding residual metal contamination in the final product. Strict regulatory limits on heavy metals in pharmaceuticals require additional, costly purification steps to ensure compliance. Furthermore, these chemical processes frequently demand harsh reaction conditions, including high pressures of hydrogen gas and extreme temperatures, which escalate energy consumption and introduce safety hazards related to flammability and explosion risks in an industrial setting.
Even within the realm of biocatalysis, earlier approaches often utilized isolated enzymes (such as Alcohol Dehydrogenases) rather than whole cells. While effective, the isolation and purification of enzymes add considerable complexity and cost to the production process. Isolated enzymes are also inherently unstable outside their native cellular environment and require the continuous addition of expensive cofactors like NADH or NADPH to sustain catalytic turnover. Without an efficient regeneration system, the stoichiometric requirement for these cofactors renders the process economically unviable for commercial scale-up, limiting the utility of such methods to small-scale laboratory synthesis rather than industrial production.
The Novel Approach
The methodology described in patent CN113462728A overcomes these hurdles by employing Cyberlindnera saturnus ZJPH1807 whole cells as a self-contained biocatalytic factory.  In this system, the yeast cells provide a protective matrix for the enzymes, enhancing their stability and operational lifetime without the need for complex immobilization techniques. Crucially, the whole-cell system facilitates in-situ cofactor regeneration; the cellular metabolism naturally recycles the necessary reducing equivalents using simple, inexpensive co-substrates like methanol. This eliminates the need for external cofactor addition and complex enzyme coupling systems, drastically simplifying the reaction setup. The process operates under mild aqueous conditions (30°C, neutral pH), significantly reducing energy inputs and eliminating the safety risks associated with high-pressure hydrogenation.
In this system, the yeast cells provide a protective matrix for the enzymes, enhancing their stability and operational lifetime without the need for complex immobilization techniques. Crucially, the whole-cell system facilitates in-situ cofactor regeneration; the cellular metabolism naturally recycles the necessary reducing equivalents using simple, inexpensive co-substrates like methanol. This eliminates the need for external cofactor addition and complex enzyme coupling systems, drastically simplifying the reaction setup. The process operates under mild aqueous conditions (30°C, neutral pH), significantly reducing energy inputs and eliminating the safety risks associated with high-pressure hydrogenation.
Mechanistic Insights into Whole-Cell Asymmetric Reduction
The core of this technology relies on the stereospecific activity of intracellular alcohol dehydrogenases (ADHs) present within the Cyberlindnera saturnus ZJPH1807 strain. These enzymes catalyze the hydride transfer from the reduced cofactor (NADPH) to the prochiral ketone substrate, 4'-bromo-2,2,2-trifluoroacetophenone. The active site of the ADH is sterically constrained, allowing it to discriminate between the two faces of the carbonyl group, thereby ensuring the exclusive formation of the (R)-enantiomer. The presence of the trifluoromethyl group adds electronic complexity to the substrate, yet the enzyme demonstrates remarkable tolerance and selectivity, achieving e.e. values up to 94.08%. This high fidelity is essential for preventing the formation of the unwanted (S)-isomer, which could act as a competitive inhibitor or toxic impurity in the final drug product.
A critical component of the mechanism is the efficient regeneration of the NADPH cofactor, which is consumed during the reduction of the ketone. In this whole-cell system, the addition of methanol as a co-substrate triggers the activity of endogenous methanol dehydrogenases or oxidases within the yeast. These enzymes oxidize methanol to formaldehyde (and subsequently formic acid), simultaneously reducing NADP+ back to NADPH. This creates a closed catalytic cycle where the cofactor is continuously recycled, allowing a catalytic amount of intracellular NADPH to drive the conversion of a large molar excess of substrate. Optimization studies revealed that a methanol concentration of 10% (v/v) provides the optimal balance between driving the regeneration cycle and maintaining cell viability, ensuring sustained catalytic activity over the 24-hour reaction period.
