Advanced Synthetic Route for Entecavir: Enhancing Purity and Scalability for Global Pharma Supply Chains
Advanced Synthetic Route for Entecavir: Enhancing Purity and Scalability for Global Pharma Supply Chains
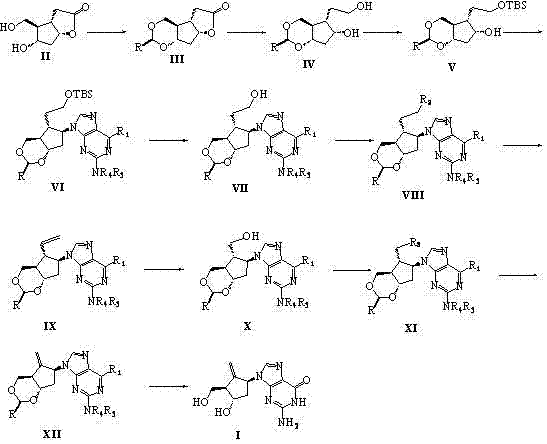
The pharmaceutical landscape for antiviral treatments continues to evolve, driven by the urgent need for cost-effective and high-purity Active Pharmaceutical Ingredients (APIs). A pivotal development in this domain is detailed in patent CN102924454A, which discloses a robust and innovative synthetic method for Entecavir, a potent guanosine nucleoside analog used extensively in the treatment of Hepatitis B Virus (HBV). This technical disclosure represents a significant leap forward from earlier methodologies, addressing critical pain points such as raw material availability, reaction complexity, and overall yield optimization. By leveraging Corey lactone diol as a strategic starting material, the process streamlines the construction of the cyclopentyl core, ensuring that the final product meets the rigorous quality standards demanded by global regulatory bodies. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable supply chain and achieving substantial cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Entecavir has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Early patents, such as US5206244 and subsequent improvements like WO98/09964, often relied on starting materials that were either difficult to source in bulk or required extensive purification protocols before use. These conventional routes frequently suffered from low overall yields due to non-selective reaction conditions, leading to the formation of difficult-to-remove impurities that compromised the final drug substance's purity profile. Furthermore, the reliance on harsh reaction conditions and expensive catalysts in traditional methods escalated the operational expenditure, making the final API prohibitively costly for widespread distribution in developing markets. The complexity of these legacy processes also introduced significant supply chain risks, as any disruption in the availability of niche precursors could halt production lines, thereby increasing lead time for high-purity antiviral agents.
The Novel Approach
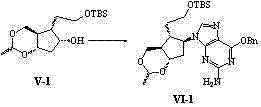
In stark contrast to these legacy challenges, the methodology outlined in CN102924454A introduces a streamlined, economically viable pathway that prioritizes both efficiency and environmental sustainability. The core innovation lies in the utilization of Corey lactone diol, a commercially accessible chiral pool material that inherently possesses the necessary stereochemistry for the Entecavir scaffold. This strategic choice eliminates the need for complex resolution steps early in the synthesis, drastically simplifying the workflow. The process involves a logical sequence of hydroxyl protection, lithium aluminum hydride reduction, and selective silylation, culminating in a highly efficient Mitsunobu coupling with 6-substituted guanine. This novel approach not only enhances the reaction yield but also ensures that the critical stereocenters are preserved with high fidelity throughout the transformation. By adopting this route, manufacturers can achieve a more robust production capability that is less susceptible to the variability often seen in older synthetic strategies.
Mechanistic Insights into Mitsunobu Coupling and Ozonolysis
A deep dive into the reaction mechanism reveals why this specific pathway offers superior control over impurity profiles compared to alternative syntheses. The pivotal step in this sequence is the Mitsunobu reaction, where the protected cyclopentyl intermediate couples with the 6-substituted guanine base. This reaction is renowned for its inversion of configuration, which is meticulously managed in this protocol to ensure the correct (1S,3R,4S) stereochemistry essential for biological activity. The use of triphenylphosphine and azodicarboxylates (DEAD or DIAD) facilitates this bond formation under mild conditions, minimizing the risk of base-catalyzed degradation of the sensitive purine ring. Following the coupling, the removal of the silicon-based protecting group and subsequent elimination reactions are carefully tuned to generate the exocyclic methylene group without affecting the integrity of the glycosidic bond. This precision in mechanistic execution is what allows R&D teams to consistently produce material that passes stringent purity specifications without requiring excessive chromatographic purification.
Furthermore, the oxidative cleavage step utilizing ozone represents a masterclass in functional group manipulation. Conducted at cryogenic temperatures ranging from -78°C to -10°C, the ozonolysis selectively targets the exocyclic double bond generated in the previous elimination step. This transformation is immediately followed by a reductive workup using sodium borohydride or lithium borohydride, which converts the unstable ozonide intermediates into the desired hydroxymethyl functionality. This two-step oxidation-reduction sequence is critical for installing the primary alcohol moiety found in the final Entecavir structure. The specificity of ozone for carbon-carbon double bonds ensures that other sensitive functional groups on the molecule remain untouched, thereby reducing the formation of side products. Such mechanistic clarity provides a solid foundation for process chemists to optimize reaction parameters, ensuring that the transition from laboratory scale to pilot plant operations proceeds with minimal technical friction.
