Advanced Organocatalytic Synthesis of (4S,5R)-Half Ester for Commercial Biotin Production
The pharmaceutical industry continuously seeks robust methodologies for synthesizing chiral building blocks, particularly for vital nutrients like Vitamin H. Patent CN101284832A introduces a groundbreaking preparation method for (4S,5R)-half ester, a critical intermediate in the total synthesis of (+)-biotin. This technology leverages a sophisticated 9-epiquinurea organocatalyst to facilitate the enantioselective ring-opening of cyclic acid anhydrides with alcohols. Unlike traditional heavy metal catalysis or stoichiometric chiral auxiliaries, this approach operates under remarkably mild conditions, typically at room temperature, achieving exceptional stereocontrol with enantiomeric excess (ee) values surpassing 98%. The structural integrity and stereochemistry of the target molecule are paramount for downstream biological activity, making this high-fidelity synthetic route a significant asset for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier for the global vitamin market.
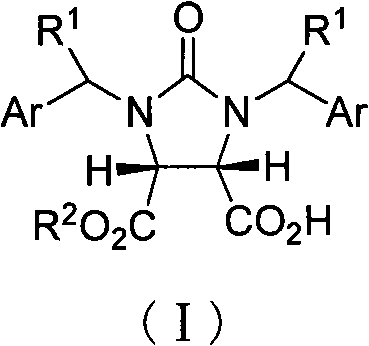
The production of high-purity biotin intermediate has historically been plagued by inefficiencies inherent in classical resolution techniques. Conventional methods, such as those reported by Gerecke et al., rely on the formation of diastereomeric salts using resolving agents like pseudoephedrine or dehydroabietamine. These processes are fundamentally flawed because they are limited by a maximum theoretical yield of 50% for the desired enantiomer, necessitating the disposal or racemization of the unwanted isomer. Furthermore, the resolving agents employed are often costly, derived from limited natural sources, and difficult to recover quantitatively, leading to substantial waste generation and inflated production costs. Alternative enzymatic approaches using pig liver esterase have also been explored but suffer from limitations in substrate scope, operational complexity, and difficulties in scaling up to industrial volumes due to enzyme stability issues and the requirement for stringent reaction controls.
In stark contrast, the novel approach detailed in the patent utilizes a chiral organocatalyst derived from 9-epiquinine to drive the asymmetric monoesterification directly. This method bypasses the yield ceiling of resolution strategies by kinetically differentiating the enantiotopic carbonyl groups of the meso-anhydride or desymmetrizing the substrate effectively. The use of 9-epiquinine thiourea derivatives allows the reaction to proceed with high turnover and selectivity without the need for cryogenic temperatures or inert atmospheres typically required for sensitive metal catalysts. By employing inexpensive and readily available alcohols such as methanol alongside green solvents like methyl tert-butyl ether (MTBE), the process significantly simplifies the operational workflow. This shift from stoichiometric chiral reagents to catalytic asymmetric synthesis represents a paradigm shift in cost reduction in vitamin manufacturing, offering a sustainable pathway that aligns with modern green chemistry principles while delivering superior product quality.
Mechanistic Insights into 9-Epiquinine Thiourea Catalyzed Ring-Opening
The efficacy of this synthesis lies in the unique dual-activation mode provided by the 9-epiquinine thiourea catalyst structure. The catalyst functions through a network of hydrogen bonding interactions that simultaneously activate the electrophilic cyclic acid anhydride and orient the nucleophilic alcohol. The thiourea moiety (where Z=S) acts as a potent hydrogen bond donor, stabilizing the developing negative charge on the anhydride oxygen during the transition state, thereby lowering the activation energy for the ring-opening event. Concurrently, the basic quinuclidine nitrogen within the catalyst scaffold may assist in deprotonating the alcohol or organizing the transition state geometry through secondary interactions. This precise spatial arrangement within the chiral pocket of the quinine backbone ensures that the nucleophilic attack occurs exclusively at one specific carbonyl carbon, dictating the formation of the (4S,5R) configuration with high fidelity. Understanding this mechanistic nuance is crucial for R&D teams optimizing reaction parameters to maintain consistent stereoselectivity across different batches.