How to Synthesize (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol Efficiently
The synthesis protocol outlined in the patent offers a robust framework for producing this valuable intermediate with high consistency. The process begins with the fermentation of the specific yeast strain to generate a high density of wet biomass, which serves as the source of the biocatalyst. The transformation is conducted in a buffered aqueous medium to maintain physiological pH, ensuring maximum enzyme activity. The reaction parameters, including temperature, agitation speed, and substrate loading, have been meticulously optimized to maximize both yield and optical purity. For detailed operational parameters and step-by-step instructions on media preparation and workup procedures, please refer to the standardized guide below.
- Ferment Cyberlindnera saturnus ZJPH1807 to obtain wet thalli, then resuspend in 0.1M phosphate buffer (pH 7.0).
- Add 4'-bromo-2,2,2-trifluoroacetophenone substrate and 10% (v/v) methanol as a co-substrate for cofactor regeneration.
- Incubate at 30°C and 200rpm for 24 hours, then extract with ethyl acetate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chemical synthesis to this whole-cell biocatalytic route offers compelling economic and logistical benefits. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing expensive transition metal catalysts and stoichiometric reducing agents (like borohydrides) with renewable yeast biomass and methanol, the direct material costs are significantly reduced. Furthermore, the elimination of heavy metals removes the need for specialized scavenging resins and extensive analytical testing for metal residues, streamlining the quality control workflow and reducing the overall cost of goods sold (COGS) for the intermediate.
- Cost Reduction in Manufacturing: The use of whole cells negates the capital expenditure and operational costs associated with enzyme purification facilities. Since the cells are used directly after fermentation and washing, the process bypasses the low-yield extraction and chromatography steps typically required to isolate pure enzymes. Additionally, the in-situ cofactor regeneration means that expensive nicotinamide coenzymes do not need to be purchased in bulk, representing a substantial saving in reagent costs that scales linearly with production volume.
- Enhanced Supply Chain Reliability: Biological catalysts are derived from renewable fermentation processes, insulating the supply chain from the geopolitical volatility often associated with the mining and refining of precious metals like platinum or palladium. The robustness of the yeast strain allows for the production of stable, storable wet cake or lyophilized powder, enabling manufacturers to build strategic inventory buffers. This reliability ensures consistent availability of the chiral intermediate, mitigating the risk of production stoppages for downstream API manufacturers due to raw material shortages.
- Scalability and Environmental Compliance: The reaction conditions are inherently green, utilizing water as the primary solvent and operating at ambient pressure. This simplifies the engineering requirements for reactor vessels, allowing for easier scale-up from pilot to commercial scale without the need for specialized high-pressure equipment. From an environmental perspective, the process generates significantly less hazardous waste compared to chemical reduction, reducing the burden on wastewater treatment facilities and lowering the costs associated with environmental compliance and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear picture of the process capabilities and limitations for potential adopters.
Q: What are the advantages of using whole-cell catalysis over purified enzymes for this intermediate?
A: Whole-cell catalysis eliminates the costly and complex steps of enzyme separation and purification. Furthermore, the cellular machinery allows for in-situ regeneration of expensive cofactors (like NADPH) using simple co-substrates like methanol, drastically reducing raw material costs compared to isolated enzyme systems.
Q: What is the optical purity achievable with the Cyberlindnera saturnus ZJPH1807 strain?
A: Under optimized conditions (substrate concentration 1.25 g/L, cell concentration 350 g/L, pH 7.0), the process achieves an enantiomeric excess (e.e.) value of up to 94.08%, which is critical for meeting the stringent stereochemical requirements of downstream API synthesis.
Q: Is this biocatalytic method scalable for industrial production?
A: Yes, the method utilizes robust fermentation-derived wet cells and mild reaction conditions (30°C, atmospheric pressure), making it highly amenable to scale-up. The avoidance of harsh chemicals and expensive metal catalysts simplifies waste treatment and enhances environmental compliance for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral chromatography systems to guarantee that every batch of (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol meets the exacting standards required for API synthesis. Our commitment to quality ensures that your downstream processes remain uninterrupted and compliant with global regulatory frameworks.
We invite you to collaborate with us to leverage this advanced biocatalytic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact us today to request specific COA data for our current inventory or to discuss route feasibility assessments for your custom synthesis projects, ensuring a secure and efficient supply chain for your critical pharmaceutical ingredients.
