Revolutionizing Alkyl Boronate Production: A Metal-Free Strategy for Commercial Scale-Up
The landscape of organic synthesis is undergoing a significant transformation driven by the urgent need for greener, more efficient, and cost-effective methodologies, particularly in the production of high-value pharmaceutical intermediates. Patent CN109575063B introduces a groundbreaking approach to the boron esterification of alkyl halides that completely bypasses the requirement for transition metal catalysis. This innovation addresses critical pain points in modern medicinal chemistry, where the removal of trace metal contaminants from Active Pharmaceutical Ingredients (APIs) remains a costly and technically challenging bottleneck. By utilizing a simple system comprising alkyl halides, diboronates, and alkoxide bases in a mixed solvent system with trace water, this technology enables the direct conversion of carbon-halogen bonds to carbon-boron bonds under remarkably mild conditions. The implications for the global supply chain of fine chemicals are profound, offering a robust alternative to traditional methods that often rely on scarce and expensive noble metals.
This novel methodology represents a paradigm shift in how we approach the synthesis of organoboron compounds, which are indispensable building blocks for Suzuki-Miyaura cross-coupling reactions. The ability to generate these key intermediates without the baggage of transition metal complexes not only streamlines the synthetic route but also aligns perfectly with the principles of green chemistry. For R&D directors and process chemists, this means access to a wider chemical space with fewer restrictions on functional group tolerance. For procurement and supply chain leaders, it signals a future where the volatility of precious metal markets has less impact on the cost of goods sold. As we delve deeper into the technical specifics of this patent, it becomes clear that this is not merely an academic curiosity but a commercially viable process ready for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkyl boronic esters has been dominated by two main strategies, both of which carry significant drawbacks for large-scale manufacturing. The first involves the use of highly reactive organometallic reagents such as Grignard reagents or organolithium compounds. While effective for simple substrates, these reagents are notoriously intolerant of functional groups, requiring extensive protection and deprotection steps that drastically increase the step count and reduce overall yield. Furthermore, the handling of these pyrophoric materials demands specialized equipment and rigorous safety protocols, inflating capital expenditure and operational risks. The second conventional approach relies on transition metal catalysis, typically employing palladium, nickel, copper, or iron complexes. Although these methods offer better functional group compatibility, they introduce a new set of challenges, including the necessity for complex ligand optimization, sensitivity to air and moisture, and the critical issue of residual metal contamination. Removing trace amounts of toxic heavy metals from the final product to meet stringent regulatory standards often requires additional purification steps, such as scavenging or recrystallization, which erode profit margins and extend lead times.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN109575063B offers a streamlined, transition metal-free pathway that leverages the intrinsic reactivity of diboronates activated by alkoxide bases. This approach eliminates the need for expensive catalysts and ligands entirely, replacing them with inexpensive and readily available alkali metal alkoxides like lithium tert-butoxide. The reaction proceeds efficiently in common organic solvents such as methanol, with the surprising addition of a small amount of water actually facilitating the process rather than inhibiting it. This tolerance to moisture is a game-changer for industrial operations, as it relaxes the stringent requirement for anhydrous conditions and inert atmospheres, thereby reducing energy consumption associated with solvent drying and nitrogen purging. The result is a robust protocol that converts primary and secondary alkyl iodides and bromides into their corresponding boronic esters with considerable yields, all while maintaining excellent functional group compatibility.
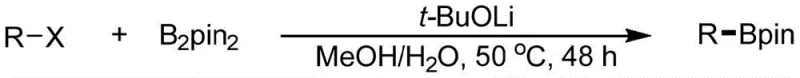
The general reaction scheme illustrates the simplicity and elegance of this transformation. An alkyl halide (R-X), where X is typically iodine or bromine, reacts with a diboron source such as bis(pinacolato)diboron (B2pin2) in the presence of an alkoxide base. The conditions are remarkably mild, typically requiring temperatures between 25°C and 65°C, with 50°C being the preferred operating point for optimal kinetics. This low thermal demand not only saves energy but also minimizes the formation of thermal degradation byproducts, ensuring a cleaner crude reaction profile. The versatility of this method is further evidenced by its applicability to a wide range of substrates, from simple linear alkanes to complex molecules bearing esters, ethers, and even other halogen atoms, making it an ideal candidate for the late-stage functionalization of complex drug candidates.
