Optimizing Clodinafop-propargyl Production: A Technical Breakthrough in Aryloxyphenoxypropionate Herbicide Synthesis
The global demand for high-efficiency, low-toxicity herbicides continues to drive innovation in agrochemical intermediate synthesis, particularly for the aryloxyphenoxypropionate class. Patent CN114292228A introduces a transformative synthetic methodology for Clodinafop-propargyl, a critical active ingredient used extensively for controlling grassy weeds in cereal crops. This technical disclosure outlines a robust two-step process that leverages a unique catalytic system involving tetrabutylammonium fluoride (TBAF) and potassium hydroxide (KOH) in dimethylformamide (DMF). Unlike traditional routes that often struggle with byproduct management and purification bottlenecks, this novel approach ensures a streamlined workflow that maintains reaction temperatures between 70°C and 73°C. For R&D directors and process chemists, the significance lies in the ability to achieve conversion rates of 100% in the initial coupling step while minimizing downstream processing complexity. As a reliable agrochemical intermediate supplier, understanding these mechanistic nuances is essential for evaluating the scalability and economic viability of sourcing this key herbicide precursor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Clodinafop-propargyl and related aryloxyphenoxypropionic acids has relied heavily on the use of potassium carbonate (K2CO3) as the acid-binding agent during the nucleophilic aromatic substitution steps. While chemically feasible, this conventional approach presents significant operational challenges that impact both yield and purity profiles. The reaction of phenolic substrates with halogenated pyridines in the presence of carbonate bases often leads to the excessive generation of inorganic salts, which complicates the filtration and isolation processes. Furthermore, a persistent issue in these traditional protocols is the formation of polymeric tars or dark-colored byproducts that are notoriously difficult to separate from the desired product. These impurities not only lower the overall yield but also necessitate energy-intensive purification steps such as column chromatography or multiple recrystallizations, which are impractical for cost reduction in herbicide manufacturing on an industrial scale. The accumulation of solid waste and the difficulty in achieving consistent optical purity have long been pain points for supply chain managers seeking efficient production routes.
The Novel Approach
The methodology detailed in patent CN114292228A represents a paradigm shift by replacing the traditional carbonate base with a synergistic system of potassium hydroxide and TBAF. This strategic modification fundamentally alters the reaction environment, effectively suppressing the formation of intractable tars and significantly reducing the volume of salt byproducts generated. By utilizing KOH, the deprotonation of the phenolic hydroxyl group is more efficient, while the fluoride ion from TBAF acts as a potent nucleophilic catalyst, enhancing the displacement of the fluorine atom on the pyridine ring. The result is a cleaner reaction profile where the resulting potassium fluoride byproduct is highly soluble in the aqueous workup phase, allowing for its removal through simple water washing rather than complex filtration of sludge. This simplification of the post-reaction treatment directly translates to higher throughput and reduced solvent consumption, addressing critical concerns regarding commercial scale-up of complex agrochemical intermediates. The process consistently delivers yields exceeding 90% with high optical purity, demonstrating superior performance over legacy methods.
Mechanistic Insights into TBAF-Catalyzed Nucleophilic Aromatic Substitution
The core chemical transformation in this synthesis involves a nucleophilic aromatic substitution (SnAr) where the phenoxide anion attacks the electron-deficient pyridine ring. In the presence of TBAF, the fluoride ion plays a dual role: it acts as a phase transfer catalyst facilitating the interaction between the organic substrate and the inorganic base, and it potentially activates the aromatic ring towards nucleophilic attack by forming a transient Meisenheimer-like complex. The specific positioning of the chlorine and fluorine atoms on the pyridine ring dictates the regioselectivity, ensuring that substitution occurs exclusively at the 2-position adjacent to the nitrogen atom. The mild thermal conditions of 70°C to 73°C are sufficient to overcome the activation energy barrier without promoting thermal degradation or side reactions that typically occur at higher temperatures. This precise control over reaction kinetics is vital for maintaining the integrity of the chiral center in the propionic acid moiety, preventing racemization which would render the herbicide ineffective.
Impurity control is another critical aspect where this mechanism excels. In traditional carbonate-mediated reactions, the basicity can sometimes lead to hydrolysis of the ester groups or elimination reactions if temperatures fluctuate. However, the KOH/TBAF system provides a buffered environment that minimizes these risks. The absence of tar formation suggests that the fluoride ion effectively scavenges reactive intermediates that might otherwise polymerize. Additionally, the solubility characteristics of the byproducts are optimized; potassium fluoride remains in solution during the aqueous quench, whereas the target Clodinafop-propargyl precipitates out or can be easily extracted. This distinct separation behavior is a direct consequence of the chosen reagents and allows for the production of high-purity OLED material grade intermediates or agrochemical actives with minimal additional refining. Understanding this mechanistic advantage allows procurement teams to appreciate the value proposition of suppliers who adopt this advanced technology.
