Advanced Biocatalytic Resolution for High-Purity Indoxacarb Intermediates and Commercial Scalability
Introduction to Patent CN110527646B and Technological Breakthroughs
The global demand for high-efficiency, low-toxicity agrochemicals has driven intense research into the synthesis of key chiral intermediates, particularly for next-generation insecticides like Indoxacarb. Patent CN110527646B, titled "Bacillus tropicalis WZZ018 and its application," represents a significant paradigm shift in the manufacturing of S-(+)-5-chloro-2,3-dihydro-2-hydroxy-1-oxo-1H-indene-2-carboxylic acid methyl ester. This specific chiral building block is critical for the production of Indoxacarb, a broad-spectrum oxadiazine insecticide known for its unique mode of action blocking sodium ion channels in insect nerve cells. The patent discloses a novel microbial strain, Bacillus tropicalis WZZ018 (preserved under accession number CCTCC NO: M2019241), which serves as a highly efficient biocatalyst for the stereoselective resolution of the racemic substrate. Unlike traditional chemical synthesis routes that struggle with complex racemization competition factors and harsh reaction environments, this biological approach leverages the intrinsic regioselectivity of the microorganism to achieve an enantiomeric excess (ee) value of up to 93.72%. This technological advancement addresses long-standing challenges in the agrochemical industry regarding optical purity, environmental sustainability, and process safety, offering a robust foundation for the industrial-scale production of high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
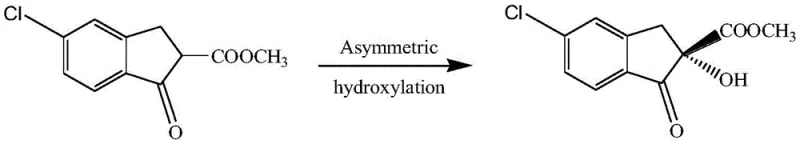
Prior to the development of this biocatalytic method, the synthesis of the target chiral intermediate relied heavily on complex chemical pathways that were fraught with inefficiencies and safety hazards. One prominent conventional method, described by Guo Li Xiang, utilized acrylic acid as a starting material, undergoing a series of rigorous transformations including hydrogenation, acyl chlorination, and alkylation to form 5-chloro-1-oxo-2,3-indan-2-carboxylic acid methyl ester. This was followed by an asymmetric hydroxylation step employing cinchonine as a chiral catalyst. As illustrated in the reaction scheme below, this chemical route is not only multi-step and labor-intensive but also suffers from suboptimal stereocontrol, yielding an ee value of only 89.4%. Furthermore, another common chemical approach involves the direct asymmetric hydroxylation of 5-chloro-2-methoxycarbonyl-1-indanone using cumene hydroperoxide and cinchonine in dichloromethane solvent. While this method can achieve slightly higher optical purity (around 92%), it necessitates the use of hazardous organic solvents and unstable peroxide oxidants. The reliance on expensive chiral ligands and the generation of toxic waste streams make these conventional chemical processes economically burdensome and environmentally unsustainable for large-scale manufacturing.
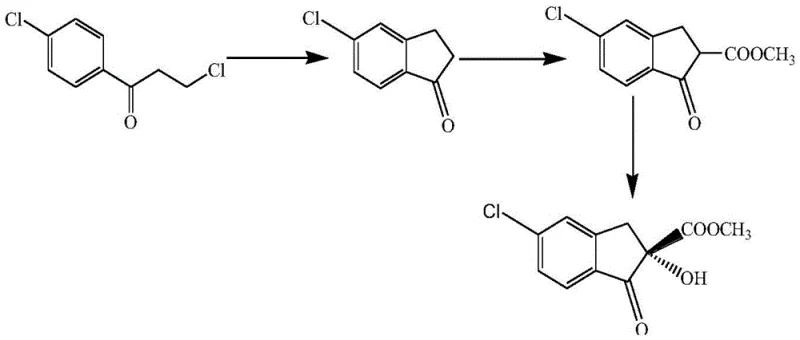
The Novel Approach
In stark contrast to the hazardous and complex chemical methodologies, the novel approach disclosed in Patent CN110527646B utilizes a whole-cell biocatalytic system that dramatically simplifies the production workflow. By employing wet cells or freeze-dried powder of Bacillus tropicalis WZZ018, the process achieves stereoselective hydrolysis of the racemic ester directly in an aqueous buffer system. This biological resolution effectively discriminates between the R and S enantiomers, hydrolyzing the unwanted R-configuration while leaving the desired S-(+)-isomer intact with high fidelity. The reaction proceeds under exceptionally mild conditions—typically at temperatures between 20°C and 45°C and a neutral pH range of 5.0 to 10.0—eliminating the need for cryogenic temperatures or aggressive reagents. This shift from organic synthesis to biocatalysis not only enhances the optical purity of the final product to over 93% ee but also fundamentally alters the safety profile of the manufacturing process, removing the risks associated with peroxide handling and volatile organic compound (VOC) emissions.
