Advanced Manufacturing of Rosuvastatin Calcium: A Technical Breakthrough in Stereocontrol and Process Efficiency
Advanced Manufacturing of Rosuvastatin Calcium: A Technical Breakthrough in Stereocontrol and Process Efficiency
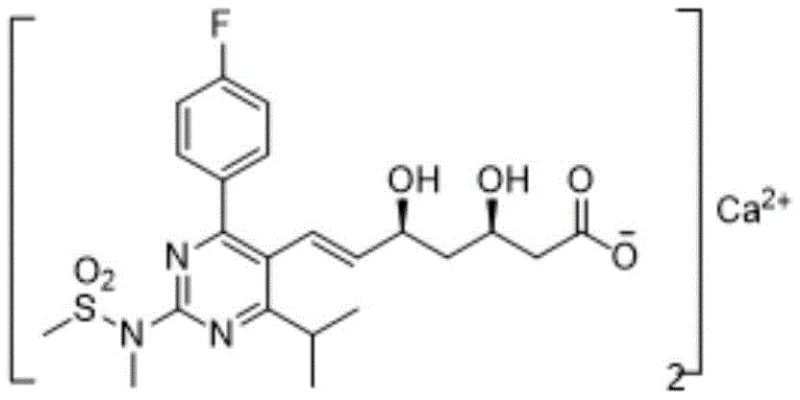
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of high-value statins, particularly Rosuvastatin Calcium, a potent HMG-CoA reductase inhibitor. Patent CN111454216B introduces a transformative synthetic methodology that addresses long-standing challenges in stereochemical control and intermediate purification. By integrating an Evans prosthetic group strategy with a Julia olefination protocol, this novel route circumvents the limitations of traditional Wittig-based syntheses. The technical significance of this patent lies in its ability to generate key intermediates as stable solids rather than oils, thereby facilitating rigorous quality control and enabling the production of high-purity active pharmaceutical ingredients (APIs) suitable for global regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Rosuvastatin Calcium has relied heavily on Wittig olefination to construct the critical carbon-carbon double bond in the side chain. However, as illustrated in prior art such as EP0521471A1 and WO0049014A1, these conventional routes suffer from inherent thermodynamic limitations that result in poor E/Z selectivity. The formation of Z-isomers as byproducts necessitates complex and yield-depleting purification steps, often involving preparative chromatography which is impractical at a commercial scale. Furthermore, many intermediates in these legacy pathways exist as viscous oils, making them susceptible to degradation, difficult to characterize, and challenging to transport between manufacturing sites without significant loss of quality. The reliance on oxidation systems to convert olefins to aldehydes in some alternative routes further introduces impurities that compromise the final drug substance purity.
The Novel Approach
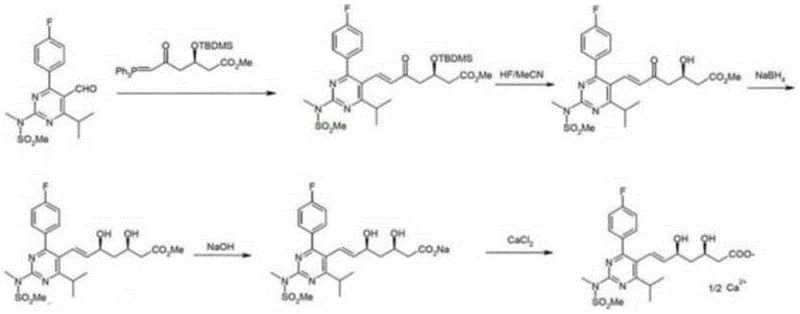
In stark contrast, the methodology disclosed in Patent CN111454216B leverages a Julia olefination strategy coupled with chiral auxiliary technology to achieve exceptional process control. The introduction of an Evans oxazolidinone moiety transforms the physical state of critical intermediates, specifically Compounds 5 and 6, from oils into crystalline solids. This phase change is pivotal for manufacturing, as crystallization serves as a powerful purification tool, effectively rejecting impurities and isomers without the need for expensive chromatographic media. The new route streamlines the synthesis by utilizing readily available reagents and avoiding the harsh oxidative conditions found in older methods. By shifting the paradigm from liquid to solid-phase intermediate processing, this approach not only enhances the chemical purity of the final API but also drastically simplifies the logistical handling and storage requirements for bulk intermediates.
