Advanced Enantioselective Hydrogenation Technology for High-Purity Pharmaceutical Intermediates
Advanced Enantioselective Hydrogenation Technology for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks more efficient pathways to synthesize chiral building blocks, particularly those serving as critical intermediates for central nervous system (CNS) active drugs. Patent CN1678562A introduces a groundbreaking method for the enantioselective preparation of amino alcohols of formula (I), which are pivotal precursors for major antidepressants such as duloxetine, fluoxetine, and atomoxetine. This technology leverages a sophisticated transition metal catalytic system to achieve high stereoselectivity directly from aminoketones, bypassing the inefficiencies of traditional racemic resolution. By utilizing non-racemic catalysts, specifically rhodium complexes coordinated with chiral diphosphine ligands, the process ensures the production of optically active forms with exceptional purity profiles. 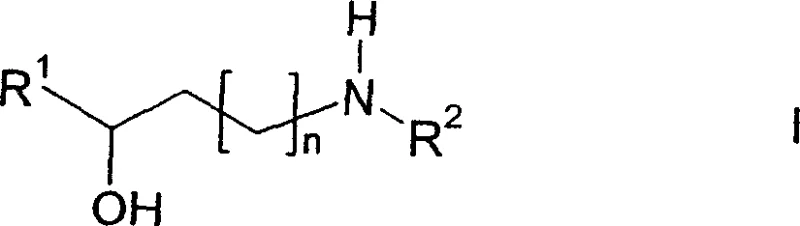 For procurement managers and supply chain directors, this represents a shift towards more sustainable and cost-effective manufacturing of reliable pharmaceutical intermediate supplier materials, reducing waste and enhancing overall process economics.
For procurement managers and supply chain directors, this represents a shift towards more sustainable and cost-effective manufacturing of reliable pharmaceutical intermediate supplier materials, reducing waste and enhancing overall process economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 3-amino-1-aryl-propanols has been plagued by inherent inefficiencies associated with classical resolution techniques. Traditional methods often involve the synthesis of a racemic mixture followed by a resolution step, which theoretically caps the maximum yield of the desired enantiomer at 50%, leaving the other half as waste or requiring complex recycling processes. Furthermore, alternative asymmetric synthesis routes described in prior art frequently rely on the stoichiometric use of expensive chiral reagents or auxiliaries, which drastically increases the cost of goods sold (COGS). Another significant drawback in existing literature involves the use of dimethyl-amino ketones which necessitate a subsequent demethylation step to obtain the target mono-methyl amine; this transformation often employs hazardous reagents and generates toxic byproducts such as methyl chloride, posing severe environmental and safety challenges for large-scale operations. These factors collectively create bottlenecks in cost reduction in API manufacturing and complicate regulatory compliance regarding impurity profiles.
The Novel Approach
The methodology disclosed in CN1678562A offers a transformative solution by enabling the direct enantioselective hydrogenation of mono-alkylated aminoketones. This approach fundamentally alters the economic landscape by allowing for nearly 100% theoretical conversion of the starting material into the desired chiral product, effectively doubling the material efficiency compared to resolution strategies. The process utilizes a catalytic amount of a chiral transition metal complex, eliminating the need for stoichiometric chiral sources and significantly lowering raw material costs. Crucially, the invention demonstrates that elimination side reactions, which are typically problematic in the hydrogenation of beta-amino ketones, are suppressed to negligible levels (less than 2%) under the optimized conditions. This results in a cleaner reaction profile that simplifies downstream purification and enhances the overall robustness of the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Rh-Catalyzed Enantioselective Hydrogenation
The core of this technological breakthrough lies in the precise design of the catalytic system, which employs a transition metal, preferably rhodium, complexed with specific chiral diphosphine ligands. The patent highlights the efficacy of ligands falling under Formula A and Formula B, where the steric and electronic properties of the phosphine groups dictate the stereochemical outcome of the hydrogen transfer. 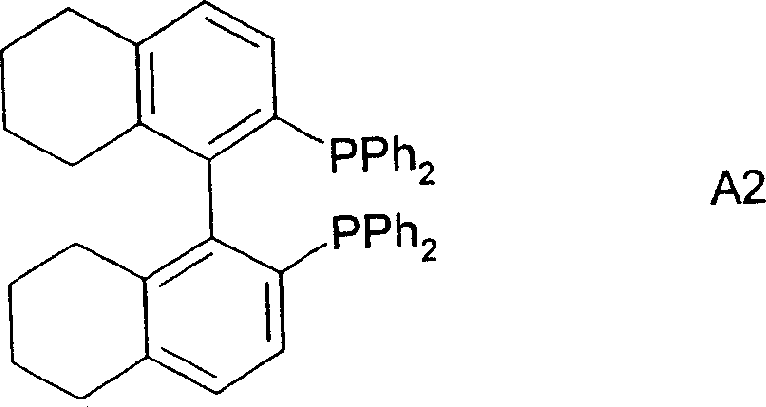 Particularly preferred embodiments utilize ligands such as (S)-BINAP and (S)-TolBINAP, where the bulky aryl substituents on the phosphorus atoms create a chiral environment that favors the formation of one enantiomer over the other during the hydride insertion step. The mechanism likely involves the formation of a cationic rhodium-dihydride species that coordinates with the olefinic or ketonic substrate, followed by migratory insertion and reductive elimination to release the chiral alcohol. The choice of counter-ions, such as triflate or tetrafluoroborate, further modulates the electrophilicity of the metal center, optimizing the reaction rate and selectivity.
