Advanced Iridium-Catalyzed Selective Synthesis of Amino Alcohols for Pharmaceutical Manufacturing
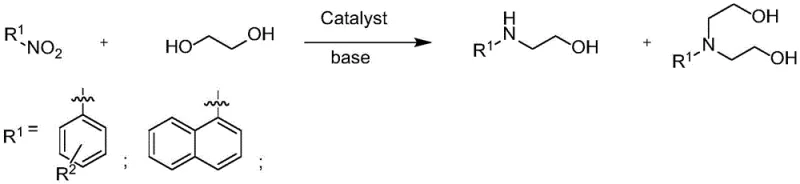
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more atom-economical synthetic routes. Patent CN114591185B, published in early 2023, introduces a groundbreaking homogeneous catalysis technology that addresses critical bottlenecks in the production of amino alcohols, which are vital scaffolds for drugs like rivaroxaban and various chiral auxiliaries. This innovation leverages a sophisticated iridium-complex catalytic system to facilitate the selective N-alkylation of nitroarenes using ethylene glycol. By integrating the reduction of the nitro group and the subsequent alkylation into a single pot, this methodology eliminates multiple processing steps and hazardous waste streams. For R&D directors and process chemists, this represents a paradigm shift from stoichiometric reductions to catalytic hydrogen borrowing, offering a pathway to high-purity intermediates with superior environmental profiles. The ability to tune the reaction towards either mono- or di-substituted products simply by adjusting base parameters adds a layer of operational flexibility that is rarely seen in traditional amine synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino alcohols has relied on methodologies that are increasingly untenable in modern regulatory and economic environments. The classical reduction of amino acids, for instance, necessitates the use of potent and dangerous reducing agents such as lithium aluminum hydride or sodium borohydride, which generate substantial quantities of inorganic waste and pose significant safety risks during handling and disposal. Alternatively, the ring-opening of epoxides with anilines, while effective, requires the use of ethylene oxide derivatives, which are classified as toxic and carcinogenic, thereby imposing strict containment costs and worker safety protocols. Furthermore, coupling halides with ethanolamines produces stoichiometric amounts of halogenated salt byproducts, leading to poor atom economy and complicating downstream purification processes. These legacy methods not only inflate the cost of goods sold due to waste treatment and raw material inefficiency but also create supply chain vulnerabilities associated with the procurement of hazardous regulated chemicals.
The Novel Approach
In stark contrast, the technology disclosed in CN114591185B utilizes a "hydrogen borrowing" or "borrowing hydrogen" strategy that fundamentally redesigns the synthetic logic. By employing nitroarenes as the starting material and ethylene glycol as both the solvent and the alkylating agent, the process achieves a remarkable convergence of step economy and safety. The reaction proceeds through a cascade where the alcohol is transiently dehydrogenated to an aldehyde, which then condenses with the in-situ reduced amine to form an imine, followed by final reduction to the amine product. Crucially, water is the only byproduct generated throughout this entire sequence, aligning perfectly with the principles of green chemistry. This approach bypasses the need for isolated amine intermediates and external hydrogen gas, simplifying the reactor setup and reducing the overall carbon footprint of the manufacturing process.
Mechanistic Insights into Iridium-Catalyzed Hydrogen Borrowing
The core of this technological breakthrough lies in the design of the iridium catalysts, specifically the Cp*-based complexes designated as Cat.A and Cat.B in the patent documentation. These complexes feature robust ligand architectures, such as N-(4-hydroxyphenyl)picolinamide or 6,6'-dihydroxy-2,2'-bipyridine, which stabilize the metal center against decomposition under the thermal stress of the reaction conditions (60-120°C). The catalytic cycle initiates with the dehydrogenation of ethylene glycol to glycolaldehyde, facilitated by the iridium center, generating a metal-hydride species. This hydride is subsequently utilized to reduce the nitroarene substrate to the corresponding aniline. The aniline then undergoes condensation with the glycolaldehyde to form an imine intermediate, which is finally reduced by the remaining metal-hydride species to yield the target amino alcohol. This intricate dance of hydrogen transfer ensures that no external reducing agent is required, and the catalyst turnover number is maximized through efficient hydride recycling.
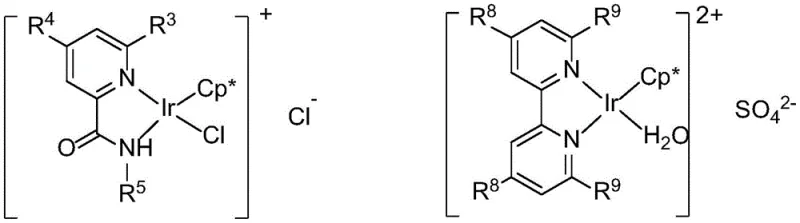
Beyond the fundamental mechanism, the patent highlights a sophisticated level of selectivity control that is critical for pharmaceutical applications where impurity profiles must be tightly managed. The system allows for the preferential formation of either N-hydroxyethyl arylamines (mono-alkylated) or N,N-dihydroxyethyl arylamines (di-alkylated) by modulating the basicity and stoichiometry of the reaction medium. For example, the use of cesium carbonate tends to favor the mono-substituted product with yields reaching up to 87%, likely due to steric or electronic modulation of the catalyst's active site. Conversely, optimizing the loading of potassium hydroxide shifts the equilibrium towards the di-substituted product. This tunability implies that a single catalytic platform can service multiple product lines, reducing the need for distinct process development campaigns for structurally similar intermediates. The substrate scope is equally impressive, accommodating various functional groups including halogens, ethers, and esters on the aromatic ring, as demonstrated by the successful conversion of substrates like 1-nitronaphthalene and 4-nitrotoluene.
