Advanced One-Step Synthesis of 5-Bromoquinazoline Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking novel heterocyclic scaffolds that offer both structural complexity for drug discovery and functional utility as catalysts. A significant breakthrough in this domain is detailed in Chinese Patent CN112300178A, which discloses the preparation and application of a unique 5-bromoquinazoline derivative, specifically identified as methyl 3-[5,5'-dibromo-3-((6-bromo-2-oxo-1,2-dihydroquinazolin-4-amino)-2'-oxo(2,3'-spirobisindolyl)]formate, referred to herein as Compound I. This molecule represents a sophisticated spiro-bisindole architecture fused with a quinazolinone core, a structural motif highly prized in medicinal chemistry for its ability to interact with diverse biological targets. The patent highlights not only the synthetic accessibility of this compound but also its dual functionality as a potent organic catalyst and a promising anticancer agent, showing inhibitory activity against leukemia, liver, colon, and breast cancer cell lines. For R&D directors and procurement specialists, understanding the synthesis and potential of Compound I opens new avenues for developing next-generation therapeutics and efficient catalytic processes.
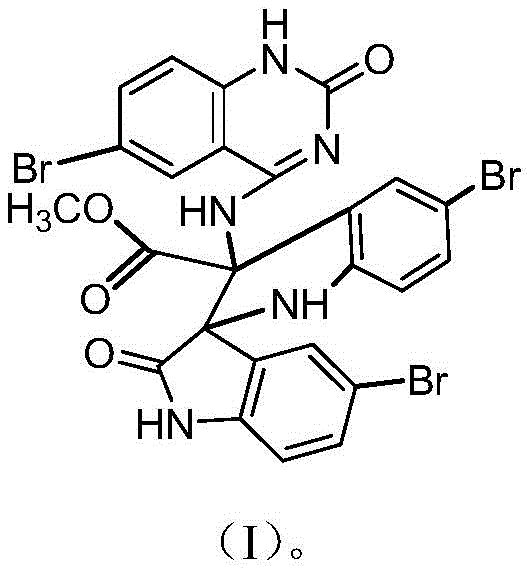
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex quinazoline derivatives, particularly those incorporating spiro-indole motifs, has been a laborious and multi-step endeavor. Conventional routes often involve the separate construction of the indole and quinazoline rings followed by difficult coupling reactions that require harsh conditions, expensive transition metal catalysts, or protecting group strategies. These multi-step sequences inherently suffer from cumulative yield losses, where the overall efficiency drops precipitously with each additional transformation. Furthermore, the purification of intermediates in such lengthy syntheses generates substantial chemical waste and increases the risk of introducing difficult-to-remove impurities, which is a critical concern for pharmaceutical intermediates intended for human use. The reliance on specialized reagents and the need for rigorous anhydrous or inert atmosphere conditions in many traditional protocols further escalate the operational costs and complexity, making the commercial scale-up of such molecules economically challenging for many manufacturers.
The Novel Approach
In stark contrast to these cumbersome traditional pathways, the methodology described in CN112300178A offers a remarkably streamlined one-step synthesis. The process utilizes readily available starting materials, specifically 5-bromoisatin and ammonium formate, reacting them in a simple anhydrous methanol solution. By heating the mixture under reflux for 48 hours, the reaction achieves a direct condensation that constructs the complex spiro-quinazoline framework in a single operation. This convergent approach eliminates the need for isolating unstable intermediates and drastically reduces the number of unit operations required. The simplicity of using ammonium formate as both a nitrogen source and a reducing agent in situ exemplifies green chemistry principles by minimizing reagent toxicity and waste. The resulting crude product can be efficiently purified via standard column chromatography using a dichloromethane and methanol system, yielding high-quality target crystals suitable for both catalytic applications and biological testing.