Mechanistic Insights into Enzymatic Stereoselective Hydrolysis
The core mechanism driving this technological breakthrough lies in the specific enzymatic activity inherent to the Bacillus tropicalis WZZ018 strain, which possesses a highly specialized esterase or lipase capable of distinguishing subtle stereochemical differences in the substrate molecule. When the racemic 5-chloro-2,3-dihydro-2-hydroxy-1-oxo-1H-indene-2-carboxylic acid methyl ester is introduced to the fermentation broth or suspended cell solution, the microbial enzymes selectively bind to the R-enantiomer due to the precise spatial arrangement of their active sites. This binding facilitates the hydrolysis of the ester bond in the R-isomer, converting it into a more polar carboxylic acid derivative that remains in the aqueous phase or is easily separated, while the S-enantiomer remains unhydrolyzed and retains its ester functionality. This kinetic resolution is governed by the enzyme's strict regioselectivity, which prevents the non-specific degradation of the desired product. The patent data indicates that the conversion rate can reach approximately 52.84%, which is theoretically consistent with a perfect 50:50 resolution of a racemate, confirming that the enzyme acts with high specificity on only one enantiomer without significant background hydrolysis of the target S-isomer.
Furthermore, the stability and robustness of the biocatalyst play a crucial role in maintaining high product quality throughout the reaction lifecycle. The strain WZZ018 demonstrates remarkable tolerance to substrate concentrations ranging from 4.2 mM to 50 mM, although higher concentrations (e.g., 150 mM) can lead to substrate inhibition and reduced conversion efficiency. The enzymatic activity is optimized in a phosphate buffer system at pH 7.0, which mimics the physiological conditions of the bacteria, ensuring maximal enzyme turnover and longevity. From an impurity control perspective, the biological nature of the reaction minimizes the formation of side products commonly associated with chemical oxidation or reduction steps, such as over-oxidized byproducts or racemized impurities. The downstream processing involves a straightforward acidification and extraction protocol, followed by silica gel column chromatography, which effectively isolates the high-purity S-(+)-isomer. This clean reaction profile significantly reduces the burden on purification units, allowing for a more streamlined isolation process that preserves the integrity of the chiral center.
How to Synthesize S-(+)-5-chloro-2,3-dihydro-2-hydroxy-1-oxo-1H-indene-2-carboxylic acid methyl ester Efficiently
To implement this advanced biocatalytic route in a laboratory or pilot setting, operators must adhere to a precise fermentation and reaction protocol that maximizes the expression of the stereoselective enzymes. The process begins with the activation of the Bacillus tropicalis WZZ018 strain on a slant culture medium, followed by seed expansion in a nutrient-rich broth containing beef extract and peptone. Once sufficient biomass is generated, the cells are harvested via centrifugation and either used immediately as wet thalli or lyophilized for storage and transport flexibility. The actual resolution step requires suspending the biocatalyst in a buffered aqueous solution containing the racemic substrate, maintaining strict control over temperature and agitation to ensure adequate mass transfer without denaturing the enzymes. For a comprehensive understanding of the operational parameters and specific stoichiometric ratios required for optimal yield, please refer to the detailed standardized synthesis steps outlined in the guide below.
- Cultivate Bacillus tropicalis WZZ018 in a specific fermentation medium containing glucose and beef extract to generate wet cells or freeze-dried powder.
- Prepare a reaction system using a phosphate buffer (pH 7.0) and suspend the biocatalyst along with the racemic substrate at a concentration of 20mM.
