Scalable Biocatalytic Synthesis of Chiral Trifluoroethanol Intermediates for Osteoporosis Drugs
The pharmaceutical industry continuously seeks robust and sustainable pathways for synthesizing chiral intermediates, particularly those serving as critical building blocks for potent therapeutic agents. Patent CN113462728A discloses a groundbreaking method for preparing (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol, a pivotal chiral alcohol intermediate, utilizing the yeast strain Cyberlindnera saturnus ZJPH1807. This biocatalytic approach represents a significant leap forward in the manufacturing of key precursors for Cathepsin K inhibitors such as Odanacatib and Tryptophan Hydroxylase inhibitors like LX-1031. The strategic application of whole-cell biocatalysis not only addresses the stringent stereochemical requirements of modern drug design but also aligns with green chemistry principles by operating under mild physiological conditions. By leveraging the intrinsic enzymatic machinery of this specific yeast strain, the process achieves high stereoselectivity without the need for complex enzyme isolation, offering a streamlined solution for the production of high-purity pharmaceutical intermediates.
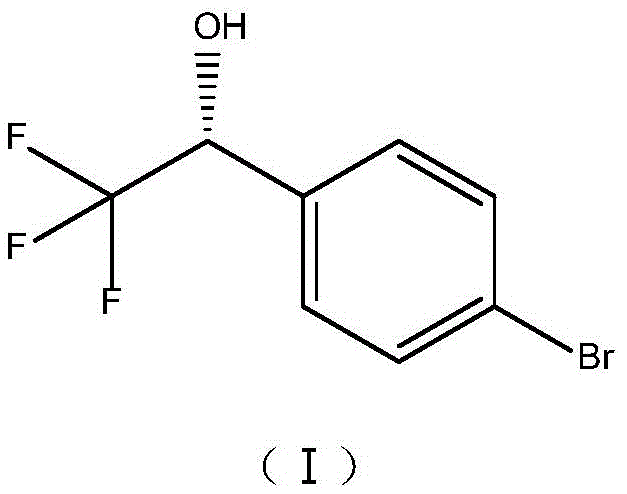
The significance of this intermediate is underscored by its role in the synthesis of advanced therapeutics targeting bone metabolism and gastrointestinal disorders. For instance, Odanacatib, a Cathepsin K inhibitor developed for postmenopausal osteoporosis, relies on the precise stereochemistry provided by this trifluoroethanol scaffold to effectively inhibit bone resorption while preserving bone formation. Similarly, LX-1031 utilizes this chiral motif to selectively inhibit TPH1 in the gastrointestinal tract, offering a targeted treatment for Irritable Bowel Syndrome with minimized central nervous system side effects. The ability to produce this specific (R)-enantiomer with high fidelity is therefore not merely a chemical exercise but a critical supply chain imperative for developing these next-generation medicines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral fluorinated alcohols has relied heavily on chemical reduction methods employing stoichiometric amounts of chiral auxiliaries or expensive transition metal catalysts. These conventional chemical routes often necessitate harsh reaction conditions, including extreme temperatures and pressures, which can compromise the integrity of sensitive functional groups and lead to the formation of difficult-to-remove impurities. Furthermore, the reliance on precious metal catalysts introduces significant cost volatility and environmental burdens associated with heavy metal waste disposal and removal. In the context of biological methods, earlier approaches utilizing purified alcohol dehydrogenases (ADH) required intricate and costly downstream processing to isolate the enzymes, alongside the continuous addition of expensive cofactors like NADPH to sustain catalytic activity. These factors collectively inflate the cost of goods sold (COGS) and complicate the regulatory approval process due to the potential for metal contamination and complex impurity profiles.
The Novel Approach
The innovative methodology described in the patent circumvents these historical bottlenecks by employing resting cells of Cyberlindnera saturnus ZJPH1807 as a self-contained biocatalytic factory. This whole-cell system inherently possesses the necessary cofactor regeneration mechanisms, thereby eliminating the external addition of costly nicotinamide cofactors and simplifying the reaction setup to a single pot. The process operates at a mild temperature of 30°C and neutral pH, drastically reducing energy consumption and equipment corrosion risks compared to thermal chemical reductions. By optimizing the co-substrate system, specifically utilizing methanol to drive the redox balance, the method achieves exceptional conversion rates and stereoselectivity. This shift from isolated enzyme systems or harsh chemical catalysis to a robust whole-cell platform fundamentally alters the economic and operational landscape of producing this high-value intermediate, making it accessible for large-scale commercial adoption.
Mechanistic Insights into Whole-Cell Asymmetric Reduction
The core of this technology lies in the stereospecific reduction of the prochiral ketone, 4'-bromo-2,2,2-trifluoroacetophenone, to the corresponding (R)-alcohol. Within the cellular environment of Cyberlindnera saturnus, specific oxidoreductases recognize the substrate and facilitate the hydride transfer from the reduced cofactor (NADPH) to the carbonyl carbon with high spatial precision. The presence of the trifluoromethyl group adjacent to the carbonyl creates significant steric and electronic challenges, yet the enzyme active site accommodates this moiety effectively to favor the formation of the (R)-enantiomer. The internal metabolic pathways of the yeast efficiently regenerate the oxidized cofactor (NADP+) back to its reduced form using the added co-substrate, creating a sustainable catalytic cycle that persists throughout the reaction duration. This intracellular cofactor recycling is the key to the process's efficiency, allowing for high turnover numbers without the accumulation of inhibitory byproducts that often plague in vitro enzyme systems.
