Advanced Synthetic Route for High-Purity Iopromide Using Diacetoxy Intermediate Technology
Introduction to Advanced Iopromide Manufacturing
The global demand for non-ionic iodine contrast media continues to rise, driven by the increasing prevalence of diagnostic imaging procedures in modern healthcare. However, the manufacturing of these complex molecules, particularly Iopromide, has historically been plagued by significant purification challenges that impact both yield and final product quality. A pivotal breakthrough in this domain is detailed in Chinese Patent CN103570580A, which discloses a novel preparation method for high-purity Iopromide. This technology addresses the critical bottleneck of removing polar byproducts that are structurally similar to the target molecule, a problem that has long hindered efficient commercial production. By introducing a specific diacetoxy intermediate, designated as Formula (19), the process enables a purification strategy based on crystallization rather than laborious chromatography or repeated recrystallization. This approach not only streamlines the synthetic workflow but also ensures that the final active pharmaceutical ingredient (API) meets the stringent impurity profiles required by major pharmacopoeias such as the USP and EP.
For pharmaceutical manufacturers and procurement specialists, understanding the mechanistic advantages of this route is essential for securing a reliable supply chain of high-quality contrast agents. The core innovation lies in the temporary modification of the hydrophilic hydroxyl groups on the side chains. In traditional synthesis, these polar groups often trap impurities within the crystal lattice or create emulsions during workup, leading to substantial yield losses. The patented method circumvents these issues by masking the hydroxyls as acetates, thereby altering the physicochemical properties of the intermediate just enough to facilitate the exclusion of unwanted side products. This strategic manipulation of solubility and crystallinity represents a significant leap forward in process chemistry, offering a robust solution for producing Iopromide with superior purity specifications while minimizing waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iopromide has relied on direct acylation strategies, such as those described in US Patent No. 4364921. In these conventional pathways, the starting material, 5-amino-2,4,6-triiodoisophthaloyl chloride, is reacted sequentially with methoxyacetyl chloride and various amino-alcohols like 3-amino-1,2-propanediol. While conceptually straightforward, this direct approach suffers from severe chemoselectivity issues. The presence of two reactive acyl chloride moieties on the aromatic ring creates a high probability of forming symmetrical and asymmetrical byproducts, such as the compound shown in Formula (5). These byproducts possess polarity and solubility characteristics that are nearly identical to the desired Iopromide, making their separation extremely difficult without resorting to excessive solvent usage or complex purification techniques.
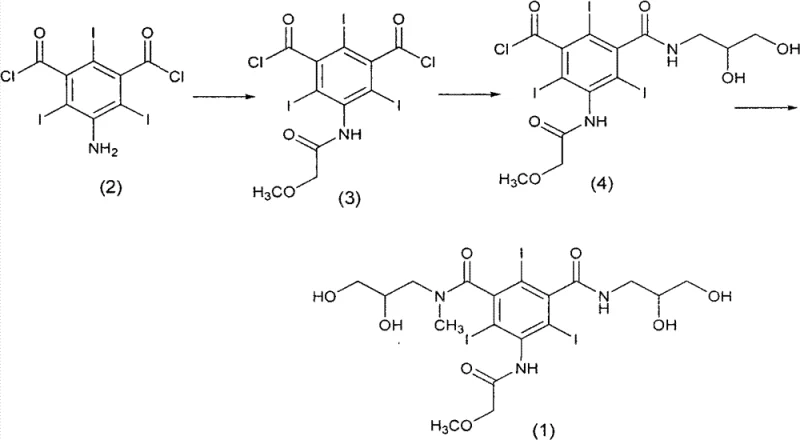
Furthermore, the direct reaction with amino-alcohols often results in a mixture of regioisomers and oligomeric impurities that are resistant to standard washing protocols. To achieve the necessary pharmaceutical grade purity, manufacturers are forced to perform multiple recrystallization steps, which drastically reduces the overall process yield and increases the environmental footprint due to high solvent consumption. The accumulation of these polar impurities not only compromises the safety profile of the contrast agent but also creates significant bottlenecks in manufacturing throughput. Consequently, the industry has long sought a method that could decouple the formation of the amide bonds from the purification challenges associated with the free hydroxyl groups, a gap that previous technologies failed to adequately fill.