- Maintain the reaction at 30°C with shaking at 180r/min for 24 hours, followed by acidification, extraction, and silica gel column chromatography for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chemical synthesis to the biocatalytic method described in Patent CN110527646B offers profound strategic advantages that extend beyond mere technical performance. The elimination of expensive chiral catalysts like cinchonine and hazardous oxidants like cumene hydroperoxide directly translates to a significant reduction in raw material costs. Moreover, the shift to an aqueous-based system removes the necessity for large volumes of organic solvents such as dichloromethane, thereby drastically cutting down on solvent procurement expenses and the associated costs of solvent recovery and waste disposal. This simplification of the bill of materials (BOM) creates a more resilient supply chain that is less vulnerable to price volatility in the petrochemical sector, ensuring more stable pricing for the final agrochemical intermediate.
- Cost Reduction in Manufacturing: The biocatalytic process inherently lowers manufacturing overheads by operating under ambient pressure and moderate temperatures, which reduces energy consumption compared to the high-energy demands of cryogenic chemical reactions. By avoiding the use of precious metal catalysts or complex chiral ligands that often require specialized recycling protocols, the overall cost of goods sold (COGS) is substantially decreased. Additionally, the high selectivity of the enzyme minimizes the formation of difficult-to-remove impurities, reducing the number of purification cycles needed and increasing the overall throughput of the production facility. These cumulative efficiencies result in a leaner, more cost-effective manufacturing operation that enhances profit margins for downstream agrochemical formulators.
- Enhanced Supply Chain Reliability: Relying on a fermentable microbial strain like Bacillus tropicalis WZZ018 provides a sustainable and renewable source of catalytic activity, unlike chemical catalysts which may face supply constraints or geopolitical sourcing issues. The ability to produce the biocatalyst in-house through standard fermentation techniques grants manufacturers greater autonomy and control over their production schedules. Furthermore, the mild reaction conditions reduce the risk of unplanned shutdowns due to safety incidents or equipment failures associated with handling hazardous peroxides, thereby ensuring a more consistent and reliable delivery of high-purity intermediates to customers. This reliability is critical for maintaining continuous production lines in the fast-paced agrochemical market.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental protection intensify globally, the green chemistry credentials of this biocatalytic method offer a distinct competitive advantage. The process generates significantly less hazardous waste and avoids the emission of volatile organic compounds (VOCs), making it easier to comply with stringent environmental regulations such as REACH or local EPA standards. The scalability of fermentation processes is well-established in the fine chemical industry, allowing for seamless expansion from pilot scale to multi-ton commercial production without the need for exotic reactor configurations. This ease of scale-up, combined with a reduced environmental footprint, positions manufacturers as responsible partners capable of meeting the sustainability goals of major multinational agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These answers are derived directly from the experimental data and specifications provided in Patent CN110527646B, ensuring accuracy and relevance for technical decision-makers evaluating this process for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this biological resolution step into existing manufacturing workflows.
Q: What is the enantiomeric excess (ee) achieved by the Bacillus tropicalis WZZ018 method?
A: The patented biocatalytic process achieves an enantiomeric excess (ee) value of up to 93.72% for the S-(+) isomer, significantly surpassing traditional chemical methods which typically yield around 89-92%.
Q: Why is the biological enzyme method preferred over chemical asymmetric hydroxylation for this intermediate?
A: The biological method eliminates the need for hazardous organic solvents like dichloromethane and expensive chiral catalysts such as cinchonine. It operates under mild aqueous conditions, reducing environmental pollution and safety risks associated with peroxides.
Q: What are the optimal reaction conditions for the enzymatic resolution?
A: The optimal conditions involve using a phosphate buffer at pH 7.0, a temperature of 30°C, and a shaking speed of 180r/min for a duration of 24 hours, ensuring maximum conversion and stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoxacarb Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Bacillus tropicalis WZZ018 mediated resolution process for the production of high-purity Indoxacarb intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in an industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities designed to meet stringent purity specifications, guaranteeing that every batch of chiral intermediate delivered meets the exacting standards required for modern agrochemical formulations. We are committed to bridging the gap between innovative academic research and practical commercial application.
We invite procurement leaders and R&D directors to collaborate with us to explore how this biocatalytic technology can optimize your supply chain and reduce overall manufacturing costs. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate the superior optical purity and consistency of our biocatalytically produced intermediates.
