Scalable Manufacturing of Prulifloxacin via Novel Ionic Liquid Catalysis
Scalable Manufacturing of Prulifloxacin via Novel Ionic Liquid Catalysis
The global demand for fourth-generation quinolone anti-infectives continues to rise, driven by the persistent threat of bacterial resistance and the need for potent therapeutic agents. Prulifloxacin, a broad-spectrum antibiotic, represents a critical asset in modern pharmacotherapy, yet its manufacturing has historically been plagued by complex purification steps and environmental concerns. A groundbreaking preparation method detailed in patent CN101565428A introduces a transformative approach utilizing ionic liquids and Lewis acid catalysis to streamline production. This technology not only enhances the chemical efficiency of the synthesis but also aligns with the rigorous sustainability standards required by modern regulatory bodies. By shifting from traditional volatile organic solvents to recoverable ionic media, manufacturers can achieve superior product purity while drastically reducing the ecological footprint of the operation.
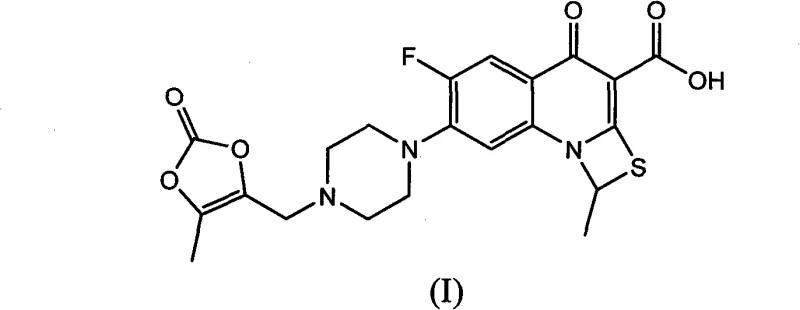
The strategic implementation of this novel pathway offers a compelling value proposition for pharmaceutical supply chains seeking reliability and quality. Unlike conventional routes that suffer from low overall yields and difficult crystallization profiles, this method leverages a 'one-pot' multi-charging strategy to synthesize key intermediates with exceptional precision. For R&D directors and procurement managers alike, understanding the mechanistic advantages of this route is essential for securing a stable supply of high-purity active pharmaceutical ingredients. The following analysis dissects the technical merits of this innovation, highlighting how it resolves long-standing bottlenecks in quinolone manufacturing and establishes a new benchmark for cost-effective, green chemistry in the antibiotic sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Prulifloxacin and its precursors has relied on pathways originating from 2,4,5-trifluorobenzoic acid or 3,4-difluoroaniline, which present significant operational challenges. These traditional methods are often characterized by cumbersome technological procedures involving multiple isolation steps, each introducing opportunities for yield loss and impurity accumulation. A critical drawback identified in prior art is the overall low yield, often calculating to merely around 40% when considering the common intermediate stages. Furthermore, Prulifloxacin exhibits instability under acidic conditions, complicating the crystallization and post-treatment of the crude product. This instability necessitates complex purification protocols to remove trace impurities, making it difficult to obtain the highly purified product required for clinical applications without substantial material degradation. Consequently, these factors render many conventional routes unsuitable for efficient industrialization, leading to higher production costs and inconsistent supply availability.
The Novel Approach
The innovative methodology described in the patent data overcomes these hurdles by employing [(3,4-difluorophenyl)amino](ethylthio)methylene malonic ester as a robust starting material. This route utilizes xylene or, more advantageously, ionic liquids as the reaction medium, coupled with Lewis acid catalysts to drive the cyclization efficiently. A standout feature of this approach is the synthesis of the 6,7-difluoro-4-hydroxy-2-(ethylthio)quinoline-3-ethyl formate intermediate via a streamlined process that removes alcohol byproducts through fractionation or nitrogen bubble distillation. This shifts the molecular equilibrium towards product formation, substantially increasing raw material utilization. Moreover, the subsequent conversion to the thiazinoquinoline core is achieved through a 'one-pot' reaction involving acetic anhydride, chlorosulfonic acid, and sodium carbonate. This telescoping of reactions eliminates the need for intermediate isolation, simplifying the technology and shortening the production cycle significantly while maintaining high product integrity.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the precise application of Lewis acid catalysis within an ionic liquid matrix to facilitate the formation of the quinoline ring system. In the initial step, the reaction of the aniline derivative with the malonic ester functionality is promoted by catalysts such as zinc chloride or indium chloride. The ionic liquid, specifically variants like [bmim]BF4, acts not merely as a solvent but as a stabilizing medium that enhances the electrophilicity of the reaction centers. This environment allows for the effective removal of ethanol generated during the condensation, driving the equilibrium forward according to Le Chatelier's principle. The use of nitrogen bubbling or fractionation at controlled temperatures, typically around 100°C to 140°C, ensures that the byproduct is continuously removed without degrading the sensitive fluorinated intermediates. This mechanistic control is vital for preventing the formation of polymeric byproducts that often plague high-temperature condensations in traditional solvents.