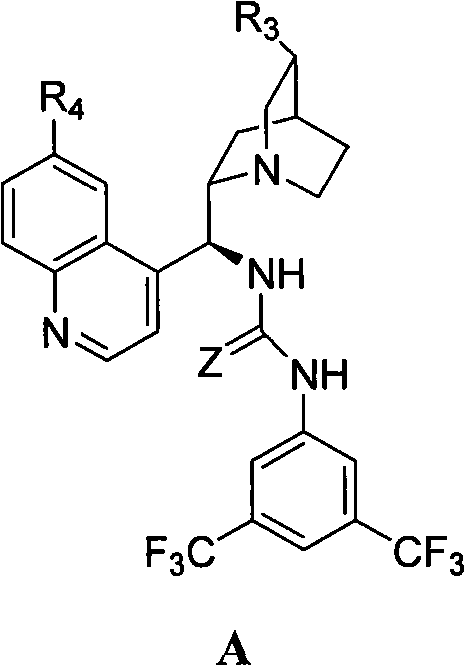
Impurity control is another critical aspect where this organocatalytic mechanism excels over traditional methods. In resolution processes, the presence of the opposite enantiomer is an intrinsic byproduct that requires rigorous and often yield-loss-inducing recrystallization steps to remove. In this catalytic system, the high enantioselectivity (ee > 98%) inherently minimizes the formation of the (4R,5S) impurity at the source. Furthermore, the mild reaction conditions (0°C to 25°C) prevent thermal degradation of the sensitive anhydride substrate or the product half-ester, which can occur under the harsher conditions sometimes needed for enzymatic or metal-catalyzed variants. The catalyst itself is designed to be chemically robust, resisting decomposition during the reaction cycle. Post-reaction workup involves a simple acid wash which protonates the basic nitrogen of the catalyst, transferring it into the aqueous phase while the neutral organic product remains in the organic layer. This phase separation strategy effectively prevents catalyst contamination in the final API intermediate, ensuring that the impurity profile meets the stringent purity specifications required for pharmaceutical grade materials.
How to Synthesize (4S,5R)-Half Ester Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this laboratory-scale innovation into a viable manufacturing process. The procedure emphasizes the importance of maintaining anhydrous conditions initially to prevent non-selective hydrolysis of the anhydride by moisture. The reaction is typically initiated by dissolving the cyclic anhydride and the chiral catalyst in a solvent like MTBE, followed by the controlled addition of the alcohol. The simplicity of the workup—acidification, phase separation, and base precipitation of the catalyst—makes this route highly attractive for process chemists looking to streamline operations. For detailed standard operating procedures and specific stoichiometric ratios optimized for your specific substrate derivatives, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by dissolving the cyclic acid anhydride and the 9-epiquinine thiourea catalyst in a suitable organic solvent such as methyl tert-butyl ether (MTBE) or 1,4-dioxane under dry conditions.
- Add the alcohol reactant (e.g., methanol or propargyl alcohol) dropwise to the mixture at a controlled temperature between 0°C and 25°C, maintaining stirring for 10 to 36 hours to ensure complete conversion.
