Advanced Solvent-Free Synthesis of DOPO: A Technical Breakthrough for High-Purity Flame Retardant Production
Advanced Solvent-Free Synthesis of DOPO: A Technical Breakthrough for High-Purity Flame Retardant Production
The global shift towards environmentally benign materials has placed immense pressure on the chemical industry to replace halogenated flame retardants with safer, high-performance alternatives. In this context, Patent CN102146097A presents a transformative methodology for the preparation of 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide, universally known in the industry as DOPO. This organophosphorus compound has emerged as the gold standard for halogen-free flame retardancy due to its exceptional thermal stability and compatibility with epoxy resins and engineering plastics. The disclosed invention addresses critical bottlenecks in existing manufacturing technologies by introducing a streamlined, solvent-free synthetic route that leverages Lewis acid catalysis and precise thermal control. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to enhance product quality while simultaneously optimizing the cost structure of polymer additive manufacturing.
Unlike conventional approaches that struggle with complex purification steps and hazardous solvent usage, this novel process achieves a yield exceeding 90% and a purity greater than 99% through a series of controlled chemical transformations. The core innovation lies in the strategic manipulation of reaction intermediates, specifically the conversion of 6-chloro-6H-dibenzo[c,e][1,2]oxaphosphorin (CDOP) into a water-soluble salt, which facilitates the removal of metallic catalyst residues prior to the final cyclization. This level of impurity control is paramount for applications in electronic materials and high-performance composites, where trace metal ions can compromise the dielectric properties or long-term reliability of the final product. By integrating this technology, manufacturers can position themselves as a reliable flame retardant intermediate supplier capable of meeting the stringent specifications of top-tier multinational corporations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of DOPO has been plagued by significant technical and economic inefficiencies that hinder large-scale adoption. Traditional protocols, such as those described in earlier literature and patents like DE 19505352Al, typically rely on the use of organic solvents to mediate the reaction between o-phenylphenol and phosphorus trichloride. These solvents not only increase the raw material costs but also necessitate complex recovery and recycling systems to meet environmental regulations, thereby inflating the operational expenditure. Furthermore, the purification of the crude product in these legacy methods often demands high-vacuum molecular distillation, a process that requires specialized, expensive equipment and consumes substantial amounts of energy to maintain the necessary pressure differentials.
Beyond the economic drawbacks, the conventional routes pose serious challenges regarding product quality and process safety. The use of high temperatures and vacuum conditions can lead to thermal degradation of the sensitive phosphorus-containing intermediates, resulting in lower yields and the formation of colored impurities that are difficult to remove. Additionally, the presence of residual zinc or chloride ions from the catalyst system is a persistent issue, as these contaminants can accelerate the degradation of the polymer matrix in downstream applications. The reliance on hazardous organic solvents also introduces significant safety risks, including flammability and toxicity concerns, which complicate plant operations and increase the burden on health and safety compliance teams.
The Novel Approach
The methodology outlined in Patent CN102146097A fundamentally reengineers the production landscape by eliminating the need for organic solvents and high-vacuum distillation entirely. This innovative approach utilizes a neat reaction system where o-phenylphenol and phosphorus trichloride react directly in the presence of a catalytic amount of Lewis acids such as zinc chloride, ferric chloride, or titanium tetrachloride. The absence of solvent simplifies the reaction mixture, allowing for more efficient heat transfer and easier control over the exothermic nature of the phosphorylation step. Following the initial cyclization, the process employs a clever alkaline hydrolysis step that converts the chloro-intermediate into a water-soluble sodium or potassium salt, enabling the physical separation of the catalyst and other insoluble impurities through simple filtration.