Impurity control is inherently superior in this biocatalytic regime due to the enzyme's exquisite substrate specificity. Unlike chemical reducers which may attack other reducible groups or cause racemization, the biocatalyst selectively targets the ketone functionality. The patent data indicates that under optimized conditions, the enantiomeric excess (e.e.) reaches values as high as 94.08%, with yields approaching 75% at practical substrate loadings. This high optical purity minimizes the need for extensive chiral resolution steps downstream, such as recrystallization or chiral chromatography, which are typically yield-loss intensive. The resulting product profile is clean, with the primary impurity being the unreacted starting material, which can be easily separated via standard extraction and chromatography techniques, ensuring the final intermediate meets the rigorous quality standards demanded by global pharmaceutical regulators.
How to Synthesize (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-performance biotransformation. The process begins with the cultivation of the ZJPH1807 strain to generate sufficient biomass, followed by the preparation of the reaction mixture where substrate concentration and co-substrate ratios are critical parameters. Methanol serves as the optimal co-substrate, driving the equilibrium towards product formation while maintaining cell viability. The detailed standardized synthetic steps below outline the precise fermentation conditions, reaction parameters, and workup procedures required to achieve the reported yields and purity levels.
- Cultivate Cyberlindnera saturnus ZJPH1807 strain on slant and seed media, followed by fermentation to obtain wet thalli as the enzyme source.
- Suspend wet cells in 0.1M phosphate buffer (pH 7.0) containing 4'-bromo-2,2,2-trifluoroacetophenone substrate and methanol as a co-substrate.
- Incubate the reaction mixture at 30°C and 200rpm for 24 hours, then extract with ethyl acetate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this biocatalytic platform offers tangible strategic benefits that extend beyond simple technical feasibility. The elimination of expensive transition metal catalysts and chiral ligands directly translates to a significant reduction in raw material costs, insulating the supply chain from the volatility of the precious metals market. Furthermore, the simplified downstream processing—requiring only extraction and standard chromatography rather than complex metal scavenging or chiral resolution—reduces the overall processing time and solvent consumption. This streamlining of the manufacturing workflow enhances the reliability of supply by reducing the number of unit operations where delays or failures can occur, ensuring a more consistent flow of critical intermediates to API manufacturing sites.
- Cost Reduction in Manufacturing: The utilization of whole-cell biocatalysis removes the substantial capital and operational expenditures associated with enzyme purification and cofactor supplementation. By leveraging the cell's natural metabolic machinery for cofactor regeneration, the process avoids the recurring cost of purchasing NADPH or glucose dehydrogenase systems. Additionally, the mild reaction conditions reduce energy costs related to heating and cooling, while the high stereoselectivity minimizes the loss of material during purification, collectively driving down the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the Cyberlindnera saturnus strain ensures consistent batch-to-batch performance, a critical factor for maintaining uninterrupted API production schedules. The use of readily available and inexpensive co-substrates like methanol further secures the supply chain against raw material shortages. The scalability of fermentation processes is well-established in the industry, allowing for rapid capacity expansion to meet surging demand without the need for specialized high-pressure reactors or exotic catalysts that often have long lead times.
- Scalability and Environmental Compliance: This green chemistry approach aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge and solvent waste. The aqueous nature of the biocatalytic step reduces the reliance on hazardous organic solvents during the reaction phase. The absence of toxic metal residues simplifies waste treatment protocols and lowers the environmental compliance burden, facilitating smoother regulatory approvals and enhancing the sustainability profile of the final drug product, which is a growing priority for global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a transparent view of the process capabilities and limitations for potential adopters and partners evaluating this synthetic route.
Q: What is the stereoselectivity of the Cyberlindnera saturnus biocatalyst?
A: The process achieves an enantiomeric excess (e.e.) value of up to 94.08% for the (R)-enantiomer, ensuring high optical purity required for downstream API synthesis.
Q: Why is methanol preferred as a co-substrate in this reaction?
A: Methanol acts as an efficient hydrogen donor for coenzyme regeneration. Optimization data indicates that a 10% (v/v) addition maximizes yield to 94.85% while maintaining high stereoselectivity.
Q: Can this biocatalytic process be scaled for industrial production?
A: Yes, the use of whole resting cells eliminates the need for expensive enzyme purification and cofactor addition, significantly simplifying the workflow and reducing operational costs for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the successful development of novel therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral chromatography systems to guarantee that every batch of (R)-1-(4-bromophenyl)-2,2,2-trifluoroethanol meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence ensures that your supply chain remains robust and compliant.
We invite you to collaborate with us to leverage this advanced biocatalytic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your overall manufacturing budget. Please contact us to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the delivery of life-saving medications to patients worldwide.