Mechanistic Insights into Julia Olefination and Chiral Induction
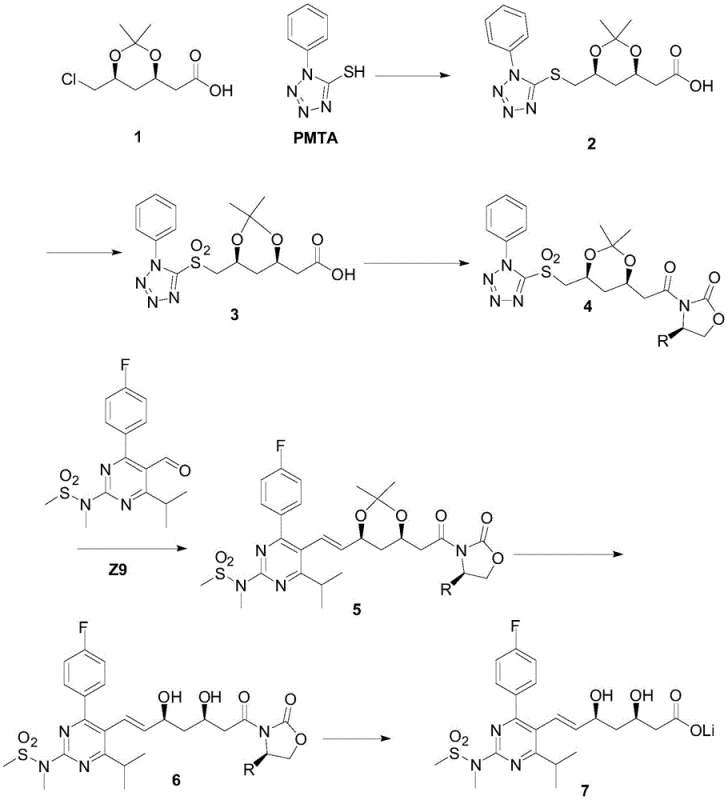
The core chemical innovation of this process resides in the stereoselective construction of the heptenoic acid side chain. The synthesis begins with the installation of an R-oxazolidinone auxiliary onto a sulfone-containing acid precursor (Compound 3) to form Compound 4. This chiral auxiliary exerts profound stereochemical influence during the subsequent Julia olefination step. When Compound 4 reacts with the pyrimidine aldehyde (Z9) in the presence of a base such as potassium tert-butoxide, the reaction proceeds through a rigid transition state that favors the formation of the E-alkene geometry with high fidelity. The bulky phenylsulfonyl group and the chiral environment provided by the oxazolidinone ring work in concert to minimize the formation of the undesired Z-isomer, ensuring that the geometric integrity of the double bond is maintained throughout the synthesis.
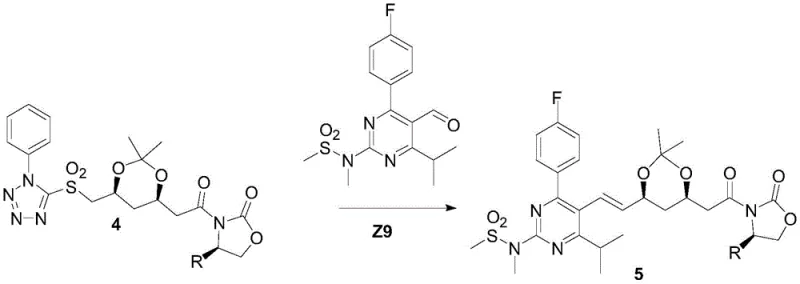
Furthermore, the mechanism ensures precise control over the absolute stereochemistry at the C3 and C5 positions. The auxiliary directs the addition of nucleophiles and subsequent functional group transformations to yield the specific (3R, 5S) configuration required for HMG-CoA reductase inhibition. Following the olefination, the auxiliary is removed under mild acidic hydrolysis conditions to yield Compound 6. This deprotection step is notably gentle, preventing the epimerization or isomerization that often plagues harsher cleavage methods. The result is a highly enantiomerically pure intermediate that requires minimal downstream processing to meet stringent pharmacopeial specifications for chiral purity, directly addressing the R&D concern of impurity profile management in complex statin synthesis.