This aqueous workup strategy is a game-changer for cost reduction in polymer additive manufacturing, as it replaces energy-intensive distillation columns with standard filtration and crystallization units that are far cheaper to install and maintain. The subsequent acidification step precipitates the hypophosphorous acid intermediate in high purity, which is then subjected to a mild thermal dehydration at 120-140°C to yield the final DOPO product. This final cyclodehydration step proceeds efficiently at atmospheric pressure, removing the need for complex vacuum systems and significantly reducing the risk of equipment failure or leakage. The result is a robust, scalable process that delivers a white crystalline product with superior purity profiles, perfectly suited for high-end applications in electronics and aerospace materials.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization and Dehydration
To fully appreciate the technical sophistication of this synthesis, one must examine the underlying chemical mechanisms that drive the transformation from simple precursors to the complex heterocyclic DOPO structure. The reaction initiates with the activation of phosphorus trichloride by the Lewis acid catalyst, which enhances the electrophilicity of the phosphorus atom, facilitating a nucleophilic attack by the hydroxyl group of o-phenylphenol. This leads to the formation of a P-O bond and the subsequent intramolecular Friedel-Crafts type cyclization, where the aromatic ring attacks the phosphorus center to close the second ring, releasing hydrogen chloride gas. The choice of catalyst, whether it be ZnCl2, FeCl3, or TiCl4, is critical in balancing the reaction rate and minimizing side reactions, with the patent specifying a catalyst loading of merely 1% to 3% by weight relative to the phenol substrate.
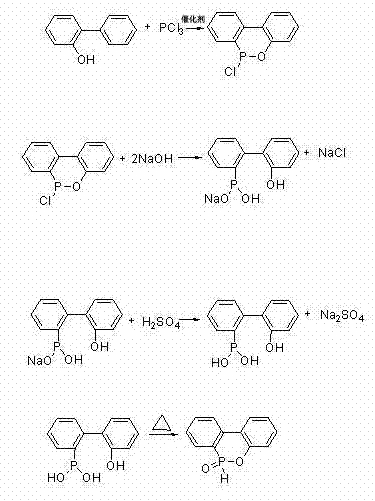
Following the formation of the chloro-intermediate (CDOP), the mechanism shifts to a hydrolytic pathway where the P-Cl bond is cleaved by hydroxide ions in an aqueous alkaline medium. This step is crucial for impurity control, as it converts the organic phosphorus species into a water-soluble phosphinate salt, leaving behind any unreacted catalyst metals or polymeric byproducts as insoluble solids that can be filtered off. The filtrate, now containing the pure phosphinate salt, is then treated with a mineral acid such as sulfuric acid or hydrochloric acid. This acidification protonates the phosphinate anion, causing the 2-hydroxybiphenyl-2-hypophosphorous acid (HPPA) to precipitate out of the solution as a crystalline solid. The final step involves a thermal dehydration where the HPPA undergoes an intramolecular condensation, losing a molecule of water to form the stable P=O double bond characteristic of the DOPO structure, completing the synthesis with high atom economy.
How to Synthesize 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and addition rates to ensure optimal yield and safety. The process begins by charging o-phenylphenol and the chosen Lewis acid catalyst into a reactor equipped with a reflux condenser and dropping funnel, followed by heating to melt the phenol. Phosphorus trichloride is then added dropwise over a period of 8 to 12 hours while maintaining the temperature between 160°C and 180°C to manage the evolution of HCl gas. Once the reaction is complete, excess phosphorus trichloride is distilled off under reduced pressure, and the resulting intermediate is subjected to alkaline hydrolysis using a 10% to 30% sodium hydroxide solution. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and crystallization parameters, are provided in the guide below to ensure reproducibility.
- React phosphorus trichloride with o-phenylphenol using a Lewis acid catalyst (e.g., ZnCl2) at 160-180°C to form the chloro-intermediate.
- Perform alkaline hydrolysis on the intermediate using sodium hydroxide solution to generate a soluble salt, followed by filtration to remove catalyst residues.
- Acidify the filtrate with sulfuric or hydrochloric acid to precipitate 2-hydroxybiphenyl-2-hypophosphorous acid crystals.
