Scalable One-Step Synthesis of Complex 5-Bromoquinazoline Derivatives for Pharmaceutical Applications
Scalable One-Step Synthesis of Complex 5-Bromoquinazoline Derivatives for Pharmaceutical Applications
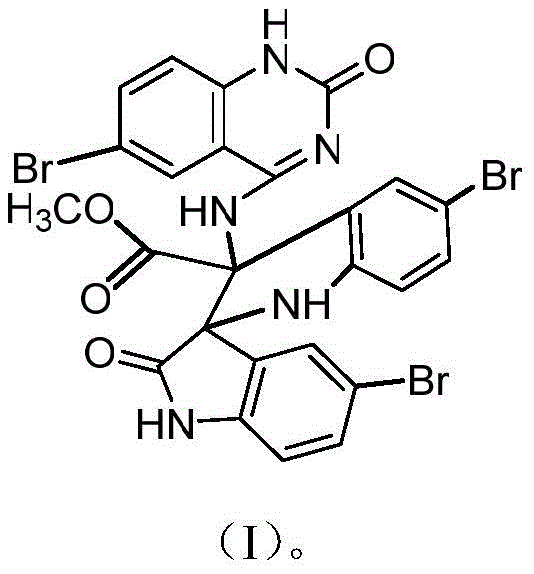
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable routes for complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN112300178B introduces a significant advancement in this domain by detailing the preparation and application of a novel 5-bromoquinazoline derivative, specifically identified as methyl 3-[5,5'-dibromo-3-((6-bromo-2-oxo-1,2-dihydroquinazolin-4-amino)-2'-oxo(2,3'-spirobisindolyl)] formate, referred to herein as Compound I. This technology addresses the persistent challenge of synthesizing highly functionalized quinazoline cores through a streamlined, one-step condensation reaction. Unlike traditional multi-step pathways that often suffer from low overall yields and extensive purification requirements, this method leverages the reactivity of 5-bromoisatin with ammonium formate in anhydrous methanol. The resulting compound exhibits a unique spiro-bisindole architecture fused with a quinazolinone moiety, offering distinct structural features that are highly valuable for medicinal chemistry campaigns targeting oncology and catalytic applications.
Beyond its structural novelty, Compound I demonstrates remarkable versatility, functioning effectively both as a potent anticancer agent and as a metal-free organocatalyst. For R&D directors evaluating new lead compounds, the presence of multiple bromine atoms on the scaffold provides excellent handles for further derivatization via cross-coupling reactions, thereby expanding the chemical space accessible for drug discovery. Furthermore, the patent data indicates that this derivative possesses intrinsic biological activity against a panel of human cancer cell lines, including leukemia, liver cancer, and breast cancer, suggesting immediate utility in preclinical screening programs. From a supply chain perspective, the simplicity of the reaction conditions—requiring only standard reflux equipment and common solvents—positions this molecule as an ideal candidate for reliable, high-volume manufacturing by a specialized pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazoline derivatives, particularly those with complex spiro-fused systems, has been plagued by synthetic inefficiencies that hinder commercial viability. Conventional methodologies often rely on the cyclization of anthranilic acid derivatives with isocyanates or amidines, processes that frequently necessitate harsh reaction conditions, such as high temperatures or strong acidic environments, which can degrade sensitive functional groups. Moreover, achieving the specific substitution patterns required for high-value API intermediates typically involves multiple protection and deprotection steps to manage the reactivity of amino and carbonyl groups. These additional synthetic operations not only increase the total processing time and labor costs but also significantly reduce the overall atom economy of the process. The accumulation of byproducts from these multi-stage sequences creates substantial downstream purification burdens, requiring extensive chromatography or recrystallization steps that lower the final isolated yield and generate large volumes of hazardous chemical waste, posing challenges for environmental compliance in large-scale manufacturing facilities.
