Scalable Synthesis of High-Purity Corey Lactone via Novel Prins Reaction Route
Scalable Synthesis of High-Purity Corey Lactone via Novel Prins Reaction Route
The pharmaceutical industry's relentless pursuit of efficient prostaglandin synthesis has long centered on the availability of high-quality Corey lactone, a pivotal chiral building block. Patent CN102190642A introduces a transformative preparation method that bypasses traditional bottlenecks by utilizing a direct Prins reaction on a mixture of precursors. This innovation addresses the critical challenge of controlling four contiguous chirality centers on a five-membered ring, a feat that has historically required cumbersome purification steps. By leveraging a robust acid-catalyzed cyclization followed by resin-mediated hydrolysis and precision crystallization, this technology offers a streamlined pathway to Formula F. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the manufacturing of key intermediates like Latanoprost and Bimatoprost, ensuring both chemical integrity and commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Corey lactone has been plagued by the difficulty of obtaining pure starting materials, specifically Compound B. Conventional routes often rely on the use of hazardous reagents such as sodium metal and expensive chiral boranes, necessitating multiple low-temperature reaction steps that are inherently risky and cost-prohibitive for large-scale operations. Furthermore, alternative bio-transformation methods, while effective in generating high enantiomeric excess, suffer from severe downstream processing challenges; the polarity of Compound A and Compound B is so similar that thin-layer chromatography Rf values differ by less than 0.1, making column chromatography separation extremely difficult and solvent-intensive. These legacy methods result in low yields, often below 10% when attempting to crystallize mixtures with near 1:1 ratios of isomers, rendering them unsuitable for the rigorous demands of modern industrial pharmaceutical manufacturing.
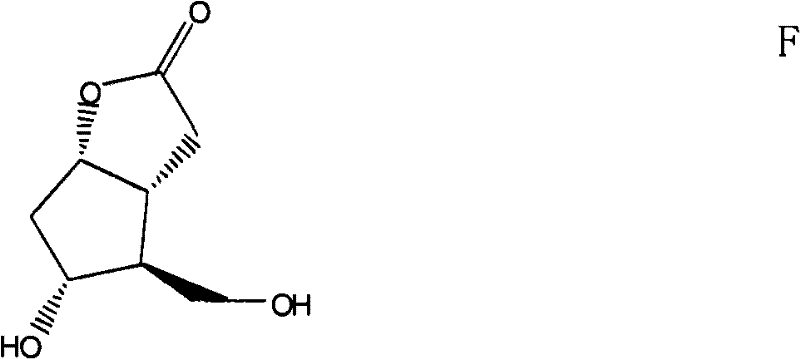
The Novel Approach
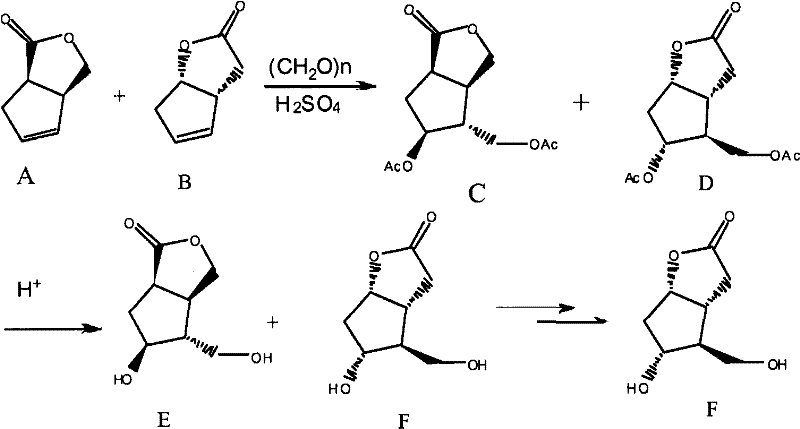
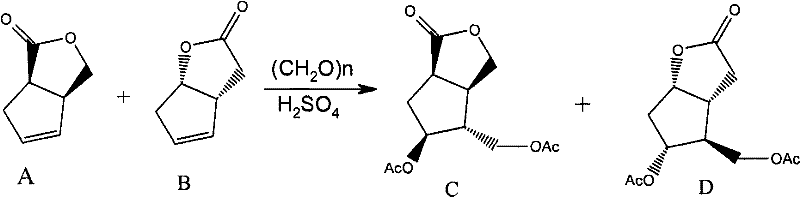
In stark contrast, the methodology disclosed in CN102190642A revolutionizes the workflow by accepting a mixture of Compound A and Compound B (in a ratio of 1:0.8 to 1:1.2) as the direct feedstock. This eliminates the need for the arduous pre-purification of Compound B, allowing the Prins reaction to proceed efficiently on the crude mixture. The process utilizes paraformaldehyde and sulfuric acid to generate a mixture of acetylated intermediates C and D, which are subsequently hydrolyzed using a strong-acid cation exchange resin. This strategic shift not only simplifies the operational complexity but also enhances the overall atom economy. The final purification is achieved through a sophisticated crystallization technique that exploits solubility differences in specific solvent systems, successfully isolating high-purity Formula F even from complex isomeric mixtures.
Mechanistic Insights into Acid-Catalyzed Prins Cyclization
The core of this synthetic breakthrough lies in the Prins reaction mechanism, where the alkene functionality of the starting material undergoes electrophilic addition with formaldehyde in the presence of a strong acid catalyst. Under the optimized conditions of 70-110°C, the oxonium ion intermediate facilitates the formation of the bicyclic framework found in Compounds C and D. The use of sulfuric acid ensures rapid kinetics while maintaining control over the stereochemical outcome, crucial for preserving the integrity of the existing chiral centers. This step is remarkably tolerant of the presence of Compound A, which concurrently reacts to form its corresponding acetylated derivative, thereby preventing the accumulation of unreacted starting material that could complicate downstream processing.