- Heat the dried crystal powder to 120-140°C to induce dehydration and cyclization, yielding the final blocky DOPO product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling advantages that extend far beyond simple chemical yield improvements. The most immediate impact is seen in the drastic simplification of the production infrastructure, which translates directly into lower capital expenditure and reduced operational overheads. By eliminating the requirement for high-vacuum molecular distillation equipment and large-scale solvent recovery systems, manufacturers can significantly reduce their facility investment costs. This leaner operational model allows for more competitive pricing strategies, making high-purity DOPO more accessible to downstream polymer compounders who are constantly seeking cost reduction in polymer additive manufacturing without compromising on performance standards.
- Cost Reduction in Manufacturing: The elimination of organic solvents from the entire process workflow removes a major variable cost component associated with raw material procurement and waste disposal. In traditional methods, the purchase, storage, and eventual incineration or recycling of tons of organic solvents represent a substantial financial burden. Furthermore, the energy savings achieved by avoiding high-vacuum distillation and operating the final dehydration step at atmospheric pressure contribute to a lower utility bill per kilogram of product. These cumulative efficiencies allow for a more robust margin structure, enabling suppliers to offer better value to their customers while maintaining profitability in a volatile market.
- Enhanced Supply Chain Reliability: The simplicity of the equipment required for this synthesis means that production lines are less prone to mechanical failures and unplanned downtime. High-vacuum systems and complex solvent distillation columns are notorious for requiring frequent maintenance and specialized spare parts, which can disrupt supply continuity. In contrast, the reactors and filtration units used in this solvent-free process are standard industry assets that are easy to maintain and operate. This reliability ensures a consistent flow of goods to customers, reducing lead time for high-purity flame retardant intermediates and allowing downstream manufacturers to optimize their own inventory levels with greater confidence.
- Scalability and Environmental Compliance: As regulatory pressures regarding volatile organic compound (VOC) emissions continue to tighten globally, a solvent-free process provides a distinct strategic advantage. This methodology inherently minimizes the generation of hazardous waste and airborne pollutants, simplifying the permitting process and reducing the risk of environmental fines. The ability to scale up production without the need for massive expansions in solvent handling infrastructure makes this technology ideal for meeting growing market demand. Companies adopting this route demonstrate a commitment to sustainable chemistry, which is increasingly becoming a prerequisite for doing business with major multinational corporations in the electronics and automotive sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DOPO synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational benefits. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new DOPO synthesis method improve product purity compared to traditional distillation?
A: Traditional methods often rely on high-vacuum molecular distillation which can lead to thermal degradation and incomplete separation of impurities. This patented process utilizes a unique alkaline hydrolysis and acidification sequence that allows for the physical removal of catalyst residues and ionic impurities via filtration before the final cyclization step, consistently achieving purity levels exceeding 99% without the need for complex distillation equipment.
Q: What are the primary cost drivers eliminated in this solvent-free manufacturing route?
A: The most significant cost reduction comes from the complete elimination of organic solvents throughout the entire synthesis pathway. Conventional processes require large volumes of solvents for reaction media and subsequent recovery systems, which incur high raw material costs, energy consumption for distillation, and waste treatment expenses. By operating in a neat phase and utilizing aqueous workups, this method drastically simplifies the downstream processing infrastructure.
Q: Is this process scalable for industrial production without specialized high-vacuum equipment?
A: Yes, the process is specifically designed for industrial scalability by avoiding the need for high-vacuum apparatus during the critical dehydration and cyclization stages. The final conversion of the hypophosphorous acid intermediate to DOPO occurs effectively at atmospheric pressure within a temperature range of 120-140°C, allowing manufacturers to utilize standard reactor vessels and reducing both capital expenditure and maintenance complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DOPO Supplier
The technical potential of this solvent-free synthesis route is immense, offering a pathway to produce flame retardant intermediates that meet the highest standards of purity and consistency. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of materials regardless of market fluctuations. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instruments to verify stringent purity specifications, guaranteeing that every batch of DOPO we deliver performs flawlessly in your final polymer applications. We understand that in the fast-paced world of electronic materials and engineering plastics, consistency is key to maintaining your brand reputation.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this optimized process. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with us, you gain access not just to a chemical product, but to a comprehensive supply chain solution designed to enhance your competitiveness in the global market.