How to Synthesize Entecavir Efficiently
Implementing this synthesis requires a disciplined approach to reaction monitoring and parameter control to maximize the benefits of the patented route. The process begins with the acetalization of Corey lactone diol, followed by reduction and selective protection, setting the stage for the crucial nitrogen heterocycle attachment. Operators must pay close attention to stoichiometry, particularly during the Mitsunobu coupling where the ratio of phosphine to azodicarboxylate can influence the completeness of the reaction. Detailed standard operating procedures (SOPs) should be established for the cryogenic ozonolysis step to ensure safety and reproducibility. While the general flow is straightforward, the devil is in the details regarding quenching procedures and workup protocols which determine the ease of isolation. For a comprehensive breakdown of the specific reagents, temperatures, and durations validated in the patent examples, please refer to the standardized guide below.
- Protect hydroxyl groups of Corey lactone diol using acetals and reduce with Lithium Aluminum Hydride.
- Selectively protect primary hydroxyl with TBSCl and perform Mitsunobu coupling with 6-substituted guanine.
- Execute elimination, ozonolysis cleavage, and final deprotection to yield Entecavir.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the bottom-line concerns of procurement managers and supply chain heads. The shift towards using Corey lactone diol as a starting material leverages a commodity chemical that is produced in significant volumes globally, thereby mitigating the risk of raw material shortages that often plague specialty syntheses. This abundance translates into a more stable pricing structure and reduces the volatility associated with sourcing niche intermediates. Moreover, the simplified reaction sequence means fewer unit operations are required to reach the final API, which inherently lowers the consumption of solvents, energy, and labor hours per kilogram of product. These operational efficiencies accumulate to provide substantial cost savings in API intermediate manufacturing, allowing companies to remain competitive in price-sensitive markets without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available reagents like lithium aluminum hydride and common silyl chlorides significantly lowers the direct material costs. Additionally, the high selectivity of the reactions reduces the burden on downstream purification processes, meaning less solvent is wasted on chromatography and crystallization steps. This lean manufacturing approach ensures that the cost of goods sold (COGS) is optimized, providing a healthier margin structure for the final drug product. By avoiding complex resolution steps early in the synthesis, the process also minimizes the loss of valuable chiral material, further enhancing the overall economic viability of the production campaign.
- Enhanced Supply Chain Reliability: Utilizing a synthesis based on widely available building blocks ensures that the supply chain is resilient against geopolitical or logistical disruptions. Since the key starting material, Corey lactone diol, is a standard item in the fine chemical catalog, lead times for procurement are significantly shorter compared to custom-synthesized precursors. This reliability allows for better inventory planning and reduces the need for safety stock, freeing up working capital. Furthermore, the robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without significant re-validation efforts, ensuring continuity of supply even if primary production facilities face unforeseen downtime.
- Scalability and Environmental Compliance: The patent explicitly highlights the environmental friendliness of the process, a critical factor for modern pharmaceutical manufacturing facing increasingly strict regulatory scrutiny. The reactions are described as easy to control, which implies a lower risk of thermal runaways or hazardous exotherms during scale-up. The use of ozone, while requiring specialized equipment, is a green oxidant that produces oxygen as a byproduct, aligning with sustainability goals. The overall simplicity of the workflow facilitates a smoother transition from kilogram-scale development to multi-ton commercial production, ensuring that the technology can meet global demand without encountering the bottlenecks typical of more convoluted synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific Entecavir synthesis pathway. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of what stakeholders can expect during technology transfer and production. Understanding these details helps in aligning expectations between the technical teams and the commercial leadership, ensuring that the project moves forward with a shared vision of success. For more specific data points regarding batch records or validation protocols, direct engagement with the technical team is recommended.
Q: What are the key advantages of this Entecavir synthesis route compared to prior art?
A: This method utilizes readily available Corey lactone diol and features concise reaction steps with easy control, significantly reducing operational complexity and raw material costs compared to older patents like US5206244.
Q: How does the process ensure high stereochemical purity?
A: The route employs highly stereoselective reactions, particularly the Mitsunobu coupling and specific elimination steps, which maintain the critical (1S,3R,4S) configuration required for antiviral activity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the reaction is easy to control, uses cheap raw materials, and is environmentally friendly, making it highly suitable for commercial scale-up and industrialized production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who understands the intricacies of complex nucleoside chemistry. Our facility is equipped with state-of-the-art reactors and analytical instruments capable of executing the precise temperature controls and anhydrous conditions required for this synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which test every batch against the highest international pharmacopeial standards to guarantee patient safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthetic technology for your Entecavir requirements. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the antiviral market through superior chemical manufacturing excellence.