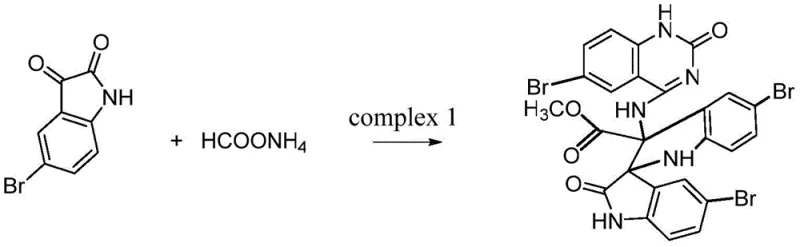
Mechanistic Insights into Spiro-Quinazoline Formation
The formation of Compound I involves a fascinating cascade of reactions initiated by the interaction between the carbonyl group of 5-bromoisatin and ammonium formate in a methanol medium. Although the full mechanistic pathway is complex, it is understood that the ammonium formate facilitates the conversion of the carbonyl functionality into an amino intermediate, which subsequently undergoes cyclization. This transformation is critical as it builds the quinazolinone ring system directly onto the indole scaffold. The presence of the bromine substituents on the aromatic rings plays a crucial role in directing the electronic properties of the molecule, enhancing its electrophilicity and stabilizing the final spiro-configuration through steric and electronic effects. The reaction proceeds through a condensation mechanism where methanol acts not just as a solvent but potentially participates in the stabilization of transition states, leading to the formation of the methyl ester moiety observed in the final structure. This one-pot transformation effectively merges three distinct molecular fragments into a single, rigid, three-dimensional architecture.
From an impurity control perspective, the robustness of this mechanism is advantageous. Because the reaction relies on thermodynamic control under reflux conditions, the system tends to favor the formation of the most stable crystalline product, which is Compound I. The use of ammonium formate minimizes the generation of heavy metal contaminants often associated with palladium or copper-catalyzed cross-couplings used in alternative routes. The patent data indicates that the final product can be obtained as distinct crystals with a melting point greater than 250°C, suggesting a high degree of lattice energy and purity. Elemental analysis data provided in the patent confirms the stoichiometry, with measured values for Carbon, Hydrogen, and Nitrogen closely matching theoretical calculations, thereby validating the structural integrity of the synthesized material. This high level of purity is essential for its subsequent application as a catalyst, where trace impurities could poison the catalytic cycle or alter selectivity.
How to Synthesize 5-Bromoquinazoline Derivative Efficiently
The synthesis of this high-value intermediate is designed for reproducibility and scalability, leveraging standard laboratory equipment that translates easily to pilot plant reactors. The protocol dictates precise stoichiometric ratios, utilizing 1.18g of 5-bromoisatin and 2.5503g of ammonium formate per 100mL of anhydrous methanol, ensuring that the reaction kinetics are optimized for maximum conversion. The extended reflux time of 48 hours allows the system to reach equilibrium, driving the condensation to completion despite the steric hindrance inherent in forming the spiro-center. Following the reaction, the workup procedure is straightforward, involving solvent removal to isolate a crude filter residue which is then subjected to chromatographic separation. This simplicity makes the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to integrate novel scaffolds into their portfolio without investing in specialized infrastructure.
- Weigh 1.18g of 5-bromoisatin and 2.5503g of ammonium formate into a 250mL round-bottom flask containing 100mL of anhydrous methanol.
- Heat the mixture under stirring and reflux conditions for 48 hours to facilitate the spiro-condensation reaction.
- Stop the reaction, remove solvent via rotation to obtain crude filter residue, and purify using column chromatography (dichloromethane/methanol 1: 1) to isolate target crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits centered around cost efficiency and supply reliability. The primary driver for cost reduction lies in the drastic simplification of the manufacturing process. By collapsing what would traditionally be a multi-step synthesis into a single reflux operation, manufacturers can significantly reduce labor hours, energy consumption, and solvent usage. The elimination of expensive transition metal catalysts, such as palladium complexes which are often required for similar heterocyclic constructions, removes a major cost center and eliminates the need for costly metal scavenging steps to meet regulatory limits for residual metals in pharmaceutical ingredients. Furthermore, the starting materials, 5-bromoisatin and ammonium formate, are commodity chemicals available from multiple global suppliers, mitigating the risk of raw material shortages and price volatility.