The Novel Approach
The innovative methodology presented in CN103570580A fundamentally restructures the synthesis timeline to prioritize purification efficiency. Instead of attempting to purify the final diol product directly, the process introduces a strategic acetylation step that converts the crude Iopromide precursor into the diacetoxy intermediate, Formula (19). This transformation is not merely a derivatization but a critical purification enabler. By converting the hydrophilic hydroxyl groups into more lipophilic acetoxy groups, the intermediate gains distinct crystallization properties that allow it to be easily separated from polar impurities, such as the fully hydroxylated byproduct Formula (20), which remains soluble in the mother liquor. This single step effectively acts as a "chemical filter," removing a wide spectrum of contaminants that would otherwise persist through to the final API.
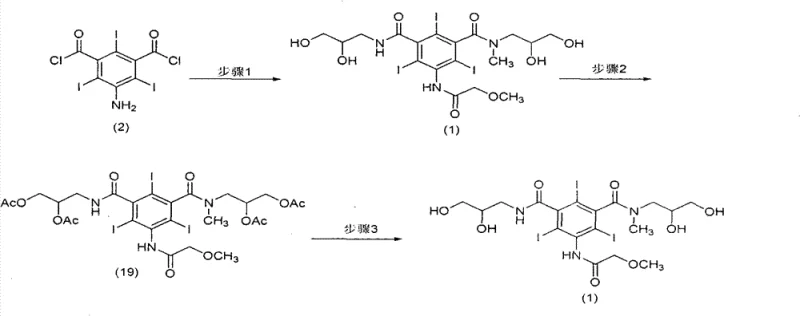
Following the isolation of the pure diacetoxy intermediate, the final step involves a mild hydrolysis under alkaline conditions to regenerate the free hydroxyl groups, yielding high-purity Iopromide. This deprotection step is highly efficient and does not reintroduce the impurities that were previously removed. The result is a process that delivers significantly higher yields compared to conventional methods, as the material loss associated with repetitive recrystallizations is eliminated. For supply chain managers, this translates to a more predictable and cost-effective manufacturing process, where the complexity of downstream processing is drastically reduced. The ability to obtain a product with purity levels exceeding 99% through simple filtration and hydrolysis underscores the commercial viability and technical superiority of this novel approach.
Mechanistic Insights into Protective Group Strategy
The success of this synthetic route hinges on the precise application of protective group chemistry to modulate molecular solubility. In the context of polyhydroxylated compounds like Iopromide, the hydrogen bonding network formed by the free hydroxyl groups creates a high-energy lattice that often incorporates solvent molecules and impurities. By reacting the crude diol precursor with acetic anhydride in the presence of a base like pyridine, the hydroxyl protons are replaced by acetyl groups. This modification disrupts the extensive hydrogen bonding capability of the molecule, reducing its polarity and increasing its compatibility with organic solvents like dichloromethane. This shift in physicochemical behavior is crucial because it allows the desired intermediate to crystallize out of solution while leaving highly polar impurities, which lack the acetyl groups or have different substitution patterns, dissolved in the supernatant.
Moreover, the stability of the acetyl groups under the reaction conditions ensures that the amide linkages on the aromatic core remain intact. The subsequent hydrolysis step is carefully controlled at temperatures between 40-60°C in an aqueous alkaline environment. These conditions are sufficiently vigorous to cleave the ester bonds of the acetyl groups but mild enough to prevent the hydrolysis of the robust amide bonds connecting the side chains to the triiodobenzene core. This chemoselectivity is vital for maintaining the structural integrity of the contrast agent. The mechanism essentially creates a "purification window" where the molecule exists in a transient state (Formula 19) that is thermodynamically favored to exclude impurities, before being reverted to its bioactive form. This level of control over the impurity profile is what distinguishes this method from prior art, providing a robust mechanism for consistent quality assurance.
How to Synthesize High-Purity Iopromide Efficiently
Implementing this advanced synthesis route requires careful attention to stoichiometry and reaction conditions to maximize the benefits of the intermediate purification strategy. The process begins with the formation of the crude diol precursor, followed by the critical acetylation step where the molar ratios of acetic anhydride and pyridine are optimized to ensure complete conversion without excessive reagent waste. The subsequent crystallization of the diacetoxy intermediate is the key operational unit, where parameters such as cooling rate and solvent volume must be tuned to achieve optimal crystal growth and impurity rejection. Finally, the hydrolysis step requires precise pH control to ensure complete deprotection while minimizing degradation. For detailed operational parameters and specific equipment requirements, please refer to the standardized synthesis guide below.