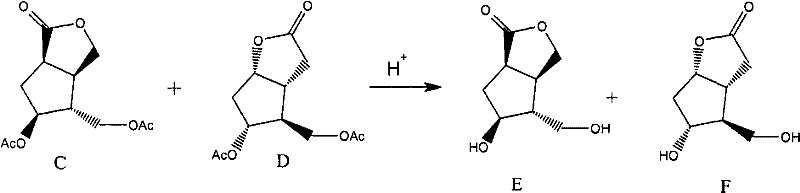
Following the cyclization, the hydrolysis step employs a strong-acid cation exchange resin, such as the 001*7 styrene type, to cleave the acetate groups and reveal the free hydroxyl functionalities in Compounds E and F. This heterogeneous catalysis approach offers distinct advantages over homogeneous acid hydrolysis, including ease of separation and reduced corrosion issues. The resin acts as a solid proton source, facilitating the nucleophilic attack of water on the ester carbonyls without introducing soluble acidic impurities that would require extensive neutralization and washing. This mechanistic elegance ensures that the resulting crude mixture of E and F is clean enough to proceed directly to the critical crystallization stage, minimizing yield losses associated with aqueous workups.
How to Synthesize Corey Lactone Efficiently
The synthesis of Corey lactone via this patented route involves a carefully orchestrated sequence of reaction and purification steps designed for maximum efficiency. The process begins with the Prins cyclization of the A/B mixture, followed by resin-mediated hydrolysis, and concludes with a multi-stage crystallization protocol. Detailed standard operating procedures for temperature control, solvent ratios, and cooling rates are essential to replicate the high purity levels described in the patent embodiments. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized protocol below.
- Mix compounds A and B with paraformaldehyde and perform a Prins reaction under acid catalysis (H2SO4) at 70-110°C to obtain a mixture of compounds C and D.
- Subject the mixture of C and D to hydrolysis using a strong-acid cation exchange resin to generate a crude mixture of compounds E and F.
- Purify the mixture of E and F through a multi-stage crystallization process using ketone/alcohol solvents and ester/ether anti-solvents to isolate pure Formula F.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers profound advantages by fundamentally altering the cost structure of Corey lactone production. The elimination of the need for pure Compound B removes a major cost driver, as the separation of A and B via chromatography typically consumes vast quantities of expensive solvents like n-hexane and methyl tert-butyl ether. By processing the fermentation broth or crude mixture directly, manufacturers can drastically reduce raw material costs and solvent procurement volumes. Furthermore, the replacement of hazardous reagents like sodium metal with stable paraformaldehyde and solid acid resins enhances workplace safety and reduces the regulatory burden associated with handling dangerous chemicals, leading to lower insurance and compliance costs.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by streamlining the synthetic route and removing the expensive purification of intermediate B. The use of solid acid resins for hydrolysis eliminates the need for complex neutralization and extraction steps, reducing labor and utility consumption. Additionally, the high selectivity of the crystallization process minimizes product loss, ensuring that the overall yield is maximized compared to traditional methods that suffer from poor recovery rates during chromatographic separation.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like paraformaldehyde and sulfuric acid rather than specialized chiral reagents, the supply chain becomes more resilient to market fluctuations. The robustness of the Prins reaction conditions allows for flexible production scheduling, as the reaction tolerates a range of temperatures and times without compromising quality. This stability ensures consistent delivery timelines for downstream customers manufacturing prostaglandin analogs, mitigating the risk of production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: The transition from batch chromatography to crystallization-based purification significantly reduces the generation of hazardous organic waste, aligning with green chemistry principles. The use of recyclable ion-exchange resins further minimizes solid waste generation. This environmental profile facilitates easier permitting for capacity expansion, allowing manufacturers to scale from pilot batches to multi-ton commercial production without encountering significant regulatory hurdles related to waste disposal or solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on practical aspects of purity, yield, and operational feasibility. Understanding these details is crucial for technical teams evaluating the adoption of this methodology for their specific manufacturing needs.
Q: Why is the direct use of A/B mixture superior to using pure Compound B?
A: Traditional methods require difficult and expensive purification of Compound B (often via chromatography or complex chemical synthesis). This patent demonstrates that the A/B mixture can undergo the Prins reaction directly without separation, significantly simplifying the workflow and reducing raw material costs.
Q: How does this method improve the purity of Corey Lactone?
A: The process utilizes a specific crystallization protocol involving solvent switching (e.g., acetone to isopropyl acetate) and controlled cooling rates. This effectively separates the target Formula F from the isomeric impurity Formula E, achieving weight ratios of F:E up to 300:1.
Q: Is this process suitable for industrial scale-up?
A: Yes. By replacing hazardous reagents like sodium metal and expensive chiral boranes with paraformaldehyde and solid acid resins, the process eliminates safety risks and simplifies waste treatment, making it highly amenable to commercial production from 100 kgs to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Corey Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of Corey lactone as a cornerstone intermediate for the global prostaglandin market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereochemical requirements of this molecule are met with precision. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the identity and quality of every batch, guaranteeing that our clients receive material that meets the highest pharmacopeial standards for API synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your competitive advantage in the pharmaceutical marketplace.