- Cost Reduction in Manufacturing: The economic impact of this one-step synthesis is profound. Traditional routes for spiro-quinazolines often involve low-yielding coupling steps that require expensive ligands and rigorous purification, inflating the cost of goods sold (COGS). In this novel process, the use of methanol as a solvent and ammonium formate as a reagent keeps input costs exceptionally low. The high thermal stability of the product (melting point >250°C) suggests that purification can potentially be achieved through recrystallization rather than expensive preparative HPLC on a large scale, further driving down processing costs. Additionally, the absence of toxic heavy metals simplifies waste disposal protocols, reducing environmental compliance costs associated with hazardous waste treatment.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-proprietary reagents. Unlike processes relying on custom-synthesized catalysts or sensitive organometallic reagents that have long lead times, the inputs for Compound I are shelf-stable and widely stocked. This ensures that production schedules are not held hostage by the availability of niche chemicals. The reaction conditions (reflux in methanol) are standard across the fine chemical industry, meaning that almost any capable manufacturing facility can execute this process without needing specialized high-pressure or cryogenic equipment. This universality allows for flexible sourcing strategies, enabling companies to qualify multiple manufacturing sites to ensure business continuity.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on homogeneous liquid-phase reactions in common solvents. Scaling from gram to kilogram or metric ton quantities primarily involves adjusting reactor volume and heat transfer capacity, without changing the fundamental chemistry. From an environmental standpoint, the atom economy is improved by avoiding protecting groups and extraneous coupling reagents. The waste stream is primarily composed of methanol and ammonium salts, which are easier to treat and recycle compared to the complex organic waste generated by multi-step syntheses involving chlorinated solvents and heavy metals. This aligns well with modern green chemistry mandates and helps pharmaceutical clients meet their sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this 5-bromoquinazoline derivative. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this compound into their existing pipelines, whether for drug discovery programs or as a specialized catalyst for organic synthesis transformations.
Q: What is the primary advantage of this synthesis route compared to traditional quinazoline methods?
A: The primary advantage is the one-step convergence. Traditional methods often require multiple steps to build the quinazoline core and install functional groups. This patent (CN112300178A) utilizes a direct condensation of 5-bromoisatin with ammonium formate in methanol, significantly reducing process complexity and potential impurity generation.
Q: What are the dual functional applications of Compound I described in the patent?
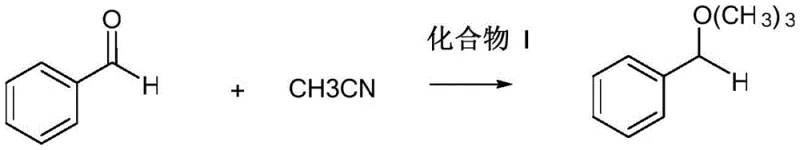
A: Compound I serves a dual purpose. Firstly, it acts as an effective organic catalyst, demonstrating significant conversion rates in benzaldehyde nitrile silicification (39%) and benzophenone hydrazone polymerization (76%). Secondly, it exhibits potent anticancer activity against leukemia (HL-60), liver cancer (SMMC-7721), colon cancer (SW480), and breast cancer (MCF-7) cell lines.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It utilizes standard reflux conditions in methanol, a common industrial solvent, and avoids exotic catalysts or extreme pressure/temperature requirements. The purification via standard column chromatography indicates a robust pathway that can be adapted for preparative HPLC or recrystallization on a metric ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromoquinazoline Derivative Supplier
The development of complex heterocyclic intermediates like Compound I requires a partner with deep technical expertise and a commitment to quality. NINGBO INNO PHARMCHEM stands ready to support your projects with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific reflux and chromatographic requirements of this synthesis, ensuring that we can deliver material that meets stringent purity specifications. We understand that for pharmaceutical applications, consistency is key; therefore, our rigorous QC labs employ advanced analytical techniques to verify the structural integrity and purity of every batch, ensuring that the spiro-quinazoline architecture remains intact and free from deleterious impurities.
We invite you to explore the potential of this versatile compound for your specific applications, whether as a lead structure for anticancer drug development or as a specialized catalyst for fine chemical synthesis. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized manufacturing processes can lower your overall procurement costs. Please contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and initiate a dialogue on how we can secure your supply chain for this high-value intermediate.