How to Synthesize N-Hydroxyethyl Arylamines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and selectivity. The process begins with the careful preparation of the iridium catalyst precursor, followed by the assembly of the reaction mixture under an inert atmosphere to prevent catalyst oxidation. The choice of base is not merely a pH adjuster but a critical selectivity switch; therefore, precise weighing and addition of bases like cesium carbonate or potassium hydroxide are essential. The reaction temperature and time must be optimized based on the specific nitroarene substrate, with electron-deficient substrates potentially requiring different conditions than electron-rich ones. Following the reaction, standard workup procedures involving dichloromethane extraction and chromatographic purification are employed to isolate the product. For a detailed, step-by-step protocol including exact molar ratios and purification techniques, please refer to the standardized guide below.
- Prepare the reaction mixture by combining nitroarenes, a specific base (such as cesium carbonate or potassium hydroxide), and the iridium catalyst in ethylene glycol under nitrogen protection.
- Heat the sealed reaction vessel in an oil bath at temperatures ranging from 60°C to 120°C and stir continuously for a duration of 6 to 30 hours depending on the desired substitution level.
- Upon completion, cool the mixture to room temperature, extract the product using dichloromethane, and purify the crude residue via column chromatography to isolate the high-purity amino alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers tangible strategic benefits that extend beyond simple yield improvements. The shift away from hazardous reagents like lithium aluminum hydride and ethylene oxide significantly reduces the regulatory burden and insurance costs associated with chemical storage and transport. By utilizing ethylene glycol, a commodity chemical with a stable and abundant global supply, the process mitigates the risk of raw material shortages that often plague specialty reagent markets. Furthermore, the generation of water as the sole byproduct drastically simplifies waste management protocols, eliminating the need for expensive treatment of halogenated or heavy metal-contaminated waste streams. This streamlined waste profile translates directly into lower operational expenditures and a reduced environmental footprint, enhancing the company's sustainability metrics which are increasingly important for corporate social responsibility reporting.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzed through the lens of total cost of ownership. By replacing stoichiometric reducing agents with a catalytic system, the consumption of expensive reagents is minimized, and the atom economy is significantly improved. The dual role of ethylene glycol as both solvent and reactant eliminates the need for purchasing and recovering separate solvent volumes, thereby reducing utility costs associated with distillation and solvent recovery. Additionally, the mild reaction conditions (60-120°C) require less energy input compared to high-pressure hydrogenation or cryogenic reduction processes, leading to substantial savings in energy consumption over the lifecycle of the production campaign.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available feedstocks. Nitroarenes and ethylene glycol are produced at massive scales globally, ensuring a consistent supply even during market fluctuations. The stability of the iridium catalysts described in the patent means they can be stored for extended periods without significant degradation, allowing for better inventory management and reducing the risk of production delays caused by catalyst spoilage. This reliability ensures that production schedules can be maintained with greater certainty, supporting just-in-time manufacturing models and reducing the need for excessive safety stock of finished intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the catalysis and the absence of exothermic hazards associated with strong reducing agents. The liquid-phase reaction is easily adaptable to standard stainless steel reactors without the need for specialized high-pressure equipment. From an environmental compliance perspective, the elimination of toxic byproducts ensures that the process meets stringent international environmental regulations, such as REACH in Europe or TSCA in the US. This compliance facilitates smoother regulatory filings for new drug applications and reduces the risk of shutdowns due to environmental non-compliance, securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iridium-catalyzed synthesis method. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of catalyst selection and base optimization is key to successfully transferring this technology from the lab to commercial production. We encourage technical teams to review these points carefully to assess the fit for their specific manufacturing requirements.
Q: What distinguishes this iridium-catalyzed method from traditional amino alcohol synthesis?
A: Unlike traditional methods that rely on hazardous reducing agents like lithium aluminum hydride or toxic epoxides, this patented process utilizes a hydrogen-borrowing strategy with ethylene glycol. This results in water as the sole byproduct, significantly improving atom economy and environmental safety while eliminating the need for dangerous reagents.
Q: How is selectivity between mono- and di-substituted products controlled?
A: The patent demonstrates that selectivity is precisely tunable by adjusting the type and stoichiometric amount of the base used. For instance, utilizing cesium carbonate favors the formation of N-hydroxyethyl arylamines (mono-substituted), whereas optimizing potassium hydroxide loading facilitates the synthesis of N,N-dihydroxyethyl arylamines (di-substituted).
Q: Is this catalytic system suitable for large-scale industrial production?
A: Yes, the system is designed for scalability. The iridium complexes employed are stable and simple to prepare, and the use of ethylene glycol serves a dual purpose as both a low-cost solvent and a reactant. The mild reaction conditions (60-120°C) and the absence of corrosive byproducts make it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from process development to full-scale supply. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of amino alcohol intermediate meets the exacting standards required for pharmaceutical applications. We understand the critical importance of consistency and quality in the supply of key intermediates, and our team is dedicated to maintaining the highest levels of operational excellence.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us to request specific COA data for our existing amino alcohol portfolio or to discuss route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain and reduce costs through innovative chemistry.