- Quench the reaction with dilute hydrochloric acid, separate the organic layer, and recover the chiral catalyst from the aqueous phase by pH adjustment for quantitative recycling, followed by solvent removal to isolate the pure half ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers tangible benefits that extend beyond mere chemical elegance. The primary value driver is the drastic simplification of the supply chain for chiral reagents. Traditional resolution methods require the continuous purchase of expensive, naturally sourced resolving agents like pseudoephedrine, the supply of which can be volatile and subject to regulatory scrutiny. By switching to a synthetic organocatalyst that can be recovered and reused, manufacturers can decouple their production costs from the fluctuating markets of natural alkaloids. This transition leads to substantial cost savings by eliminating the need for stoichiometric quantities of chiral pool materials and reducing the waste disposal costs associated with the unwanted enantiomer generated in resolution processes.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the quantitative recovery of the catalyst. Since the catalyst can be extracted from the aqueous waste stream simply by adjusting the pH and filtering the precipitated solid, the effective cost per kilogram of product decreases significantly with each cycle. Additionally, the use of commodity chemicals like methanol and MTBE as solvents, rather than specialized halogenated solvents or expensive buffer systems required for enzymatic routes, further drives down the raw material expenditure. The elimination of transition metals also removes the costly and time-consuming step of heavy metal scavenging and testing, which is a mandatory regulatory requirement for many pharmaceutical intermediates, thereby reducing overall processing time and analytical costs.
- Enhanced Supply Chain Reliability: Reliability in the supply of high-purity biotin intermediate is critical for downstream formulation companies. This synthetic route enhances reliability by utilizing robust, shelf-stable reagents that do not require cold-chain logistics, unlike enzymes which often need refrigeration and have limited shelf lives. The synthetic catalyst is chemically stable and can be stockpiled without degradation, ensuring that production schedules are not disrupted by reagent spoilage. Furthermore, the broad substrate scope of the catalyst means that the same catalytic system can potentially be used for various analogues, allowing for flexible manufacturing lines that can adapt to changing market demands for different vitamin H derivatives without requiring extensive retooling or requalification of new enzymatic systems.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat transfer and mixing challenges, but this reaction operates effectively at near-ambient temperatures, mitigating the risks of thermal runaway associated with exothermic reactions. The preference for MTBE, a solvent with favorable physical properties for separation and recycling, aligns with increasingly strict environmental regulations regarding volatile organic compounds (VOCs). The ability to recycle both the solvent and the catalyst minimizes the volume of hazardous waste generated, simplifying compliance with environmental discharge standards. This green profile not only reduces the environmental footprint but also future-proofs the manufacturing site against tightening regulatory frameworks, ensuring long-term operational continuity without the need for costly retrofits to waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organocatalytic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is essential for making informed decisions about technology transfer and process validation.
Q: What are the primary advantages of this organocatalytic method over traditional chiral resolution?
A: Unlike traditional resolution methods which are limited to a maximum theoretical yield of 50% and require expensive resolving agents like pseudoephedrine, this organocatalytic approach achieves yields exceeding 95% with ee values greater than 98%, while allowing for the quantitative recovery and reuse of the catalyst.
Q: Is the 9-epiquinine catalyst stable and recoverable for industrial use?
A: Yes, the patent explicitly details a recovery process where the catalyst is extracted from the acidic aqueous layer by adjusting the pH to 14 with sodium hydroxide, allowing it to precipitate as a solid that can be filtered, dried, and reused without significant loss of activity.
Q: What represent the optimal reaction conditions for scaling this synthesis?
A: The process operates efficiently at mild temperatures ranging from 0°C to 25°C using environmentally friendly solvents like MTBE, with a preferred molar ratio of anhydride to alcohol to catalyst of approximately 1:3-10:0.01-1.1, ensuring safety and ease of scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4S,5R)-Half Ester Supplier
The technological potential of the 9-epiquinine catalyzed synthesis represents a significant opportunity for optimizing the global supply of Biotin intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such complex asymmetric synthesis routes from patent literature into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high stereoselectivity observed in the lab is maintained at the plant scale. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of (4S,5R)-half ester meets the exacting standards required by top-tier pharmaceutical clients.
We invite you to collaborate with us to leverage this innovative chemistry for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that compares this organocatalytic route against your current manufacturing methods. We encourage you to contact us to obtain specific COA data from our pilot runs and comprehensive route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more efficient and cost-effective sourcing strategy.