Mechanistic Insights into Alkoxide-Mediated Radical Borylation
Understanding the mechanistic underpinnings of this reaction is crucial for R&D teams looking to implement this technology effectively. Unlike transition metal-catalyzed cycles that involve oxidative addition and reductive elimination steps, this metal-free process is believed to proceed via a radical mechanism initiated by single electron transfer (SET). Under basic conditions, the alkoxide anion attacks the diboronate species, generating a nucleophilic boron species. This activated complex then engages with the alkyl halide through a single electron transfer process, resulting in the formation of an alkyl radical and a boron-centered radical intermediate. The subsequent coupling of these radical species leads to the formation of the desired carbon-boron bond. This radical pathway explains the method's unique selectivity and its ability to tolerate functional groups that might otherwise interfere with polar organometallic mechanisms. The absence of a metal center also means there are no coordination spheres to manage, simplifying the kinetic profile and reducing the likelihood of catalyst deactivation.
From an impurity control perspective, this mechanism offers distinct advantages. Traditional metal-catalyzed reactions often suffer from homocoupling side reactions or beta-hydride elimination, especially with secondary alkyl substrates. The radical nature of this new method, combined with the specific reaction conditions involving trace water, appears to suppress these competing pathways effectively. The water plays a subtle but vital role, potentially stabilizing intermediate species or facilitating the heterogeneity of the reaction mixture, as indicated by the formation of white precipitates (likely LiX adducts) during the process. This controlled environment ensures that the final product profile is clean, with minimal byproduct formation, which translates directly to easier downstream processing. For quality control laboratories, this means simpler analytical methods and faster release times for batches, further enhancing the overall efficiency of the manufacturing workflow.
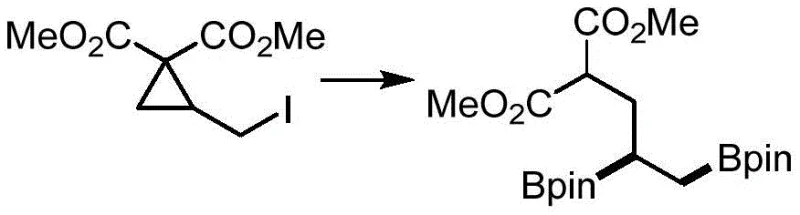
The scope of this reaction extends beyond simple mono-borylation, as demonstrated by specific embodiments in the patent where di-borylation products are achieved with high efficiency. For instance, substrates containing multiple reactive sites can be selectively functionalized, opening up new avenues for the synthesis of complex building blocks used in material science and agrochemicals. The ability to install boron motifs at specific positions on a carbon chain without affecting other sensitive moieties is a powerful tool in the synthetic chemist's arsenal. This level of precision, achieved without the crutch of directing groups or specialized ligands, underscores the fundamental robustness of the alkoxide-mediated activation strategy. It suggests that this chemistry is not just a niche alternative but a foundational method that could redefine how alkyl boronates are produced on a global scale.
How to Synthesize Alkyl Boronates Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific parameters to ensure reproducibility and high yield. The process is designed to be operationally simple, avoiding the complexities associated with handling air-sensitive catalysts. The key lies in the precise stoichiometry of the reagents and the maintenance of the recommended temperature profile. By following the standardized protocol derived from the patent data, manufacturers can reliably produce high-purity alkyl boronates suitable for downstream coupling reactions. The detailed标准化 synthesis steps are provided below to guide your technical team in adopting this innovative methodology.
- Prepare the reaction mixture by dissolving 1 equivalent of alkyl halide, 3 to 4 equivalents of diboronate (e.g., B2pin2), and 1.5 to 2 equivalents of alkoxide base in an organic solvent like methanol.
- Add water equivalent to 0.5% to 2% of the organic solvent volume to the mixture to facilitate the heterogeneous reaction environment.