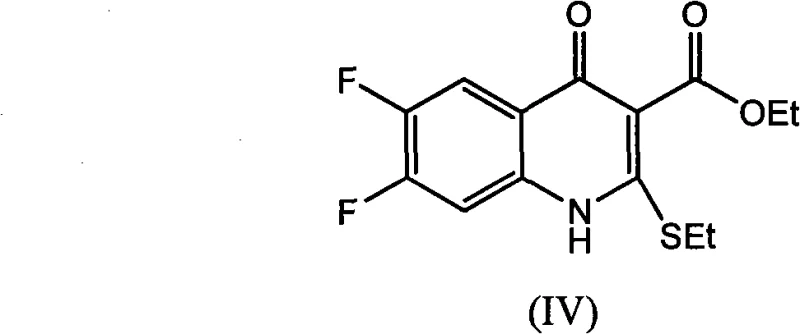
Furthermore, the impurity profile is rigorously controlled through the 'one-pot' synthesis of the bicyclic thiazinoquinoline system. By sequentially adding acetic anhydride, chlorosulfonic acid, and sodium carbonate in the same vessel, the process avoids exposing unstable intermediates to harsh workup conditions that could induce hydrolysis or decomposition. The chlorosulfonic acid facilitates the activation of the carbonyl group for subsequent ring closure, while the sodium carbonate neutralizes acidic byproducts in situ, maintaining a pH environment conducive to cyclization rather than degradation. This careful orchestration of reagent addition ensures that the final crude product possesses a purity exceeding 98.5% even before recrystallization. Such high initial purity reduces the burden on downstream purification units, minimizing solvent consumption and waste generation, which is a critical consideration for sustainable pharmaceutical manufacturing.
How to Synthesize Prulifloxacin Efficiently
The synthesis of Prulifloxacin via this advanced route involves a sequence of highly optimized chemical transformations designed for maximum efficiency and minimal waste. The process begins with the cyclization of the aniline precursor to form the quinoline core, followed by a telescoped reaction to construct the thiazine ring. Subsequent nucleophilic displacement with piperazine and final functionalization yield the target antibiotic. This methodology is particularly noted for its adaptability to large-scale production, utilizing reagents that are commercially available and reaction conditions that are safe to operate. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles, are outlined below to guide process engineers in replicating this high-yield pathway.
- Cyclization of [(3,4-difluorophenyl)amino](ethylthio)methylene malonic ester using Lewis acid catalyst in ionic liquid to form the quinoline core.
- One-pot reaction involving acetylation, chlorosulfonation, and cyclization to generate the thiazinoquinoline intermediate.
- Nucleophilic substitution with piperazine followed by hydrolysis and final condensation to yield Prulifloxacin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-based synthesis route offers profound strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures. By eliminating multiple isolation and purification steps associated with traditional methods, the facility requirements are minimized, and the throughput is significantly enhanced. The use of ionic liquids, which are non-volatile and thermally stable, removes the need for complex solvent recovery distillation columns typically required for volatile organic compounds. This not only lowers capital investment in equipment but also reduces energy consumption, contributing to a leaner and more cost-effective production model that can withstand market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the recyclability of the reaction medium. Since the ionic liquid can be recovered and reused multiple times without significant loss of catalytic activity, the recurring cost of solvents is virtually eliminated. Additionally, the high yield of the 'one-pot' reaction means that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold. The reduction in waste disposal costs, due to the eco-friendly nature of the solvents and the minimized generation of hazardous byproducts, further enhances the financial viability of this route for large-scale commercial operations.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of readily available starting materials such as 3,4-difluoroaniline derivatives and common Lewis acids, which are not subject to the same supply constraints as exotic reagents. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without compromising product quality, ensures consistent batch-to-batch performance. This reliability is crucial for meeting the strict delivery schedules of global pharmaceutical clients, reducing the risk of stockouts and ensuring that the pipeline for critical anti-infective medications remains uninterrupted even during periods of high demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant advancement in green chemistry. The replacement of toxic volatile solvents with ionic liquids aligns with increasingly stringent global environmental regulations, mitigating the risk of regulatory fines and shutdowns. The scalability of the process is proven by its successful demonstration in multi-kilogram batches, with straightforward translation to metric ton scales. The simplified workup procedures, involving simple filtration and water washing, reduce the complexity of waste treatment facilities, making it easier for manufacturing sites to maintain compliance with local discharge standards while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Prulifloxacin using this novel ionic liquid technology. These insights are derived directly from the patent specifications and experimental data, providing clarity on the process capabilities and quality outcomes. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this supply source into their existing procurement strategies.
Q: What are the advantages of using ionic liquids in Prulifloxacin synthesis?
A: Ionic liquids serve as both solvent and catalyst support, allowing for easier product separation, solvent recyclability, and significantly reduced environmental pollution compared to traditional volatile organic solvents.
Q: How does the one-pot synthesis method improve yield?
A: The one-pot method eliminates the need for isolating unstable intermediates, reducing material loss during filtration and purification steps, thereby increasing the overall cumulative yield of the final API.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the process utilizes readily available starting materials and robust reaction conditions (e.g., moderate temperatures, standard Lewis acids), making it highly adaptable for commercial scale production from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prulifloxacin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of essential antibiotics like Prulifloxacin. Our team of expert chemists has extensively evaluated this ionic liquid-catalyzed pathway and confirmed its potential for delivering superior product consistency. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest international standards for pharmaceutical intermediates and APIs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can enhance your production efficiency and secure your position in the competitive anti-infective market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