How to Synthesize Rosuvastatin Calcium Efficiently
The implementation of this synthetic route involves a logical sequence of coupling, oxidation, and salt formation steps designed for scalability. The process initiates with the preparation of the sulfone scaffold, followed by the critical attachment of the chiral controller. Detailed operational parameters, including solvent systems, stoichiometric ratios, and temperature profiles for each transformation, are essential for reproducing the high yields and purity reported in the patent literature. For process chemists looking to adopt this methodology, understanding the nuances of the workup procedures—particularly the extraction and crystallization protocols for the solid intermediates—is key to maximizing throughput. The detailed standardized synthesis steps for this advanced pathway are outlined below.
- Preparation of Sulfone Intermediate: React PMTA with chloro-acetaldehyde derivative followed by oxidation using heptamolybdate and hydrogen peroxide to form the key sulfone precursor.
- Chiral Auxiliary Attachment: Couple the sulfone acid with an R-oxazolidinone using DCC and DMAP to install the Evans chiral auxiliary, creating a solid intermediate.
- Julia Olefination and Final Assembly: Perform the Julia coupling with the pyrimidine aldehyde (Z9) using t-BuOK, followed by hydrolysis and calcium salt formation to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers substantial strategic benefits for procurement and supply chain management. The transition to solid intermediates fundamentally alters the risk profile of the manufacturing process, reducing the likelihood of batch failures due to intermediate instability. By eliminating the need for complex chromatographic purifications associated with oily intermediates, the process significantly lowers the consumption of silica gel and organic solvents, leading to a direct reduction in raw material costs and waste disposal expenses. Moreover, the use of common, non-proprietary reagents such as PMTA and standard oxidants ensures a resilient supply chain that is not vulnerable to the bottlenecks often associated with specialized catalysts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage for chromatography translate into significant operational savings. The ability to purify intermediates via crystallization rather than column chromatography allows for larger batch sizes and higher throughput, effectively lowering the cost of goods sold (COGS) for the final API. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, further contributing to overall manufacturing efficiency and cost competitiveness in the global market.
- Enhanced Supply Chain Reliability: The stability of the solid intermediates (Compounds 5 and 6) allows for extended storage periods and safer transportation between different manufacturing facilities if a multi-site production strategy is employed. This stability mitigates the risk of supply disruptions caused by the degradation of sensitive liquid intermediates. Furthermore, the reliance on commodity chemicals for the synthesis ensures that raw material sourcing remains consistent and predictable, shielding the production schedule from the volatility of the specialty chemical market.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with the majority of steps proceeding at or near room temperature, minimizing the engineering constraints associated with cryogenic reactors. The simplified workup procedures, which involve straightforward extractions and filtrations, are easily adaptable to large-scale reactor vessels. From an environmental standpoint, the reduction in hazardous waste generation and the avoidance of heavy metal residues align with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Rosuvastatin Calcium synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the practical application of the technology. Understanding these aspects is crucial for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source.
Q: Why is Julia Olefination preferred over Wittig reaction for Rosuvastatin synthesis?
A: Julia olefination offers superior E/Z stereoselectivity compared to the Wittig reaction, which often produces mixtures requiring difficult separation. Additionally, the Julia route described in CN111454216B utilizes solid intermediates, simplifying purification and avoiding the handling of unstable oily substances common in Wittig protocols.
Q: What is the role of the Evans prosthetic group in this synthesis?
A: The Evans oxazolidinone auxiliary serves a dual purpose: it controls the stereochemistry at the C3 and C5 positions to ensure the correct (3R, 5S) configuration essential for biological activity, and it converts liquid intermediates into crystalline solids, significantly enhancing process robustness and purity.
Q: Does this process require extreme cryogenic conditions?
A: While the Julia coupling step utilizes moderate cooling (around -40°C), the majority of the synthetic sequence, including the auxiliary attachment and oxidation steps, proceeds efficiently at room temperature. This contrasts with traditional routes that may require harsher low-temperature conditions throughout, thereby reducing energy costs and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the pharmaceutical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in Patent CN111454216B can be seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Rosuvastatin Calcium meets the highest global standards for potency and safety.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain security. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your statin manufacturing operations.