The Novel Approach
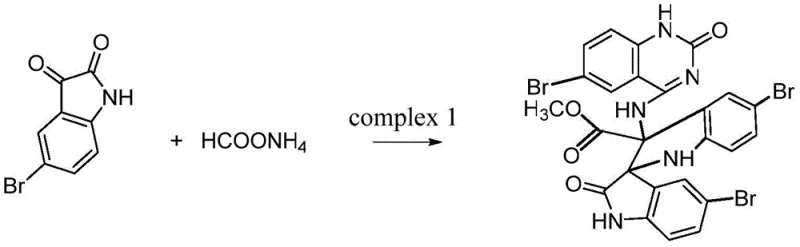
In stark contrast to these cumbersome traditional routes, the methodology disclosed in patent CN112300178B offers a paradigm shift through a direct, one-pot condensation strategy. By utilizing 5-bromoisatin as the foundational building block and reacting it directly with ammonium formate, the process bypasses the need for pre-functionalized precursors or complex catalytic systems involving expensive transition metals. The reaction proceeds smoothly in anhydrous methanol under reflux conditions, leveraging the inherent nucleophilicity of the formate species to facilitate ring closure and spiro-center formation simultaneously. This telescoped approach drastically reduces the number of unit operations required, effectively collapsing what would traditionally be a multi-day synthesis into a single heating cycle. The result is a process that is not only chemically elegant but also operationally superior, minimizing solvent consumption and simplifying the workup procedure to a straightforward filtration and column chromatography sequence, which is highly advantageous for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into the Formation of Spiro-Quinazoline Scaffolds
The formation of the complex spiro-bisindole quinazoline structure in Compound I involves a fascinating cascade of condensation and cyclization events that merit detailed mechanistic consideration for process optimization. Although the patent text alludes to potential palladium-mediated pathways in certain mechanistic discussions, the primary embodiment relies on a thermal condensation driven by the reactivity of the isatin carbonyl group with the ammonium source. The initial step likely involves the nucleophilic attack of ammonia (generated in situ from ammonium formate decomposition) on the ketone carbonyl of the 5-bromoisatin, forming an imine intermediate. Subsequent intramolecular cyclization involving the adjacent amide nitrogen and the formate carbon leads to the formation of the quinazolinone ring system. The unique spiro-connection at the C3 position of the indole ring suggests a rearrangement or a concurrent reaction with a second equivalent of the isatin derivative, facilitated by the specific electronic properties imparted by the bromine substituents. Understanding these electronic effects is crucial for R&D teams, as the electron-withdrawing nature of the bromine atoms activates the aromatic ring towards nucleophilic attack while stabilizing the transition states involved in the spiro-center formation.
Furthermore, the control of impurities in this synthesis is inherently managed by the stoichiometry and the choice of solvent. The use of anhydrous methanol is critical, as the presence of water could hydrolyze the reactive imine intermediates or promote the formation of open-chain byproducts rather than the desired cyclic spiro-compound. The reaction temperature, maintained at the reflux point of methanol, provides sufficient thermal energy to overcome the activation barrier for the cyclization without inducing thermal degradation of the sensitive heterocyclic core. For quality control purposes, the distinct crystallographic data provided in the patent, including specific bond lengths and angles, serves as a rigorous fingerprint for verifying the structural integrity of the product. This level of structural definition ensures that any batch produced will meet the stringent purity specifications required for pharmaceutical applications, minimizing the risk of genotoxic impurities or structurally related analogs that could complicate regulatory filings.
How to Synthesize 5-Bromoquinazoline Derivative Efficiently
The practical execution of this synthesis is designed to be accessible to standard organic chemistry laboratories while maintaining the rigor necessary for reproducible results. The protocol begins with the precise weighing of 5-bromoisatin and ammonium formate, which are then suspended in anhydrous methanol within a standard round-bottom flask equipped with a reflux condenser. The mixture is heated to maintain a gentle boil with continuous magnetic stirring to ensure homogeneous heat transfer and mass transport throughout the reaction vessel. After a prolonged reaction period of approximately 48 hours, the completion of the reaction is monitored, and the mixture is allowed to cool to room temperature. The resulting precipitate is collected via filtration, yielding a crude solid that contains the target spiro-compound along with minor soluble impurities. To achieve the high purity required for biological testing or catalytic applications, the crude material is subjected to column chromatography using a binary solvent system of dichloromethane and methanol, which effectively separates the product based on polarity differences.