- Seal the reaction system and stir at a temperature between 25°C and 65°C (preferably 50°C) for more than 12 hours to obtain the alkyl boronate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transition metal-free borylation technology presents a compelling value proposition centered on cost stability and operational resilience. The traditional reliance on palladium and nickel catalysts exposes manufacturers to significant price volatility driven by geopolitical factors and mining constraints. By eliminating these precious metals from the process entirely, companies can decouple their production costs from the fluctuating commodities market. Furthermore, the removal of the catalyst removal step—often a resource-intensive process involving silica treatment or specialized scavengers—drastically reduces the consumption of auxiliary materials and waste generation. This simplification of the workflow leads to substantial cost savings in both raw materials and waste disposal, contributing directly to a healthier bottom line.
- Cost Reduction in Manufacturing: The economic benefits of this method are multifaceted, starting with the direct elimination of expensive transition metal catalysts and ligands. In traditional processes, the cost of the catalyst can account for a significant portion of the raw material expense, especially when high loadings are required for difficult substrates. By switching to inexpensive alkoxides and diboronates, the direct material cost is significantly lowered. Additionally, the simplified workup procedure reduces the labor and time required for purification. There is no need for extensive metal scavenging or repeated recrystallizations to meet strict residual metal specifications. This streamlined process flow enhances throughput and reduces the overall cost of goods sold, making the final pharmaceutical intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, where disruptions can halt production lines and delay drug launches. This new method relies on commodity chemicals like methanol, alkyl halides, and alkoxides, which are produced in vast quantities globally and have stable supply chains. Unlike specialized catalysts that may have long lead times or single-source suppliers, these reagents are readily available from multiple vendors. This diversification of supply sources mitigates the risk of shortages and ensures consistent production schedules. Moreover, the robustness of the reaction to moisture and air reduces the dependency on high-purity dry solvents and inert gas supplies, further insulating the manufacturing process from logistical bottlenecks.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden challenges, but this metal-free borylation is inherently scalable due to its mild operating conditions. The reaction runs at moderate temperatures (around 50°C), which reduces the energy load on heating and cooling systems compared to cryogenic or high-temperature alternatives. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and solid waste disposal, helping facilities meet increasingly stringent environmental regulations. The reduced ecological footprint not only lowers compliance costs but also enhances the company's sustainability profile, a factor that is becoming increasingly important for corporate social responsibility reporting and customer preference.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free borylation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. They are intended to provide clarity for decision-makers evaluating the feasibility of integrating this process into their existing manufacturing portfolios. Understanding these nuances is essential for maximizing the benefits of this innovative synthetic route.
Q: What are the primary advantages of this metal-free borylation method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of transition metal catalysts such as palladium, nickel, or copper. This removes the need for expensive ligand screening, eliminates the risk of heavy metal contamination in the final API, and significantly simplifies the downstream purification process, leading to substantial cost savings and environmental benefits.
Q: Does this method tolerate sensitive functional groups commonly found in drug intermediates?
A: Yes, the method exhibits broad functional group compatibility. It successfully converts primary and secondary alkyl iodides and bromides containing esters, ethers, hydroxyl groups, and even other halides into their corresponding boronic esters under mild conditions, preserving the integrity of complex molecular architectures.
Q: Is the reaction scalable for industrial production of pharmaceutical intermediates?
A: Absolutely. The reaction operates under mild temperatures (25-65°C) and uses readily available reagents like alkoxides and diboronates. The absence of air-sensitive catalysts and the use of common solvents like methanol make the process highly amenable to commercial scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Boronate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener and more efficient synthetic methods is not just a trend but a necessity for the future of the chemical industry. Our team of expert process chemists has thoroughly analyzed the potential of the metal-free borylation technology described in CN109575063B and is prepared to assist clients in scaling this route from gram-scale discovery to multi-ton commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical manufacturing setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of alkyl boronate meets the highest standards required for pharmaceutical applications.
We invite you to explore how this advanced synthesis method can optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific product portfolio. By partnering with us, you gain access to not just a reliable supplier, but a strategic partner committed to innovation and quality. Please contact us today to request specific COA data and route feasibility assessments for your target compounds, and let us help you navigate the next generation of chemical synthesis.