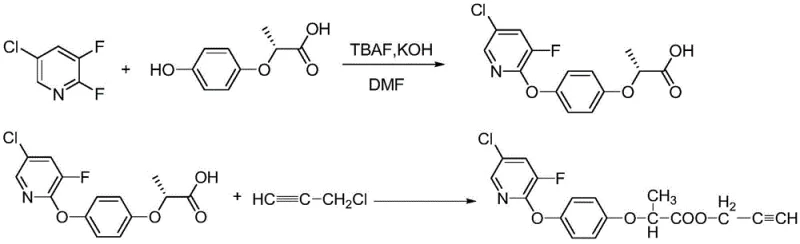
How to Synthesize Clodinafop-propargyl Efficiently
Implementing this synthesis requires strict adherence to the stoichiometric ratios and temperature profiles outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction mixture in DMF, where the precise molar ratio of TBAF to the hydroxy-acid substrate is maintained between 0.06:1 and 0.08:1 to optimize catalytic activity without excess cost. The subsequent addition of 2,3-difluoro-5-chloropyridine must be controlled to manage the exotherm, followed by a sustained incubation period at 70-73°C to drive the first coupling to completion. Once the intermediate acid is formed, the second step involves alkylation with propargyl chloride under similar thermal conditions. The detailed standardized synthetic steps, including specific addition rates and workup procedures, are provided in the guide below to assist technical teams in replicating this high-yield pathway.
- Dissolve TBAF and potassium hydroxide in DMF, then add R-2-(4-hydroxyphenoxy)propionic acid and heat to 70-73°C before adding 2,3-difluoro-5-chloropyridine.
- Maintain reaction at 70-73°C under normal pressure to generate the intermediate R-2-(4-(5-chloro-3-fluoropyridin-2-yloxy)phenoxy)propionic acid.
- Cool to 50-53°C, add propargyl chloride, return to 70-73°C for alkylation, then remove solvent and precipitate product with water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the TBAF/KOH catalytic system offers substantial strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the purification workflow, which directly correlates to reduced operational expenditures and faster batch turnover times. By eliminating the need for extensive filtration of tar-like residues and reducing the volume of inorganic salt waste, manufacturers can significantly lower their waste disposal costs and environmental compliance burdens. This efficiency gain is crucial for maintaining competitive pricing in the volatile agrochemical market, where margin pressures are constant. Furthermore, the reliance on commercially available and stable reagents such as DMF, KOH, and TBAF ensures a resilient supply chain that is less susceptible to the shortages often associated with exotic or highly specialized catalysts.
- Cost Reduction in Manufacturing: The elimination of difficult-to-separate tars and the reduction of salt byproducts mean that the downstream processing units require less energy and fewer solvents for purification. Instead of multiple recrystallization steps or chromatographic separations, a simple water wash is sufficient to remove soluble potassium fluoride, leading to substantial cost savings in utility consumption and labor. This streamlined process flow enhances the overall economic feasibility of producing Clodinafop-propargyl, allowing suppliers to offer more competitive pricing structures without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, including potassium hydroxide and DMF, are commodity chemicals with robust global supply networks, reducing the risk of production stoppages due to raw material scarcity. The mild reaction conditions (70-73°C) also reduce the stress on reactor equipment, extending asset life and minimizing maintenance downtime. For supply chain planners, this translates to more predictable lead times and the ability to scale production volumes rapidly in response to seasonal demand fluctuations in the herbicide market, ensuring continuous availability for downstream formulators.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solid waste generation aligns with increasingly stringent global regulations on industrial effluent and hazardous waste. The process generates significantly less hazardous sludge compared to carbonate-based methods, simplifying the permitting process for manufacturing facilities. Moreover, the high yield (>90%) means that less raw material is wasted per unit of product, improving the atom economy of the synthesis. This sustainability profile is increasingly important for multinational agribusinesses seeking to partner with suppliers who demonstrate a commitment to green chemistry principles and responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Clodinafop-propargyl using the patented TBAF/KOH method. These insights are derived directly from the experimental data and beneficial effects described in patent CN114292228A, providing clarity on yield expectations, purification mechanisms, and scalability factors. Understanding these details helps stakeholders make informed decisions regarding supplier selection and process validation.
Q: What is the primary advantage of using TBAF and KOH over traditional potassium carbonate in this synthesis?
A: The use of TBAF as a catalyst combined with potassium hydroxide significantly reduces salt generation and eliminates the formation of difficult-to-separate tar, allowing for simplified purification via water washing.
Q: What yield and purity levels can be achieved with this patented method?
A: According to patent CN114292228A, this method achieves a yield of at least 90%, with optical purity and content reaching approximately 97%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates at mild temperatures (70-73°C) and utilizes common solvents like DMF, making it highly scalable and safe for industrial production without requiring extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clodinafop-propargyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the TBAF-catalyzed route are fully realized in our manufacturing plants. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify optical purity and chemical content, guaranteeing that every batch of Clodinafop-propargyl meets the exacting standards required for modern herbicide formulations.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can lower your total cost of ownership. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring full transparency and confidence in our capability to serve as your long-term strategic partner for high-quality agrochemical intermediates.