- Combine 5-bromoisatin and ammonium formate in anhydrous methanol within a round-bottom flask under inert atmosphere conditions.
- Heat the reaction mixture to reflux with continuous stirring for approximately 48 hours to ensure complete conversion.
- Isolate the crude product via filtration and purify using column chromatography with a dichloromethane and methanol solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary driver for cost efficiency lies in the drastic simplification of the raw material portfolio; by relying on commodity chemicals like ammonium formate and commercially available 5-bromoisatin, the process eliminates the dependency on exotic, high-cost reagents that are often subject to volatile market pricing and long lead times. This stability in raw material sourcing translates directly into more predictable budgeting and reduced exposure to supply chain disruptions. Additionally, the elimination of transition metal catalysts, such as palladium or platinum, which are frequently required in alternative quinazoline syntheses, removes the necessity for expensive metal scavenging steps and the associated regulatory burden of proving low residual metal levels in the final API. This qualitative reduction in processing complexity inherently lowers the operational expenditure per kilogram of product produced.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the consolidation of multiple synthetic steps into a single operation, which significantly reduces labor hours, energy consumption for heating and cooling cycles, and solvent usage. By avoiding the isolation of unstable intermediates, the process minimizes material loss during transfer and purification stages, leading to a higher effective throughput of the manufacturing facility. The absence of precious metal catalysts further contributes to substantial cost savings, as there is no need to recover or dispose of expensive heavy metals, nor is there a requirement for specialized equipment resistant to corrosive metal salts. This lean manufacturing approach allows for a more competitive pricing structure for the final intermediate, providing a clear margin advantage for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is a critical factor for maintaining uninterrupted supply to pharmaceutical clients. Since the reagents are stable and widely produced on a global scale, the risk of raw material shortages is minimized, ensuring that production schedules can be met reliably even during periods of market volatility. The simplicity of the workup procedure, involving basic filtration and standard chromatography, means that the process can be easily transferred between different manufacturing sites or scaled up without requiring highly specialized technical expertise or custom-built reactor configurations. This flexibility enhances the resilience of the supply chain, allowing for rapid response to fluctuations in demand for this key pharmaceutical intermediate.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is significantly greener than traditional alternatives due to the reduced generation of chemical waste. The high atom economy of the condensation reaction means that a larger proportion of the starting materials end up in the final product, reducing the volume of effluent that requires treatment. Furthermore, the solvents used, primarily methanol and dichloromethane, are well-understood and can be efficiently recovered and recycled using standard distillation infrastructure, aligning with modern sustainability goals. The ability to scale this process from gram-scale laboratory synthesis to multi-kilogram or metric-ton production is facilitated by the lack of exothermic hazards or pressure requirements, making it a safe and compliant choice for large-scale industrial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this 5-bromoquinazoline derivative. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is essential for partners looking to integrate this intermediate into their own development pipelines or supply networks.
Q: What are the primary advantages of this synthesis route compared to traditional methods?
A: The primary advantage is the significant simplification of the process into a single step, eliminating the need for complex multi-stage protection and deprotection sequences often required for quinazoline scaffolds. This reduces operational time and minimizes waste generation.
Q: Can this compound be utilized effectively as an organocatalyst?
A: Yes, experimental data confirms its efficacy as a Lewis base catalyst, demonstrating substantial conversion rates in nitrile silicification reactions and the self-polymerization of benzophenone hydrazone without requiring heavy metal additives.
Q: Is the production of this intermediate scalable for commercial supply?
A: The process utilizes readily available starting materials like 5-bromoisatin and ammonium formate in common solvents like methanol, making it highly amenable to scale-up from laboratory gram quantities to metric ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromoquinazoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development of novel therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering this complex 5-bromoquinazoline derivative with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify structural integrity and impurity profiles. Our capability to handle complex heterocyclic synthesis allows us to offer a reliable supply of this key building block, supporting your R&D timelines and commercial launch goals with unwavering consistency.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your project economics. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can add value to your supply chain, ensuring you have access to high-purity pharmaceutical intermediates when you need them most.