Particularly preferred embodiments utilize ligands such as (S)-BINAP and (S)-TolBINAP, where the bulky aryl substituents on the phosphorus atoms create a chiral environment that favors the formation of one enantiomer over the other during the hydride insertion step. The mechanism likely involves the formation of a cationic rhodium-dihydride species that coordinates with the olefinic or ketonic substrate, followed by migratory insertion and reductive elimination to release the chiral alcohol. The choice of counter-ions, such as triflate or tetrafluoroborate, further modulates the electrophilicity of the metal center, optimizing the reaction rate and selectivity.
From an impurity control perspective, the mechanism is finely tuned to prevent the dehydration of the beta-amino alcohol product, a common side reaction that leads to enamines or imines. The patent data indicates that by carefully selecting the solvent system—preferably a mixture of hydrocarbons like toluene and alcohols like methanol—and maintaining specific hydrogen pressures between 55 and 200 bar, the equilibrium is driven strongly towards the saturated alcohol. The use of acid addition salts of the starting aminoketones is also a strategic mechanistic choice; protonating the amine nitrogen reduces its coordinating ability to the metal center, preventing catalyst poisoning while still allowing the ketone moiety to undergo reduction. This delicate balance ensures that the catalyst remains active throughout the reaction cycle, delivering high turnover numbers and consistent quality suitable for high-purity OLED material or pharmaceutical applications.
How to Synthesize (S)-3-Methylamino-1-(2-thienyl)-1-propanol Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of operations that can be readily adapted for industrial reactors. The process begins with the in situ or ex situ formation of the active catalyst species by mixing a rhodium precursor, such as [Rh(COD)Cl]2, with the chiral ligand in an inert solvent under nitrogen or argon protection. This catalyst solution is then transferred to a pressure vessel containing the substrate, typically a 3-monoalkylamino-1-aryl-1-propanone, dissolved in a methanol-toluene mixture. The detailed standardized synthesis steps see the guide below for specific molar ratios and safety protocols required to handle high-pressure hydrogenation safely and effectively.
- Preparation of the chiral catalyst solution by mixing a rhodium precursor like [Rh(COD)Cl]2 with a chiral diphosphine ligand such as (S)-TolBINAP in an inert solvent like toluene.
- Loading the aminoketone substrate into a pressure reactor with a solvent mixture of methanol and toluene, followed by the addition of the prepared catalyst solution under inert atmosphere.
- Conducting the hydrogenation reaction at elevated pressures (e.g., 55-120 bar) and moderate temperatures (50°C), followed by workup involving pH adjustment and crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented hydrogenation technology translates into tangible strategic benefits that extend beyond simple chemistry. By shifting from a resolution-based workflow to a direct asymmetric synthesis, manufacturers can significantly reduce the volume of raw materials required to produce a fixed amount of active pharmaceutical ingredient (API). This efficiency gain directly impacts the bottom line by lowering the cost per kilogram of the final intermediate, making the supply chain more resilient against fluctuations in raw material pricing. Furthermore, the elimination of stoichiometric chiral reagents and toxic demethylation agents simplifies the waste management profile, reducing the environmental burden and associated disposal costs which are increasingly scrutinized in global manufacturing hubs.
- Cost Reduction in Manufacturing: The most profound economic advantage stems from the doubling of theoretical yield compared to classical resolution methods. Since the process converts the ketone substrate directly into the desired enantiomer with high selectivity, there is no inherent 50% loss of material, which effectively halves the raw material cost contribution for that specific step. Additionally, the catalytic nature of the chirality source means that expensive chiral ligands are used in minute quantities relative to the substrate, rather than in stoichiometric amounts, leading to substantial cost savings in reagent procurement. The avoidance of complex downstream demethylation steps further reduces utility consumption and labor hours, streamlining the overall production budget.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically the mono-alkylated aminoketones, are readily accessible and can be synthesized via well-established commodity chemical routes. This ensures a stable and continuous supply of feedstock, mitigating the risk of production stoppages due to specialized reagent shortages. Moreover, the robustness of the rhodium catalyst system under the described conditions (temperatures of 20-70°C and pressures up to 200 bar) allows for flexible scheduling and reliable batch completion times. This predictability is crucial for maintaining just-in-time inventory levels and meeting the stringent delivery deadlines demanded by multinational pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram scale in the laboratory to multi-hundred gram scales in autoclaves, with parameters that translate well to tonnage production. The use of common solvents like methanol and toluene, which are easily recovered and recycled, aligns with green chemistry principles and facilitates compliance with increasingly strict environmental regulations. By minimizing the generation of hazardous byproducts like methyl chloride and reducing the overall solvent intensity through higher concentration reactions, the technology supports sustainable manufacturing practices that are essential for long-term operational licenses and corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enantioselective hydrogenation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is critical for R&D teams planning process validation and for procurement teams assessing vendor capabilities.
Q: What are the primary advantages of this hydrogenation method over traditional resolution?
A: Unlike traditional resolution which limits theoretical yield to 50%, this enantioselective hydrogenation allows for near-quantitative conversion of the starting ketone directly into the desired chiral alcohol, effectively doubling the potential output from the same amount of raw material.
Q: Does this process require toxic demethylation steps?
A: No, a significant advantage of this patented route is that it utilizes mono-alkylated aminoketones directly, thereby eliminating the need for subsequent demethylation steps that often generate hazardous byproducts like methyl chloride.
Q: What level of enantiomeric excess (ee) can be achieved?
A: The process is capable of achieving high enantiomeric purity, with experimental data showing initial ee values around 92-94%, which can be further upgraded to >99% ee through a simple crystallization step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino Alcohols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving CNS medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are equipped with state-of-the-art high-pressure hydrogenation facilities and rigorous QC labs capable of verifying stringent purity specifications, including enantiomeric excess values exceeding 99% ee as required by modern pharmacopeial standards. Our commitment to quality assurance guarantees that every batch of amino alcohol intermediate meets the exacting demands of global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