- React 5-amino-2,4,6-triiodoisophthaloyl chloride with methoxyacetyl chloride to form the mono-amide intermediate, followed by reaction with 2,3-dihydroxypropylamine and 3-methylamino-1,2-propanediol to generate the crude diol precursor.
- Convert the crude diol precursor into the diacetoxy intermediate (Formula 19) using acetic anhydride and pyridine, allowing for the crystallization and removal of polar byproducts.
- Hydrolyze the purified diacetoxy intermediate in an alkaline aqueous solution at 40-60°C to remove the acetyl protecting groups and yield high-purity Iopromide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this diacetoxy intermediate route offers transformative advantages for the procurement of Iopromide and related contrast media intermediates. Traditional manufacturing processes are often characterized by low overall yields due to the cumulative losses incurred during multiple purification stages. By consolidating the purification into a single, high-efficiency crystallization step, this new method significantly reduces the amount of starting material required to produce a given quantity of API. This improvement in atom economy directly correlates to a reduction in raw material costs, which is a primary driver for procurement managers seeking to optimize their bill of materials. Furthermore, the elimination of complex chromatographic separations or extensive solvent swaps simplifies the manufacturing infrastructure, allowing for production in standard multipurpose reactors without the need for specialized purification equipment.
- Cost Reduction in Manufacturing: The streamlined workflow eliminates the need for large volumes of solvents typically used in repetitive recrystallizations, leading to substantial savings in both material costs and waste disposal fees. The higher yield per batch means that fixed costs, such as labor and energy, are distributed over a larger output of saleable product, effectively lowering the cost of goods sold (COGS). Additionally, the use of common reagents like acetic anhydride and pyridine ensures that the supply chain for inputs remains stable and cost-effective, avoiding reliance on exotic or expensive catalysts.
- Enhanced Supply Chain Reliability: The robustness of the crystallization-based purification makes the process less sensitive to minor variations in reaction conditions, resulting in more consistent batch-to-batch quality. This reliability is critical for maintaining continuous supply to downstream formulation partners, reducing the risk of stockouts caused by failed batches or off-spec material. The simplified workup also shortens the overall cycle time per batch, increasing the manufacturing capacity of existing facilities and allowing suppliers to respond more agilely to fluctuations in market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, as the physical principles governing the crystallization of the diacetoxy intermediate translate linearly from laboratory to pilot and commercial scales. The reduction in solvent usage and the avoidance of heavy metal catalysts or hazardous reagents align with green chemistry principles, facilitating easier regulatory approval and reducing the environmental compliance burden. This sustainability aspect is increasingly important for multinational corporations aiming to meet their corporate social responsibility (CSR) goals while securing a long-term supply of essential medical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Iopromide synthesis method. These insights are derived directly from the patent data and are intended to clarify the operational benefits and chemical rationale behind the diacetoxy intermediate strategy. Understanding these details can help technical teams evaluate the feasibility of adopting this route for their own manufacturing needs or for qualifying new suppliers.
Q: How does the diacetoxy intermediate improve Iopromide purity?
A: The introduction of acetyl groups increases the lipophilicity and crystallinity of the intermediate, allowing polar byproducts (such as Formula 20) to remain in the mother liquor during crystallization, which is difficult to achieve with the highly polar diol forms.
Q: What are the main impurities removed by this new process?
A: This process effectively removes polar byproducts generated during the acylation steps, specifically compounds like N,N'-bis-(2,3-dihydroxypropyl)-2,4,6-triiodoisophthalamide derivatives that co-elute or co-crystallize in conventional methods.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the route replaces complex chromatographic purifications or extensive recrystallizations with a simple filtration step after acetylation, significantly simplifying the workup and making it highly amenable to multi-kilogram or ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iopromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the production of high-purity diagnostic agents like Iopromide requires not only advanced chemical knowledge but also rigorous process control. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactor systems capable of handling the specific temperature and mixing requirements of the acetylation and hydrolysis steps described in this patent. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of Iopromide intermediate or API meets the exacting standards of the global pharmaceutical industry, including USP and EP monographs.
We invite procurement leaders and R&D directors to collaborate with us to leverage this innovative synthesis technology for your supply chain. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate the superior purity profile achievable through this diacetoxy intermediate method. Let us help you secure a sustainable and cost-efficient supply of high-quality Iopromide for your diagnostic imaging portfolio.
